Aromatase inhibitors (AIs) are a first-line therapy for postmenopausal women diagnosed with estrogen receptor-positive breast cancer. Because of the estrogen suppression caused by the AIs, changes in bone structure frequently occur, leading to or exacerbating osteoporosis. Survival rates have increased, and women treated for breast cancer frequently return to the primary care setting. Practitioners need to be aware of the enhanced risk of osteoporosis in this population, how to assess, and know treatment guidelines.
Breast cancer is the most frequent, noncutaneous cancer diagnosis in women. The lifetime risk of breast cancer in women is 13%, that is, 1 in 8 women will receive a breast cancer diagnosis during their lifetime.1 The average age of diagnosis of breast carcinoma in women is 62 years. The risk of breast cancer in women continues to increase with age. Most breast cancers diagnosed in postmenopausal women are estrogen receptor positive (ER+), and long-term use of aromatase inhibitors (AIs) has become standard treatment. This therapy blocks conversion of endogenous androgenic steroids into estrogen, lowering serum estrogen levels, and significantly impeding the growth and spread of ER+ postmenopausal breast cancer. Treatment of breast cancer with AIs in postmenopausal women does carry risks that must be addressed. Among these risks is the occurrence of bone loss at twice the rate that occurs physiologically, thus increasing the risk of fragility fracture.2
Loss of bone health places at peril independence, quality of life, and survival in women as they age. Estimates globally are that over 200 million people have osteoporosis and that 9 million fractures occur annually.3 Hip fractures are associated with increased mortality, with the cumulative mortality after 1 year ranging between 20% and 40%.4 Only 33% of older women who have a hip fracture will be able to return to independence.3
The focus on maintaining bone health in women with a history of breast cancer has received greater attention in recent years because survival rates continue to improve, with 69% of all newly diagnosed breast cancer patients currently living more than 5 years post treatment.1 As breast cancer patients live longer and transition to routine follow-up provider care, including with nurse practitioners in primary care practice, maintaining and enhancing quality of life while managing and/or preventing long-term and often treatment-related adverse effects will become priorities of care. This article focuses on the assessment and management of bone health in women with a history of breast cancer treated with AIs.
Aromatase inhibitors
Treatment decisions in the newly diagnosed breast cancer population are based on many factors including tumor size, lymph node involvement, comorbidities, and estrogen receptor status. The current guidelines for the postmenopausal woman diagnosed with a small estrogen-positive breast cancer tumor (< 0.5 cm) and no lymph node involvement are surgery followed by 5 years of AI therapy.5 Women with lymph node involvement usually receive surgery followed by possible adjuvant chemotherapy, concluding with AI therapy for 5 years. Women with lymph node involvement may benefit from extended AI therapy from 7.5 to 10 years. An alternative for women with lymph node involvement is to receive tamoxifen for 2 to 6 years followed by AI for another 4 to 8 years for a total of 10 years of endocrine suppression therapy.
AIs are the standard of care for the management of ER+ postmenopausal breast cancer. Of postmenopausal women diagnosed with breast cancer, 80% are ER+ and are placed on AIs.6
Aromatase inhibitors have been demonstrated as superior to tamoxifen regarding disease-free survival and time to recurrence with disease. Tamoxifen increases the risk of thromboembolic events and endometrial cancer.7 Furthermore, the longevity and quality-of-life benefits of AI therapy are considered to significantly outweigh AI-related long-term adversities.8
The fact remains that AIs are associated with a decrease in bone mineral density (BMD) and subsequent increase in fracture risk.9–11 As AIs diminish sex steroid hormone production, there is a resultant increase in bone remodeling and resorption and an increased loss of BMD occurs.12,13
Bone health assessment
Physiologic postmenopausal bone loss is approximately 1% to 2% change per year as compared to women who receive AIs, losing an additional 2% to 3% bone per year.2 Women treated with AIs have an increased risk of fractures over the 5-year treatment period. The risk of bone fracture begins to decrease over the next 5 years after treatment is completed. Given the increased risk of bone demineralization and fracture in postmenopausal patients receiving AI therapy, it is essential to conduct a thorough bone health assessment that includes identifying modifiable risk factors for osteoporosis throughout all phases of treatment, including smoking, alcohol use, physical inactivity, and low body weight. A thorough assessment also includes BMD measurement and use of a validated risk factor tool such as the Fracture Risk Assessment tool (FRAX).
