Vaginal estrogen can improve postmenopausal vaginal dryness as well as a variety of related lower urinary tract symptoms and improve a woman’s quality of life. Despite the fear and negativity surrounding the use of vaginal estrogen, it is safe and effective for the treatment of vaginal dryness in most postmenopausal women. Education is key when prescribing vaginal estrogen so that the woman is confident and comfortable with its use.
Menopause is defined as the final menstrual period and reflects loss of ovarian follicular function.1 Menopause is confirmed when a woman has not had a menstrual period for 12 consecutive months. The average age of menopause in the United States is 51. The final step in the midlife journey is postmenopause. Postmenopause encompasses the entire period of one’s life after menopause. The average US woman will spend 30 years in the postmenopausal state, which equates to over one third of a woman’s life.1
Estrogens and Their Metabolism
Longer estrogen exposure improves cognitive function
Every life stage is a new transition, and the postmenopausal years are no exception. Unfortunately, many postmenopausal women experience unwelcome health changes as they enter this life transition. Among the changes is genitourinary syndrome of menopause (GSM), affecting up to 84% of postmenopausal women.2 The symptoms of GSM can significantly affect a woman’s quality of life (QOL), specifically their sexual health. Despite the prevalence of GSM and the impact it has on a woman’s QOL, only 50% have used any type of therapy.2 This article provides readers with an overview of the physiologic changes in the vagina that occur with menopause and associated genitourinary symptoms. The author then describes benefits and barriers and recommended use of various formulations of vaginal estrogens for treatment and addresses safety concerns.
Physiology
The cytology of a premenopausal woman’s vaginal tissue is characterized by wide columns of superficial epithelial cells that are rich in glycogen. Estrogen helps maintain the thick glycogen-rich epithelial cells. In the presence of glycogen, lactobacillus proliferates and contributes to a low or acidic pH (4.5–5.5). The presence of lactobacillus and acidic pH helps protect against vaginal infections and urinary tract infections (UTIs).3 The high level of estrogen present in premenopausal females contributes to the thick epithelium; the rugated vaginal surface that allows for elasticity and expansion; and the vascularity and lubrication of the vaginal tissue.
With the absence of estrogen, the cytology of a postmenopausal woman’s vaginal tissue changes and is characterized by a decrease in superficial epithelial cells and an increase in intermediate cells.3 The epithelium thins, the vaginal rugae diminish, and the tissue becomes avascular and dry. Without glycogen-rich epithelial cells there is a resultant lack of lactobacillus, and so postmenopausal women experience a high or alkaline vaginal pH (6.5–7.0), which contributes to a higher risk of UTIs and vaginal infections.3
Genitourinary syndrome of menopause
Genitourinary syndrome of menopause is the term used to describe the large spectrum of symptoms women experience mainly due to the lack of estrogen.4 Common symptoms of GSM include vaginal dryness, dyspareunia, dysuria, urinary urgency, and urinary frequency. The urinary symptoms related to GSM can be particularly difficult for patients to understand.4
Most patients assume they have a UTI if they experience urinary symptoms. Educating the patient that her urinary symptoms are likely related to vaginal dryness can be difficult. The patient may insist she has an infection because antibiotics improve her symptoms. This phenomenon is commonly observed because antibiotics have anti-inflammatory properties that can initially improve symptoms. If the dryness is not addressed, the symptoms will eventually return.4
There certainly are patients with GSM who have culture-positive recurrent UTIs. This happens because the alkaline vaginal environment allows for colonization of pathogens that can migrate to the urinary tract. A urine culture should be considered in the assessment of urinary symptoms in women with GSM to rule out a UTI.4
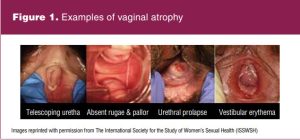 The pictures in Figure 1 are courtesy of the International Society for the Study of Women’s Sexual Health. They represent exam findings of patients with vulvovaginal and urethral changes due to lack of estrogen. The telescoping urethra image depicts a compressed urethra or one that collapses on itself. The lack of rugae, previously discussed, is illustrated. Urethral prolapse is common and is portrayed as a complete eversion of the urethralmucosa. The image of vestibular erythema presents another common finding due to the inflammation related to the lack of estrogen.5
