Endometriosis is associated with pelvic pain, dysmenorrhea, dyspareunia, infertility, and lower health-related quality of life. Unfortunately, it takes an average of 8 to 10 years between initial presentation of symptoms and diagnosis of this often chronic and progressive condition. Nurse practitioners in primary care settings who have comprehensive knowledge about endometriosis are key to early diagnosis and initiation of treatment. Attention to quality of life, patient education, and shared decision making is essential.
Continuing education approval: This activity has been evaluated and approved by the Continuing Education Approval Program of the National Association of Nurse Practitioners in Women’s Health (NPWH) for 1.0 continuing education contact hours including 0.50 contact hours of pharmacology content now through December 31, 2024.
Intended audience: This continuing education (CE) activity has been designed to meet the educational needs of nurse practitioners and other clinicians who provide healthcare for women.
Goal statement: Nurse practitioners and other clinicians who provide healthcare for women will increase their knowledge about the diagnosis and management of endometriosis.
Needs assessment: Endometriosis is associated with pelvic pain, dysmenorrhea, dyspareunia, infertility, and lower health-related quality of life. It has a prevalence rate of 10% in reproductive-age women and is often chronic and progressive. Unfortunately, it takes an average of 8 to 10 years between initial presentation of symptoms and diagnosis. An understanding of recommended assessment, diagnostic criteria, and management options for endometriosis is essential to provide care that is appropriate and attentive to individual needs and desires.
Educational objectives: At the conclusion of this educational activity, participants should be able to:
- Identify clinical indicators and imaging studies helpful in the diagnosis of endometriosis.
- Discuss a patient-centered approach for relief from symptoms and improved quality of life.
- Describe indications, mechanism of action, efficacy, adverse events, and contraindication for pharmacologic options in treating endometriosis.
The authors have no actual or potential conflicts of interest in relation to the contents of this article, but as journal editor in chief, Dr. Kelsey did not participate in any aspects of peer review or decision on approval of this manuscript.
Disclaimer: Participating faculty members determine the editorial content of the CE activity; this content does not necessarily represent the views of NPWH. This content has undergone a blinded peer review process for validation of clinical content. Although every effort has been made to ensure that the information is accurate, clinicians are responsible for evaluating this information in relation to generally accepted standards of care and integrating the information in this activity with that of established recommendations of other authorities, national guidelines, and individual patient characteristics.
Commercial support: This activity is supported by an educational grant from Myovant Sciences and Pfizer Inc.
Successful completion of the activity: Successful completion of this activity, J-22-06, requires participants to do the following:
- 1. “Sign in” at the top right-hand corner of the page pathlms.com/npwh/courses/48068 if you have an NPWH account. You must be signed in to receive credit for this course. If you do not remember your username or password, please follow the “Forgot Password” link and instructions on the sign-in page. If you do not have an account, please click on the “Create an Account.”*
- Read the learning objectives, disclosures, and disclaimers.
- Study the material in the learning activity during the approval period (now through December 31, 2024).
- Complete the post-test and evaluation. You must earn a score of 70% or higher on the post-test to receive CE credit.
- Print out the CE certificate after you have successfully passed the post-test and completed the evaluation.
*If you are an NPWH member, were once a member, or have completed CE activities with NPWH in the past, you have a username and password in our system. Please do not create a new account. Creation of multiple accounts could result in loss of CE credits as well as other NPWH services. If you do not remember your username or password, either click on the “Forgot Username” or “Forgot Password” link or call the NPWH office at (202) 543-9693, ext. 1.