The National Comprehensive Cancer Network (NCCN) and American Society of Clinical Oncology (ASCO) have established guidelines for assessing bone health in cancer patients.14,15 The guidelines assume that BMD is proportionate to bone strength and include a baseline BMD scan and screening every 2 years after. One retrospective study of over 25,000 women with early-stage breast cancer found that a baseline BMD scan for osteoporosis 1 year before or 6 months after AI therapy started was associated with a decreased risk of fractures and a higher likelihood for receiving antiresorptive therapies.8
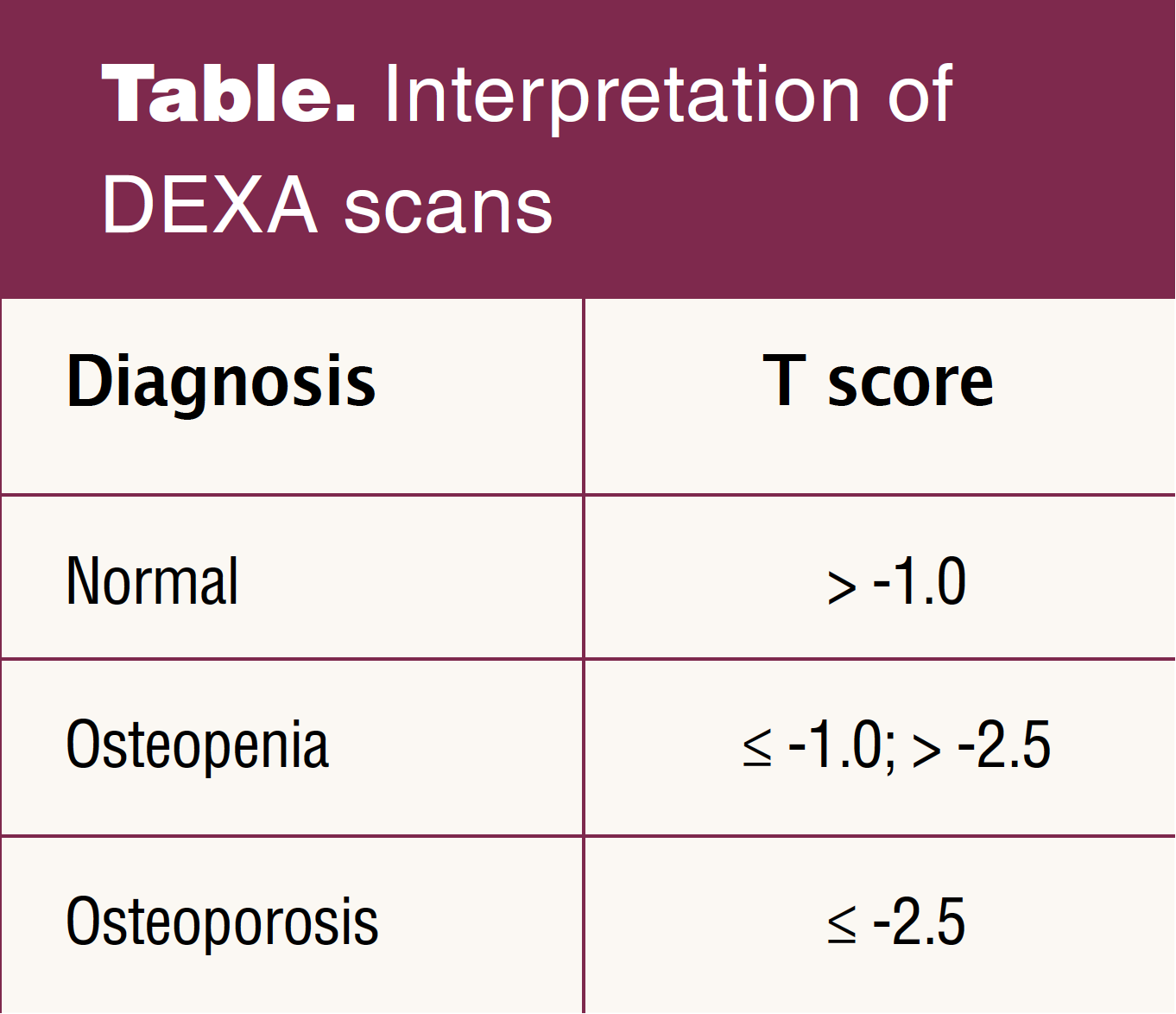
Bone densitometry or dual-energy X-ray absorptiometry (DEXA) is considered the gold standard for measuring BMD. DEXA results reveal the amount the bone density differs from the mean and is predictive of the risk of osteoporotic fracture in addition to clinical indicators. DEXA results are reported as T scores representing the number of standard deviations (SDs) from young adult mean values. Negative T scores indicate lower BMD, and positive scores indicate higher bone density. There is approximately a 2-fold increase in fracture risk for every 1 SD decrease in BMD.16,17 The World Health Organization identifies the international reference standard to describe osteoporosis in postmenopausal women as a femoral neck BMD of 2.5 SD or more below the mean of a 20- to 29-year-old Caucasian woman.18 Osteopenia is described as a T score at the femoral neck between -1.0 and -2.5. Other sites such as the lumbar spine or total hip can be used for diagnosis in clinical practice. A T score higher than -1.0 indicates normal bone density (Table).16
BMD is used as a surrogate marker of bone strength, but the test accounts for only 60% of differences in bone fragility and it is unable to depict differences in bone material composition and structural design, important attributes of bone strength.19,20 Several studies found large numbers of postmenopausal women who experienced a fracture had T scores above -2.5.21,22 The addition of trabecular bone scores using data from the DEXA scan to evaluate the microarchitecture of bone is promising but is not yet considered standard of care. Bone mineral density remains an indirect indicator of osteoporosis and fracture risk.23 The addition of a validated risk factor tool such as the FRAX calculator improves assessment accuracy of a person’s risk for a fracture over the BMD alone.24