The pictures in Figure 1 are courtesy of the International Society for the Study of Women’s Sexual Health. They represent exam findings of patients with vulvovaginal and urethral changes due to lack of estrogen. The telescoping urethra image depicts a compressed urethra or one that collapses on itself. The lack of rugae, previously discussed, is illustrated. Urethral prolapse is common and is portrayed as a complete eversion of the urethralmucosa. The image of vestibular erythema presents another common finding due to the inflammation related to the lack of estrogen.5
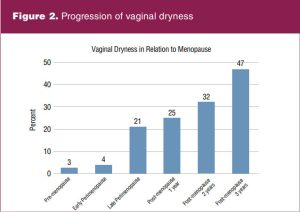 Figure 2 demonstrates how vaginal dryness progresses over time. Data were obtained from a longitudinal, population-based study of 438 women age 45 to 55 years. Results reveal the percentage of women reporting vaginal dryness increased progressively as women approached and passed through menopause. Only 3% of premenopausal women reported vaginal dryness compared with 25% of women postmenopausal by 1 year and 47% of women postmenopausal by 3 years.6,7
Figure 2 demonstrates how vaginal dryness progresses over time. Data were obtained from a longitudinal, population-based study of 438 women age 45 to 55 years. Results reveal the percentage of women reporting vaginal dryness increased progressively as women approached and passed through menopause. Only 3% of premenopausal women reported vaginal dryness compared with 25% of women postmenopausal by 1 year and 47% of women postmenopausal by 3 years.6,7
Benefits and barriers to treatment with vaginal estrogen
The use of vaginal estrogen can provide many benefits for postmenopausal women with GSM. Use should be considered for women in whom symptoms are moderate to severe or for those who do not obtain relief with nonhormonal lubricants and moisturizers.8
First, vaginal estrogen can aid with restoration of the vaginal pH to approximately 5.5 within the normal acidic premenopausal range. This acidic vaginal environment favors the growth of lactobacillus and discourages colonization of pathogens that can contribute to UTIs.2,9 Protection against UTIs with the use of estrogen is unique to local vaginal estrogen. Oral estrogen has less effect on the vaginal tissue and therefore is not recommended to use as treatment for recurrent UTIs.3
Vaginal estrogen stimulates maturation of vaginal epithelial cells, which thickens the glycogen-containing superficial cell layer. Vaginal blood flow, elasticity, and lubrication are made better. By improving the integrity of the vaginal tissue, the GSM symptoms of vaginal dryness and dyspareunia are lessened. Women using oral estrogen for vasomotor symptoms can benefit from vaginal estrogen for complete resolution of vulvovaginal symptoms.3 The peak effect of vaginal estrogen is approximately 3 months, although new users may experience improvements within the first few weeks.2,9
Despite the benefits of vaginal estrogen, barriers to its use exist. The Women’s EMPOWER survey was an internet-based survey of US women at least 45 years of age with symptoms of vaginal atrophy. The survey assessed women’s awareness of vaginal atrophy and their behaviors and attitudes associated with treatment of the symptoms. The results demonstrated women were most concerned with safety-related issues, including side effects, increased risk of breast cancer, and systemic absorption.10
Cost can also play a role as to whether or not a woman will use vaginal estrogen. Even women with a drug plan associated with their insurance might have to pay a hefty price for the medication. It is not uncommon for a 1-month supply of vaginal estrogen to cost nearly $400.11 When prescribing vaginal estrogen it is helpful to have information on prices at local pharmacies so that the patient knows what to expect. Some pharmaceutical companies do offer savings cards and/or assistance programs for low-income patients. Alternatives such as over-the-counter moisturizers and lubricants can be considered if the patient cannot afford vaginal estrogen. Vaginal lubricants for use during sexual activity can be as cheap and simple as coconut, olive, or vegetable oil from the kitchen pantry. Lubricants and moisturizers that also lower vaginal pH specifically marketed for vaginal dryness can be purchased at any local retail store.10
Vaginal estrogen formulations