A panel of six experienced women’s health nurse practitioners convened for two roundtable discussions to share insights and expertise on essential aspects of endometriosis care. They agreed that key components were being astute at identifying at-risk patients for early diagnosis and treatment, conducting a thorough assessment, individualizing evidence-based treatments to meet patient goals, and providing attentive follow-up to assess treatment efficacy and patient satisfaction. The impact of endometriosis on quality of life underscored their discussion. They agreed that nurse practitioners (NPs) have an important role in providing adolescents and adult women with supportive communication. Knowledge about menstruation is needed so that patients understand what is normal and what symptoms suggest the need to see a healthcare provider. This article provides an overview of endometriosis, reviews current modalities for assessment and diagnosis, discusses medication therapies, and addresses the importance of quality of life, patient education, and shared decision making.
Endometriosis overview
Endometriosis is an estrogen- dependent gynecologic disorder characterized by the presence of endometrial tissue outside the endometrial cavity. Endometriotic lesions typically occur on the ovaries, pelvic peritoneum, and uterosacral ligaments. Lesions also can be identified on the bladder, ureter, or extra pelvic sites.1 Endometriosis is associated with pelvic pain, dysmenorrhea, dyspareunia, and infertility.1,2 The cause of endometriosis is not clearly understood and likely multifactorial.1,2 Possible risk factors include early age at menarche, low body mass index, short menstrual cycles, increased menstrual flow, nulliparity, and family history.1,3 Endometriosis has a strong familial component. The individual with a first-degree relative with endometriosis has a 7- to 10-fold increased risk of having the condition.1 Endometriosis is a disorder primarily affecting reproductive-age females with a prevalence rate of 10%. Estimates vary but up to 21% of women hospitalized for pain and 50% of women with infertility are diagnosed with endometriosis.3 It is the most common cause of secondary dysmenorrhea in adolescents.4 Although endometriosis can remain stable or improve over time, it is often a chronic and progressive condition.5
Studies have demonstrated that endometriosis is associated with lower health-related quality of life (HRQL). Women with symptomatic endometriosis have reported significant disruptions in everyday activities; negative impact on sexual, social, and emotional wellbeing; poor sleep quality; loss of sense of control; feelings of uncertainty with diagnostic delays; increased stress and anxiety; and feelings of lack of support or understanding.6–9 Women who experience dyspareunia report feelings of being an inadequate partner, guilt and shame, low self-esteem, fear of pain, and willingness to endure pain to please the partner or achieve pregnancy.10 The potential for infertility caused by endometriosis also impacts HRQL.5,9
Delay in diagnosis remains problematic. It takes an average of 8 to 10 years between initial presentation of symptoms and diagnosis.1,5 The delay in diagnosis is longer in adolescents, averaging 12 years from onset of symptoms.11 Such delays may in part be attributed to a lack of knowledge about menstruation in general and specifically endometriosis, perceptions that pelvic pain is normal, and embarrassment in discussing symptoms with healthcare providers.5 The delay in diagnosis also is attributed to healthcare providers’ lack of knowledge about the disease and its possible symptoms, including the overlap of symptoms with other pain-associated syndromes. Delays in the diagnosis and treatment of endometriosis also occur when healthcare providers are hesitant to establish the diagnosis based on clinical findings without direct laparoscopic confirmation and tissue biopsy of visible lesions.1,9,12,13 Delays in diagnosis and initiation of treatment increase negative symptoms and can result in a disease state more difficult to treat.5
Qualitative studies have demonstrated that diagnosis of the disease can result in feelings of relief, legitimation, and empowerment. A diagnosis of endometriosis validates a woman’s symptoms and allows for the start of targeted treatment.9 Early diagnosis and treatment have the potential to avoid central sensitization, a complicated response causing pain hypersensitivity. In addition, early diagnosis and treatment reduces chronic pain and infertility and changes the trajectory of patients’ lives.13
Assessment and diagnosis
To prevent delays in diagnosis and treatment, the inclusion of a thorough menstrual history to screen for symptoms and risk factors is essential. The patient may not always report such clues unless asked, and it provides an excellent opportunity for the NP to share information about normal menses. An important part of the menstrual history is to ask about a family history of endometriosis or painful periods. The sexual, obstetric, and pelvic-abdominal surgery history and review of systems can further identify symptoms and other factors that trigger the need for further investigation.