FRAX has been validated as a predictive tool for bone fracture in the general population but does have limitations. The predictive accuracy of FRAX is diminished if information is entered incorrectly and may under or overestimate the risk of bone fracture. The tool should be used prior to any intervention or treatment for osteoporosis to avoid an overestimation of risk. According to researchers, FRAX scores may over or underestimate the 10-year probability of fracture, skewing treatment decisions.17 Investigators also point out that the data used in FRAX include the patient’s femoral neck BMD but do not take the lumbar spine density into consideration.24 Fall history is not considered when computing the FRAX score.
There are additional limitations to the predictive value of FRAX for postmenopausal women with breast cancer receiving AI therapy. Specific cancer treatments commonly used and known to affect bone health are not included in FRAX because the tool was developed for the general population. Although AI therapy is not included as a clinical risk factor in FRAX, the provider may enter yes for “secondary osteoporosis” when using the FRAX algorithm.14 FRAX calculator instructions indicate to enter yes to the secondary osteoporosis category if a patient has a disorder strongly associated with osteoporosis.25 This may render a more complete assessment, but researchers found that when secondary osteoporosis was added into the calculation for breast cancer AI users, FRAX overestimated risk for future fractures.26
The NCCN task force currently recommends screening for fracture risk in all patients with breast cancer initiating AIs by obtaining a clinical history, DEXA scan, and FRAX calculation.14,25 A study of early-stage breast cancer patients who received AI and had FRAX assessment added to BMD identified an additional 9% of women pretreatment and 2% posttreatment with elevated fracture risk.27
Although guidelines for screening bone health in the oncology population are established, significant variation in clinical practice frequently exists. Current research findings support the integration of FRAX into clinical assessment of the higher-risk oncology population and the proactive management of osteoporosis in oncology patients.28 Nurse practitioners in primary care practice can implement routine assessment of all patients on AI therapy to identify those at increased risk who may require targeted patient education and treatment intervention to minimize long-term impact on bone health-related quality of life. Incorporation of FRAX has demonstrated increased identification of fracture risk in the oncology population. The FRAX calculator is readily accessible online and is simple to complete as an adjunct to BMD, providing an opportunity to proactively treat patients who may have been overlooked with standardized, routine screening.