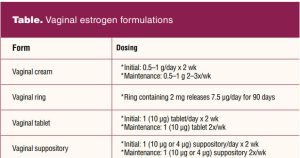 Various formulations exist for vaginal estrogen therapy. There is not one form that is better than another, but there are certain forms that are better for some women than others. All forms should be discussed along with the advantages and disadvantages so each woman can make an informed choice of what might be best for her. The Table compares all four forms and dosages of vaginal estrogen. Most formulations utilize the bioidentical hormone estradiol, which is the most plentiful and potent estrogen naturally produced by the body during the reproductive years.2
Various formulations exist for vaginal estrogen therapy. There is not one form that is better than another, but there are certain forms that are better for some women than others. All forms should be discussed along with the advantages and disadvantages so each woman can make an informed choice of what might be best for her. The Table compares all four forms and dosages of vaginal estrogen. Most formulations utilize the bioidentical hormone estradiol, which is the most plentiful and potent estrogen naturally produced by the body during the reproductive years.2
It is important to stress to patients as previously stated that full symptom relief may take 1 to 3 months to occur, although some improvement usually happens within a few weeks. It is also important to explain that vaginal estrogen is usually a long-term treatment. If the medication is discontinued, the effect will diminish and symptoms are likely to recur.2
The first form of vaginal estrogen is cream. Two vaginal creams are available: estradiol vaginal cream and conjugated estrogen vaginal cream. The difference between them is that estradiol cream solely contains estradiol as the active ingredient, whereas the conjugated estrogen cream contains a mixture of estrogenic compounds. Both are applied with an applicator. Recommended dosing is 0.5 to 1 g per day for 2 weeks, followed by maintenance dosing of 0.5 to 1 g vaginally two to three times per week. Dosages vary depending on the type of vaginal estrogen cream being used. It is important to use no more than the dose indicated on the prescription. Although the best way to administer vaginal estrogen is with the applicator, a small amount of the cream within the limits of the prescribed dose can be applied digitally to the vulva and vestibular tissues if the patient is unable to utilize the applicator.2
Cream tends to be easy to utilize and may be covered by insurance companies more readily than other forms of vaginal estrogen. Drawbacks to cream include messiness and burning with the application. Although it is recommended the cream be inserted at bedtime, it is usually not absorbed by the morning. As a result, the patient may need to wear a pad for cream expelled from the vagina. In regard to burning, if a woman is severely atrophic, applying the cream may burn because of the preservatives in the formula. In those cases, an alternative form is recommended.2
The estradiol vaginal ring provides another form of vaginal estrogen delivery. Individuals who are unable to apply cream or tablets or who have difficulty remembering to use estrogen at least a few times per week may find this an attractive option. It is placed in the upper one-third of the vagina. The vaginal ring contains a total of 2 mg of estradiol and releases 7.5 μg daily. After 90 days, the ring is removed and replaced with a new one. Some patients remove and replace the ring themselves, while others present to the clinic every 3 months for the provider to remove and replace it. The ring is not suitable for patients with vaginal prolapse who are not utilizing a pessary because it will not be retained. However, it is ideal for women with a prolapse who are using a pessary because the use of pessaries poses the risk of developing vaginal ulcers if the tissue is atrophic. The use of vaginal estrogen with a pessary will improve the integrity of the vaginal tissue and decrease the incidence of tissue breakdown. The estradiol vaginal ring should not be mistaken for the estradiol acetate vaginal ring that provides a higher dose of estrogen with systemic absorption to treat both vasomotor and vaginal symptoms.2
The estradiol vaginal tablet is a third option for vaginal estrogen delivery. The tablet is preloaded in an applicator and plunged into the vagina. Initial dosing is one tablet every day for 2 weeks, then one tablet per vagina two times per week. The tablet contains 10 μg estradiol. If the patient is able to use the applicator and insert the tablet, this may provide a less messy alternative to using cream. If a patient has severe dryness, they may not be suited for this form of estrogen because it may be difficult for the tablet to dissolve.2