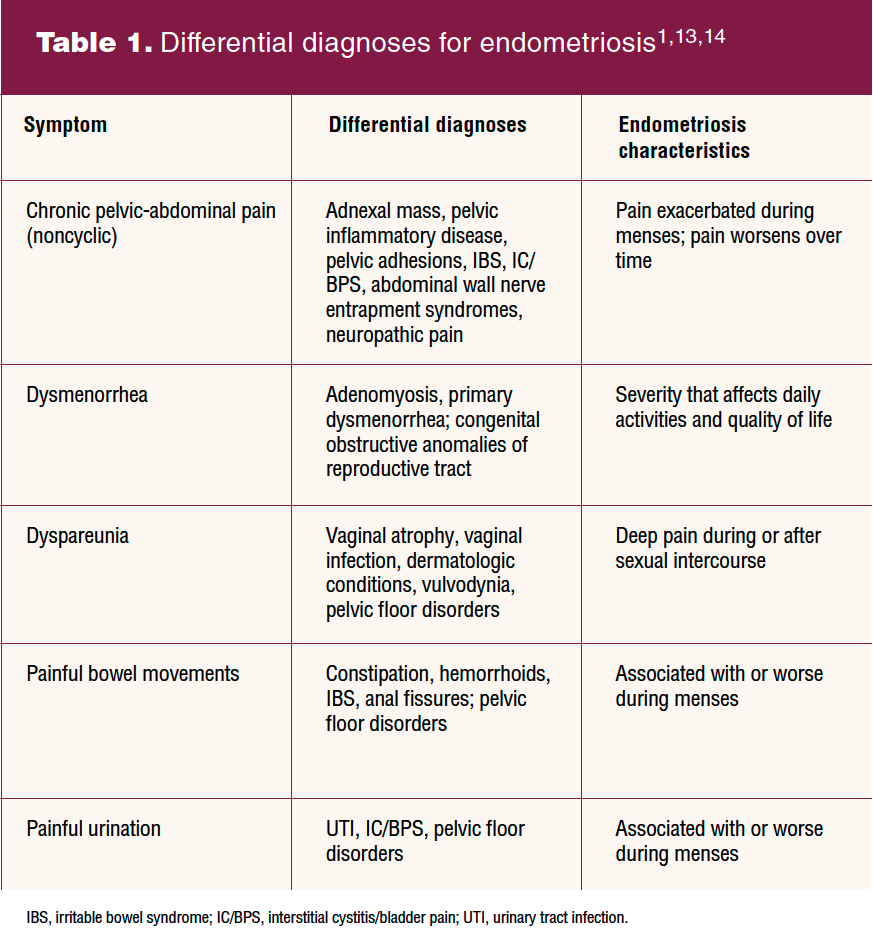
Patients who present with dysmenorrhea that negatively impacts daily activities and quality of life, dyspareunia with deep penetration, cyclic gastrointestinal or genitourinary symptoms, chronic pelvic pain, or infertility in association with one or more these symptoms should alert the NP to include endometriosis in the differential diagnosis.5 As these symptoms are not disease specific, it is essential to consider other diagnoses that may be a primary or contributing etiology of the patient’s pain. Table 1 displays differential diagnoses for consideration.1,13,14
An abdominal and pelvic examination may reveal findings suggestive of endometriosis and aid in eliminating other etiologies and coexisting pain syndromes. Physical examination findings consistent with endometriosis include bluish lesions on the cervix or upper vagina, tenderness and/or palpable nodules in the uterosacral ligaments or posterior cul-de-sac, and a fixed retroverted uterus.1,15 It is important to note that the individual with endometriosis may or may not have physical examination findings indicative of endometriosis. Adolescents who have not had sexual intercourse may or may not be able to tolerate a vaginal examination. Discussion and informed consent are critical.