Interventions to reduce bone fracture risk
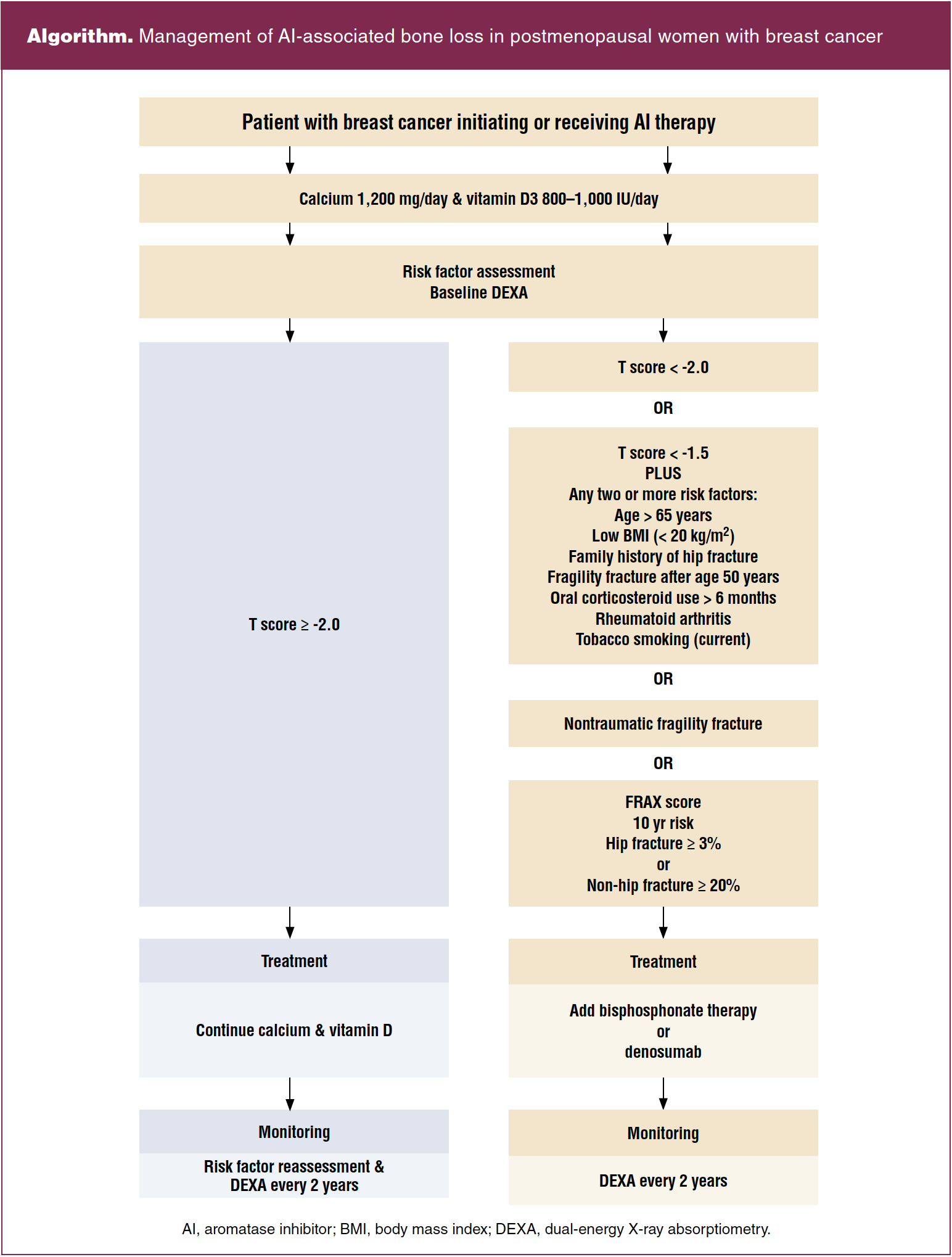
Patient education is a critical cornerstone for empowering women with breast cancer to understand and seek appropriate treatment, anticipate and negotiate adverse effects of the cancer and related treatments, and avoid bone fractures. In one study, researchers found that the great majority (80%) of women being treated with AIs for postmenopausal breast cancer wanted to be proactive to prevent bone loss but that they had little knowledge about osteoporosis.29 The women in the study knew the importance of calcium and vitamin D supplementation, but most did not know the importance of weight-bearing exercise and strength training.29 Thus, initial interventions include education with a focus on reducing modifiable risk factors. Lifestyle changes that promote bone health include cessation of cigarette smoking and reduction or cessation of alcohol consumption, increasing physical activity including weight-bearing activities, and minimizing the risk of falling.30 Women should participate in at least 150 minutes of exercise weekly and in strength training at least twice weekly. Education to reduce fall risks is considered vital. All postmenopausal women should have an intake of calcium 1,200 mg/day and vitamin D3 800 to 1,000 IU/day through diet and supplementation.30 An OH-D3 level can be obtained as a baseline measurement of vitamin D status. Higher doses of vitamin D may be indicated if a deficiency is identified. It should be noted that vitamin D and/or calcium supplementation alone has been demonstrated as ineffective in preventing fractures.2 However, benefits of vitamin D and calcium supplementation include decreased postmenopausal BMD loss and strengthened muscle, which reduces fall risks that lead to fractures.31–33
Several well-respected cancer and bone societies have reached a consensus on management of AI-associated bone loss in postmenopausal women with breast cancer.14,15,34 Recommendations are based on BMD T scores and risk factors (Algorithm). Patients with T scores greater than -2.0 and no additional risk factors should be advised on getting regular exercise and adequate calcium and vitamin D intake while monitoring risk and repeating BMD measurement at 1- to 2-year intervals. Patients with T scores lower than -1.5 with 2 or more risk factors and those with T scores lower than -2.0 should begin bisphosphonate therapy with repeat BMD measurement every 2 years.2