The fourth form for vaginal estrogen delivery is the estradiol vaginal suppository. The patient digitally inserts the suppository into the vagina. Dosing is similar to the tablet, as it is inserted once a day for 2 weeks, then two times per week. The suppository is supplied in both a 10-μg and 4-μg form. The 4-μgsuppository provides the lowest dose of vaginal estrogen available on the market.2
Safety concerns
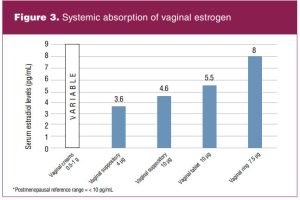 A major concern for some patients considering vaginal estrogen is systemic absorption and thus potential systemic side effects and risks. Figure 3 details the systemic absorption of vaginal estrogen based on the formulation. The normal reference range for postmenopausal estradiol concentration is less than 10 pg/mL. All vaginal estrogen formulations demonstrate serum estradiol levels to remain within the postmenopausal reference range, which is reassuring regarding safety.2,12 Still, concerns related to cancer and cardiovascular disease remain major barriers to vaginal estrogen use in women from both the patient and provider perspective. Therefore, this section discusses the risks and benefits of vaginal estrogen in women with specific health risks or health conditions.10 Two large prospective cohort studies provide reassuring results concerning the safety of vaginal estrogen for the majority of women experiencing symptoms of GSM. These studies are the Women’s Health Initiative (WHI) Observational Study and the Nurses’ Health Study.13,14
A major concern for some patients considering vaginal estrogen is systemic absorption and thus potential systemic side effects and risks. Figure 3 details the systemic absorption of vaginal estrogen based on the formulation. The normal reference range for postmenopausal estradiol concentration is less than 10 pg/mL. All vaginal estrogen formulations demonstrate serum estradiol levels to remain within the postmenopausal reference range, which is reassuring regarding safety.2,12 Still, concerns related to cancer and cardiovascular disease remain major barriers to vaginal estrogen use in women from both the patient and provider perspective. Therefore, this section discusses the risks and benefits of vaginal estrogen in women with specific health risks or health conditions.10 Two large prospective cohort studies provide reassuring results concerning the safety of vaginal estrogen for the majority of women experiencing symptoms of GSM. These studies are the Women’s Health Initiative (WHI) Observational Study and the Nurses’ Health Study.13,14
Endometrial hyperplasia
Studies have demonstrated that the use of low-dose vaginal estrogen without progesterone does not contribute to endometrial hyperplasia. It should be noted that there are no studies evaluating endometrial hyperplasia beyond 1 year and so long-term safety has not been established. Based on the results of available studies, the North American Menopause Society (NAMS) and the American College of Obstetricians and Gynecologists (ACOG) state that low-dose vaginal estrogen can be used indefinitely, if needed, in women at low risk for endometrial cancer. Both NAMS and ACOG do not advise the routine use of concomitant progesterone to protect the endometrium. Additionally, women at low risk for endometrial cancer do not require routine endometrial surveillance.2,3,13,15
If postmenopausal spotting or bleeding occur, a thorough evaluation with pelvic examination and transvaginal ultrasound (US) or endometrial biopsy is required. Although studies are lacking, based on expert opinion, NAMS states that for women at increased risk for endometrial cancer (eg, obesity) endometrial surveillance via transvaginal US or intermittent progesterone withdrawal may be considered.2,16
Breast cancer
Women with no personal history of breast cancer and not at high risk for breast cancer can safely use vaginal estrogen. Women at high risk for breast cancer because of family history or who have a personal history of breast biopsy can also safely use vaginal estrogen. A 2018 consensus recommendation from NAMS suggests systemic estrogen therapy does not increase the risk of breast cancer in this group; therefore, it was determined low-dose vaginal estrogen is safe.8