In patients with suspected endometriosis, further diagnostic steps should be considered even if the clinical examination is normal.5,12,15 Laparoscopy was once regarded as the diagnostic gold standard for endometriosis. With advances in the quality and availability of imaging modalities, ultrasound can be utilized with less risk and cost to patients.12 Transvaginal ultrasound (TVU) is an effective technique for confirming or excluding the diagnosis of endometriosis and also can aid in detecting other causes for pelvic pain such as adenomyosis.1,5,12,15 The sensitivity of TVU, however, is variable depending on the location and size of endometrial lesions.12 A negative finding on ultrasound does not exclude endometriosis, particularly superficial peritoneal disease.12 If a TVU is not appropriate due to age, inability to tolerate vaginal examinations, or other factors, transabdominal ultrasound or magnetic resonance imaging (MRI) may be considered.5,12 When imaging with ultrasound or MRI is negative, empiric medical treatment is unsuccessful, or if it is the patient’s preference, laparoscopy is recommended for the diagnosis and treatment of suspected endometriosis.5,12,15
Treatment of endometriosis-related pain
Key to successful treatment of endometriosis-related pain is a patient-centered approach that meets patient goals for relief from symptoms and improvement of HRQL. Changes should be made as needed based on patient response to treatment and personal and reproductive goals. All treatment options should be discussed with review of the benefits, risks, and potential side effects. Patients should be informed that both medical and surgical treatments may offer complete or partial relief of pain symptoms. However, symptoms often recur after discontinuation of hormonal suppressive therapy or a period of time after surgical treatment.12,15 Baseline and repeated assessment of pain levels and HRQL aid in the ongoing determination of treatment effectiveness. The frequency and type of follow-up should be individualized based on symptom severity. Pain levels can be documented using a scale of 0 to 10. What may be more important than assigning a number, however, is to talk with the patient about how pain is affecting all aspects of their daily life.1 The EHP-30 [Endometriosis Health Profile-30] has demonstrated reliability and validity for assessment of HRQL.16 The generic quality-of-life questionnaire SF-36 also has been validated for use in endometriosis.17 Individual circumstances and priorities should guide referrals for counseling and other support services.
The most commonly prescribed pharmacologic treatments for endometriosis include nonsteroidal anti-inflammatory drugs (NSAIDs) and drugs that modify the hormonal environment either by suppressing ovarian activity or acting directly on steroid receptors and enzymes found in the lesions. These include combined hormonal contraceptives (CHCs), progestins, gonadotropin- releasing hormone (GnRH) agonists, and GnRH antagonists. Aromatase inhibitors, although not approved by the US Food and Drug Administration for this indication, are an off-label option for severe endometriosis-related pain refractory to other medical or surgical treatments. They may be used in combination with hormonal medications because they suppress extraovarian estrogens.12