Bisphosphonates (alendronate, risedronate, zoledronic acid) are antiresorptive agents that work by decreasing bone loss and increasing bone strength. They slow bone resorption by reducing osteoclast function, which results in improved bone density and reduced risk of fractures. Bisphosphonates have a long-term safety record and are cost-effective. Contraindications include having significantly reduced renal function or hypocalcemia. Rare adverse effects include osteonecrosis of the jaw and atypical subtrochanteric femur fractures occurring most often with long-term use. Instructions for the use of oral bisphosphonates include strategies to enhance absorption and minimize acid reflux.
Women with a history of breast cancer compose the largest group of cancer survivors. Currently, there are 3.5 million women who survive breast cancer with the majority transitioning back to their primary care provider for their healthcare needs.35 Care of survivors of breast cancer includes screening for long-term and late effects of cancer treatment. Osteoporosis related to AI therapy is well documented. Routine screening for osteoporosis in breast cancer survivors who receive AI therapy can reduce the risk of fracture from occurring.
Kristin K. Ownby is Associate Professor of Nursing at the Clinical Cizik School of Nursing at UTHealth in Houston, Texas. Lydia T. Madsen is an adjunct faculty member at the Cizik School of Nursing at UTHealth. Faith A. Strunk is Assistant Professor of Nursing at the Clinical Cizik School of Nursing at UTHealth. The authors have no actual or potential conflicts of interest in relation to the contents of this article.
References
1 American Cancer Society. Cancer Facts & Figures. 2021. https://www.cancer.org/cancer/breast-cancer/about/how-common-is-breast-cancer.html.
2 Hadji P, Aapro MS, Body JJ, et al. Management of aromatase inhibitor-associated bone loss (AIBL) in postmenopausal women with hormone sensitive breast cancer: joint position statement of the IOF, CABS, ECTS, IEG, ESCEO IMS, and SIOG. J Bone Oncol. 2017;7:1-12.
3 Porter JL, Varacallo M. Osteoporosis. StatPearls Publishing; February 12, 2022.
4 Guzon-Illescas O, Perez Fernandez E, Crespí Villarias N, et al. Mortality after osteoporotic hip fracture: incidence, trends, and associated factors. J Orthop Surg Res. 2019;14(1):203.
5 Gradishar WJ, Anderson BO, Abraham J, et al. Breast cancer, Version 3.2020, NCCN Clinical Practice Guidelines in Oncology. J Natl Compr Canc Netw. 2020;18(4):452-478.
6 Majumder A, Singh M, Tyagi SC. Post-menopausal breast cancer: from estrogen to androgen receptor. Oncotarget. 2017;8(60):102739-102758.
7 Chlebowski RT, Anderson GL, Aragaki AK, et al. Association of menopausal hormone therapy with breast cancer incidence and mortality during long-term follow-up of the Women’s Health Initiative randomized clinical trials. JAMA. 2020;324(4):369-380.