For women with estrogen receptor-positive breast cancer on tamoxifen, ACOG, the Endocrine Society, and NAMS cautiously support the use of low-dose vaginal estrogen if needed in consultation with the woman’s oncologist. Tamoxifen acts by binding to estrogen receptors in breast tissue, thus blocking estrogen action. Even if there was some systemic absorption of low-dose vaginal estrogen, it would be blocked from breast cells. Women with estrogen receptor-positive breast cancer on aromatase inhibitors (AIs) should not be treated with vaginal estrogen. Aromatase inhibitors act by blocking the conversion of adrenal androgens to estrogen and are only effective after the ovaries are no longer producing estrogen. Any systemic absorption from vaginal estrogen could theoretically increase the risk for cancer recurrence.8 Nonhormonal treatments should be first-line treatment for these women. For those with severe vaginal symptoms not managed with nonhormonal treatments, providers may consider consulting with the woman’s oncologist on switching from an AI to tamoxifen.8,14,16
No data address the safety or efficacy of vaginal estrogen in survivors of triple-negative breast cancer. However, for women who are long-term survivors of estrogen receptor-negative disease for which risk of recurrence is low and symptomatology is troubling, consideration of vaginal estrogen is reasonable.8
No data are available to inform decisions regarding the use of vaginal estrogen in women with metastatic disease. If symptoms are severe and quality of life is the priority, the decision to use vaginal estrogen in women with receptor-positive metastatic disease may be reasonable after a discussion of the risks and benefits.8
Heart disease
Analysis of data from the WHI Observational Study and the Nurses’ Health Study provided the same conclusion. There was no elevated risk for stroke, deep vein thrombosis, myocardial infarction, or pulmonary embolism among postmenopausal vaginal estrogen users. Findings from both of these studies support the safety of vaginal estrogen in regard to any cardiovascular disease risk.13,14
Gynecologic cancer survivors
A retrospective cohort study was conducted including 224 women diagnosed with endometrial, ovarian, or cervical cancer from January 1, 1991, to December 31, 2017, who were subsequently treated with vaginal estrogen for GSM. Results demonstrated that adverse outcomes, including recurrence and thromboembolic events, were infrequent. Based on these data, vaginal estrogen may be considered safe in gynecologic cancer survivors.17
Estrogen: A controversial history
One cannot conclude a discussion regarding vaginal estrogen without examining some of the controversy behind its use. The concerns about vaginal estrogen and its connection with cardiovascular events and breast cancer stem largely from the WHI study published in 2002. Nearly 20 years later, we are still trying to demonstrate the safety of vaginal estrogen and clearly distinguish it from oral estrogen. The initial results of the 2002 WHI study demonstrated hormone replacement therapy (HRT) had more risks than benefits for all women, specifically increasing the incidence of cardiovascular events and breast cancer. Additionally, all forms of HRT were considered the same, which meant that oral and vaginal estrogen were considered equally as risky. Despite subsequent studies focused on the use of vaginal estrogen that include the WHI Observational Study and the Nurses’ Health Study over the past several years demonstrating safety and efforts to distinguish vaginal estrogen from oral estrogen, fear and negativity persist.12,18,19
The fact that the black box warning for estrogen is on vaginal estrogen formulations does not help ameliorate the confusion of estrogen safety because the boxed warnings are based on systemic estrogen or combination estrogen-progesterone hormone therapy. In hopes of improving the care of women, proposals for alternative labeling for vaginal estrogen have been shared with the US Food and Drug Administration, but the boxed warning remains on vaginal estrogen. This lack of clarity demonstrates how imperative it is to educate women on the difference between oral and vaginal estrogen and that vaginal estrogen is indeed safe for most women to use.8
Conclusion
Postmenopausal women affected by GSM are grossly undertreated due to lack of knowledge and communication.3 This is unfortunate because vaginal estrogen is safe and effective for most women. The women’s health nurse practitioner is well positioned to recognize the need for vaginal estrogen, educate the patient on safety concerns, and prescribe it as treatment when indicated. Women may not routinely volunteer they are suffering from GSM symptoms, so it is imperative to ask specific questions. Additionally, women with a personal or family history of breast cancer often think they are not candidates for vaginal estrogen. These women need adequate counseling on the safety of vaginal estrogen for them to make an informed decision. Advocate and inform your patients so they are not a GSM statistic.