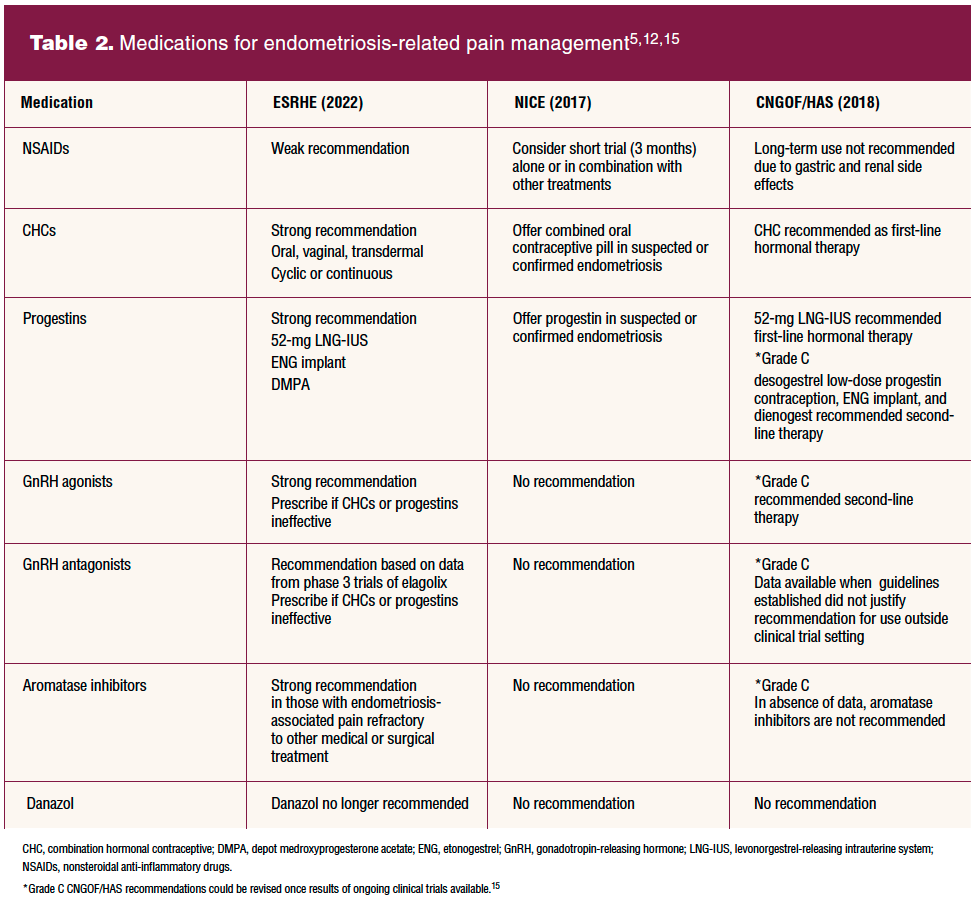
Three European-based groups have provided guidelines for the diagnosis and management of endometriosis based on scientific reviews of evidence regarding efficacy and safety. These groups are the National Institute for Health and Care Excellence (NICE), European Society of Human Reproduction and Embryology (ESHRE), and the French National Authority for Health and the French College of Gynecologists and Obstetricians (CNGOF/HAS).5,12,15 All three groups note that the aim of clinical practice guidelines is to aid healthcare professionals in everyday clinical decisions about appropriate and effective care, not precluding clinical judgment and individual patient preference. Table 2 compares their recommendations. It is important to note that the timing of the NICE and CNGOF/HAS review of evidence preceded some of the most recent available study results on oral GnRH antagonists.5,12,15
The NP in a primary care practice can initiate first-line therapies (NSAIDs, CHCs, progestins) when the patient exhibits mild-to-moderate symptoms consistent with endometriosis. First-line therapies are those combining known efficacy with good safety profiles, minimal side effects, ease in administration, and lower cost. For patients who have previously tried one or more of the first-line therapies without appreciable pain relief or with unacceptable side effects, other options should be considered. These patients may benefit from second-line medication therapies such as GnRH agonists, GnRH antagonists, and aromatase inhibitors. The use of these medications generally requires laparoscopic confirmation of endometriosis. Second-line medication therapies have more potential for side effects, related loss of bone mineral density (BMD), and are more expensive than first-line medications. They should, however, be considered options for individuals for whom first-line medications have not provided satisfactory pain relief.
NSAIDs
Nonsteroidal anti-inflammatory drugs are a commonly employed first-line therapy partly because they are readily available and relatively inexpensive. They work by inhibiting cyclooxygenase-mediated prostaglandin production. Prostaglandins are mediators of localized inflammation and are considered to be responsible for dysmenorrhea.2 Evidence does support the use of NSAIDs as an effective treatment for mild-to-moderate primary dysmenorrhea.18 Nevertheless, available evidence is limited to support the use of NSAIDs for endometriosis-related pain management.19 Additionally, these medications do not suppress hormonal-dependent endometrial growths.
There are minimal risks to the use of NSAIDs although they do have side effects, most specifically gastrointestinal side effects.12 Because they have a general anti-inflammatory effect, NSAIDs can be used in conjunction with hormonal therapies. For women who are attempting pregnancy, this may be the only medication option for pain relief.
Combined hormonal contraceptives
Data from two systematic reviews have demonstrated that CHC use results in statistically significant reductions in endometriosis- related pain and improved quality of life.20,21 Although more data are available on combined oral contraceptives, vaginal and transdermal CHCs demonstrated efficacy in endometriosis-related pain reduction in both systematic reviews. A continuous-use regimen appears to be more efficacious in regard to dysmenorrhea, with nonsignificant differences between continuous and cyclic regimens for chronic pelvic pain and dyspareunia.22
Progestins
A systematic review including progestin-only pills, the 52-mg levonorgestrel intrauterine system (LNG-IUS), etonogestrel (ENG)- releasing subdermal implant, and depot medroxyprogesterone acetate found all to be effective in reducing endometriosis-related pain.19 A recent randomized controlled trial reported that both the ENG implant and LNG-IUS significantly reduced endometriosis- related pain, dysmenorrhea, and chronic pelvic pain.23
GnRH agonists
Gonadotropin-releasing hormone agonists have demonstrated efficacy in relieving endometriosis-related pain.5,12,15,24 They bind to GnRH receptors on the anterior pituitary, causing a down-regulation of the pituitary-ovarian axis and profound but reversible hypoestrogenism.24
Commonly used GnRH agonists that are FDA approved for up to 12 months for treatment of endometriosis-related pain are goserelin SC, leuprolide IM, and nafarelin depot via nasal spray.1 There has been no demonstrated difference in efficacy or reported side effects related to route of administration.24 The most reported side effects are vaginal dryness, hot flushes, headaches, and joint pain.1 The use of GnRH agonists is contraindicated during pregnancy.
Reduction of BMD is a major concern with GnRH agonist treatment continuing for longer than 6 months. The addition of add-back therapy prevents bone loss and does not affect the efficacy of the GnRH agonist treatment.25 Commonly used add-back regimens include the progestins medroxyprogesterone acetate and norethindrone, a low-dose estrogen-progestin combination, and bisphosphonates.25 Considering the possible impact on BMD, GnRH agonists should only be used after careful consideration in young women and adolescents if first-line treatments are ineffective.12
GnRH antagonists
Two oral GnRH antagonists have been FDA approved for the management of moderate-to-severe pain associated with endometriosis. Elagolix, which is available in two dosages, was approved in 2018.26 Relugolix combined with estradiol and norethindrone acetate was approved in 2022.27 GnRH antagonists suppress follicle-stimulating hormone, luteinizing hormone, and estrogen and may have a quicker symptom relief profile than GnRH agonists as well as the advantage of oral administration. The most frequently reported side effects across both medications include hot flushes, night sweats, and irregular bleeding.26,27 The use of GnRH antagonists is contraindicated in pregnancy.