Kendra L. Roloff is a certified women’s health and urology nurse practitioner at Pelican Health in Bismarck, North Dakota. The author has no actual or potential conflicts of interest in relation to the contents of this article.
References
- Shiren, J, Gass, M. The North American Menopause Society recommendations for clinical care of midlife women. Menopause. 2014;21(10).
- North American Menopause Society. The 2020 genitourinary syndrome of menopause position statement of The North American Menopause Society. Menopause. 2020;27(9):976-992.
- North American Menopause Society. Menopause Practice: A Clinician’s Guide, 6th ed. September 25, 2019.
- Angelou K, Grigoriadis T, Diakosavvas M, et al. The genitourinary syndrome of menopause: an overview of the recent data. Cureus. 2020;12(4):e7586
- International Society for the Study of Women’s Sexual Health, Inc. Slide deck: female sexual function and dysfunction. 2021.
- Dennerstein L, Dudley EC, Hopper JL, et al. A prospective population-based study of menopausal symptoms. Obstet Gynecol. 2000;96(3):351-358.
- Waetjen L, Crawford S, Chang P, et al. Factors associated with developing vaginal dryness symptoms in women transitioning through menopause: a longitudinal study. Menopause. 2018;25(10):1094-1104.
- Faubion S, Larkin L, Stuenkel C, et al. Management of genitourinary syndrome of menopause in women with or at high risk for breast cancer: consensus recommendations from The North American Menopause Society and The International Society for the Study of Women’s Sexual Health. Menopause. 2018;25(6):596-608.
- Archer DF. Efficacy and tolerability of local estrogen therapy for urogenital atrophy. Menopause. 2010;17(1):194-203.
- Krychman M, Graham S, Bernick B, et al. The Women’s EMPOWER survey: women’s knowledge and awareness of treatment options for vulvar and vaginal atrophy remains inadequate. J Sexual Med. 2017;14(3):425-433.
- Estrace vaginal cream prices, coupons and patient assistance programs. Drugs.com. October 11, 2021.
- Pinkerton J, Liu J, Santoro N, et al. Workshop on normal reference ranges for estradiol in postmenopausal women: commentary from the North American Menopause Society on low-dose vaginal estrogen labeling. Menopause. 2020;27(6):611-613.
- Bhupathiraju S, Grodstein F, Stampfer M, et al. Vaginal estrogen use and chronic disease risk in the Nurses’ Health Study. Menopause. 2018;26(6):603-610.
- Crandall CC, Hovey K, Andrews C, et al. Breast cancer, endometrial cancer, and cardiovascular events in participants who used vaginal estrogen in the Women’s Health Initiative Observational Study. Menopause. 2018;25(1):11-20.
- American College of Obstetricians and Gynecologists. ACOG practice bulletin No. 114: management of menopausal symptoms. Obstet Gynecol. 2014;123(1):202-216.
- American College of Obstetricians and Gynecologists. The use of vaginal estrogen in women with a history of estrogen-dependent breast cancer. Updated 2020.
- Chambers LM, Herrmann A, Michener CM, et al. Vaginal estrogen use for genitourinary symptoms in women with a history of uterine, cervical, or ovarian carcinoma. Int J Gynecol Cancer.2020;30(4):515-524.
- Cagnacci A, Venier M. The controversial history of hormone replacement therapy. Medicina. 2019;55(9):602.
- North American Menopause Society. Why the product labeling for low-dose vaginal estrogen should be changed. Menopause. 2014;21(9):911-916.
KEY WORDS: vaginal estrogen, genitourinary syndrome of menopause, postmenopause, vaginal dryness, lower urinary tract symptoms
Womens Healthcare. 2022;10(2):30-35. doi:10.51256/WHC042230