Like GnRH agonists, reduction of BMD is a concern with long-term GnRH antagonist use. Prescribing information recommends limiting duration of use to 24 months for elagolix 150 mg and relugolix combined with estradiol and norethindrone acetate.26,27 The higher dose of elagolix (200 mg twice daily), while more effective in pain relief, is associated with more bone loss than the lower dose with recommended use limited to 6 months.26 Considering the possible impact on BMD, GnRH antagonists should only be used after careful consideration in young women and adolescents if first-line treatments are ineffective.12
Other considerations
Neuromodulators such as tricyclic antidepressants, selective reuptake inhibitors, and anticonvulsants have shown promise as a component of the management of endometriosis-related pain but are associated with dose-limiting side effects. Pelvic physical therapy and cognitive behavioral therapy also may be of benefit to some women.2
It has been suggested that pain syndromes such as endometriosis may interact with other pain syndromes (eg, irritable bowel syndrome, fibromyalgia, migraine headaches) and that this interaction may feature increased pain sensitivity stemming from the effects of nociceptive inputs on the nervous system. Perceptual responses to pain can become exaggerated, prolonged, and widely spread (central sensitization).28 If central sensitization is suspected, further pain evaluation and pain management is recommended. This often requires a multidisciplinary team that includes a pain specialist, physical therapist, psychologist, and primary healthcare provider.15
For patients who have undergone laparoscopic surgery, postoperative use of a CHC or progestin is recommended to prevent the risk of disease recurrence when pregnancy is not desired.12,15 Hormone replacement therapy can be offered to postmenopausal women who have undergone surgical treatment, although endometriosis symptoms could recur.12,15
Endometriosis-related infertility management should consider current pain as well as the results of the pretreatment infertility evaluation.5,15 The benefits and risks of surgery prior to assisted reproductive therapy should be discussed to include the potential negative impact on ovarian reserve.12 There is insufficient evidence to support extended GnRH agonist, CHC, or progestin use prior to assisted reproductive therapies to increase live birth rates.12,15
Implications for NP practice
Nurse practitioners can play a key role in the early diagnosis and treatment of endometriosis. Comprehensive knowledge of endometriosis that includes risk factors, symptoms, potential physical examination findings, and current imaging modalities empower the NP to clinically diagnose endometriosis and to recognize when consultation or collaboration is beneficial. Special considerations should be given to adolescent assessment. Providing patient education about normal menses and symptoms that require healthcare attention can be incorporated into the patient history. A patient-centered approach to endometriosis treatment considers the patient’s individual goals, HRQL, and follow-up to evaluate for any needed adjustment based on treatment efficacy, patient satisfaction, and change in reproductive goals.
Key words: endometriosis, endometriosis-related pain, pelvic pain, dysmenorrhea, dyspareunia
Womens Healthcare. 2022;10(6):4-10. doi: 10.51256/WHC122208
References
1 Falcone T, Flyckt R. Clinical management of endometriosis. Obstet Gynecol. 2018;131(3):557-571.
2 Zondervan KT, Becker CM, Missmer SA. Endometriosis. N Engl J Med. 2020;382(13):1244-1256.
3 Shafrir AL, Farland LV, Shah DK, et al. Risk for and consequences of endometriosis: a critical epidemiologic review. Best Pract Res Clin Obstet Gynaecol. 2018;51:1-15.
4 American College of Obstetricians and Gynecologists. Committee opinion no. 760: dysmenorrhea and endometriosis in the adolescent. Obstet Gynecol. 2018;132(6):e249-258.
5 National Institute for Health and Care Excellence. Endometriosis: diagnosis and management. NICE guideline. September 6, 2017. www.nice.org.uk/guidance/ng73.
6 Pope CJ, Sharma V, Sharma S, Mazmanian D. A systematic review of the association between psychiatric disturbances and endometriosis. J Obstet Gynaecol Can. 2015;37(11):1006-1015.
7 Marinho MCP, Magalhaes TF, Fernandez LFC, et al. Quality of life in women with endometriosis: an integrative review. J Womens Health (Larchmt). 2018;27(3):399-408.
8 Soliman AM, Coyne KS, Zaiser E, et al. The burden of endometriosis symptoms on health-related quality of life in women in the United States: a cross-sectional study. J Psychosom Obstet Gynaecol. 2017;38(4):238-248.
9 Rush G, Misajon R. Examining subjective wellbeing and health-related quality of life in women with endometriosis. Health Care Women Int. 2018;39(3):303-321.
10 Faccine F, Buggio L, Dridi D, et al. The subjective experience of dyspareunia in women with endometriosis: a systematic review with narrative synthesis of qualitative research. Int J Environ Res Public Health 2021;18(22):12112.
11 Geysenbergh B, Dancet EAF, D’Hooghe T. Detecting endometriosis in adolescents: why not start from self-report screening questionnaires for adult women? Gynecol Obstet Invest. 2017;82(4):322-328.
12 European Society of Human Reproduction and Embryology. ESHRE guideline endometriosis. February 2, 2022. https://www.eshre.eu/Guidelines-and-Legal/Guidelines/Endometriosis-guideline.
13 Agarwal SK, Chapron C, Giudice LC, et al. Clinical diagnosis of endometriosis: a call to action. Am J Obstet Gynecol. 2019;220(4):354.e1-354.e12.
14 American College of Obstetricians and Gynecologists. Practice bulletin no. 218: chronic pelvic pain. Obstet Gynecol. 2020;135(3):e98-e109.
15 Collinet P, Fritel X, Revel-Delhom M, et al. Management of endometriosis CNGOF/HAS clinical practice guidelines – short version. J Gynecol Obstet Hum Reprod. 2018;47(7):265-274.
16 Pokrzywinski R, Soliman AM, Chen J, et al. Responsiveness evaluation and recommendation for responder thresholds for Endometriosis Health Profile-30: analysis of two-phase III clinical trials. J Womens Health (Larchmt). 2020;29(2):253-261.
17 Stoll DE, Wasiak R, Kreif N, et al. Validation of the SF-36 in patients with endometriosis. Qual Life Res. 2014;23(1):103-117.
18 Marjoribanks J, Ayeleke RO, Farquhar C, et al. Nonsteroidal anti-inflammatory drugs for dysmenorrhoea. Cochrane Database Syst Rev. 2015(7):CD001751.
19 Brown J, Crawford TJ, Allen C, et al. Nonsteroidal anti-inflammatory drugs for pain in women with endometriosis. Cochrane Database Syst Rev. 2017;1(1):CD004753.
20 Grandi G, Barra F, Ferrero S, et al. Hormonal contraception in women with endometriosis: a systematic review. Eur J Contracept Reprod Health Care. 2019;24(1):61-70.
21 Jensen JT, Schlaff W, Gordon K. Use of combined hormonal contraceptives for the treatment of endometriosis-related pain: a systematic review of the evidence. Fert Steril. 2018;110(1):137-152.e1.
22 Muzii L, Di Tucci C, Achilli C, et al. Continuous versus cyclic oral contraceptives after laparoscopic excision of ovarian endometriomas: a systematic review and metaanalysis. Am J Obstet Gynecol. 2016;214(2):203-211.
23 Margatho D, Carvalho NM, Bahamondes L. Endometriosis-associated pain scores and biomarkers in users of the etonogestrel-releasing subdermal implant or the 52-mg levonorgestrel-releasing intrauterine system for up to 24 months. Eur J Contracept Reprod Health Care. 2020;25(2):133-140.
24 Brown J, Pan A, Hart RJ. Gonadotrophin-releasing hormone analogues for pain associated with endometriosis. Cochrane Database Syst Rev. 2010(12):CD008475.
25 Wu D, Hu M, Hong L, et al. Clinical efficacy of add-back therapy in treatment of endometriosis: a meta-analysis. Arch Gynecol Obstet 2014;290(3):513-523.
26 Orilissa [elagolix]. US Food and Drug Administration approved product information. Revised February 2021. https://www.rxabbvie.com/pdf/orlissa_pi.pdf.
27 Myfembree [relugolix, estradiol, and norethindrone]. US Food and Drug Administration approved product information. Revised August 2022. https://www.myovant.com/wp-content/uploads/2022/08/Approved-MYFEMBREE-PI-and-PPI_05August2022.pdf.
28 Woolf CJ. Central sensitization: implications for the diagnosis and treatment of pain. Pain. 2011;152(suppl 3):S2-S15.