8 Ruhstaller T, Giobbie-Hurder A, Colleoni M, et al. Adjuvant letrozole and tamoxifen alone or sequentially for postmenopausal women with hormone receptor-positive breast cancer: long-term follow-up of the BIG 1-98 trial. J Clin Oncol. 2019;37(2):105-114.
9 Fukumoto S, Soen S, Taguchi T, et al. Management manual for cancer treatment-induced bone loss (CTIBL): position statement of the JSBMR. J Bone Miner Metab. 2020;38(2):141-144.
10 Kawahara T, Fusayasu S, Izumi K, et al. Bone management in Japanese patients with prostate cancer: hormonal therapy leads to an increase in the FRAX score. BMC Urol. 2016;16(1):32.
11 Lu H, Lei X, Zhao H, et al. Bone mineral density at the time of initiating aromatase inhibitor therapy is associated with decreased fractures in women with breast cancer. J Bone Miner Res. 2021;36(5):861-871.
12 Guise TA, Mohammad KS, Clines G, et al. Basic mechanisms responsible for osteolytic and osteoblastic bone metastases. Clin Cancer Res. 2006;12(20 Pt 2):6213s-6216s.
13 Kwan ML, Lo JC, Laurent CA, et al. A prospective study of lifestyle factors and bone health in breast cancer patients who received aromatase inhibitors in an integrated healthcare setting. J Cancer Surviv. 2021;10.1007/s11764-021-00993-0. Online ahead of print.
14 Gralow JR, Biermann JS, Farooki A, et al. NCCN Task Force Report. Bone health in cancer care. J Natl Compr Canc Netw. 2013;11 suppl 3:S1-S51.
15 Runowicz CD, Leach CR, Henry NL, et al. American Cancer Society/American Society of Clinical Oncology Breast Cancer Survivorship Care Guideline. CA Cancer J Clin. 2016;66(1):43-73.
16 US Preventive Services Task Force; Curry SJ, Krist AH, Owens DK, et al. Screening for osteoporosis to prevent fractures: US Preventive Services Task Force Recommendation Statement. JAMA. 2018;319(24):2521-2531.
17 Lewiecki EM. Osteoporosis: clinical evaluation. In: Feingold KR, Anawalt B, Boyce A, et al, eds. Endotext [Internet]. South Dartmouth, MA: MDText.com, Inc.; 2000-. Updated June 7, 2021. https://www.ncbi.nlm.nih.gov/books/NBK279049/.
18 Kanis JA; World Health Organization Scientific Group. Assessment of osteoporosis at the primary health-care level. Technical Report. World Health Organization Collaborating Centre for Metabolic Bone Diseases, University of Sheffield, UK. 2007.
19 McClung MR. The relationship between bone mineral density and fracture risk. Curr Osteoporos Rep. 2005;3(2):57-63.
20 Osterhoff G, Morgan EF, Shefelbine SJ, et al. Bone mechanical properties and changes with osteoporosis. Injury. 2016;47(suppl 2):S11-S20.
21 Schuit SC, van der Klift M, Weel AE, et al. Fracture incidence and association with bone mineral density in elderly men and women: the Rotterdam Study. Bone. 2004;34(1):195-202.
22 Sornay-Rendu E, Boutroy S, Munoz F, Delmas PD. Alterations of cortical and trabecular architecture are associated with fractures in postmenopausal women, partially independent of decreased BMD measured by DXA: the OFELY study. J Bone Miner Res. 2007;22(3):425-433.
23 Bluemke DA, Lui S. Principles and Practice of Clinical Research, 3rd ed. London, UK: Academic Press, 2012.
24 Zekri J, Farag K. Assessment of bone health in breast cancer patients starting adjuvant aromatase inhibitors: a quality improvement clinical audit. J Bone Oncol. 2016;5(4):159-162.
25 World Health Organization Collaboration Center for Metabolic Bone Diseases, University of Sheffeld. FRAX WHO fracture risk assessment tool. https://shef.ac.uk/FRAX/tool.
26 Leslie WD, Morin SN, Lix LM, et al. Performance of FRAX in women with breast cancer initiating aromatase inhibitor therapy: a registry-based cohort study. J Bone Miner Res. 2019;34(8):1428-1435.
27 Mariotti V, Page DB, Davydov O, et al. Assessing fracture risk in early stage breast cancer patients treated with aromatase-inhibitors: an enhanced screening approach incorporating trabecular bone score. J Bone Oncol. 2016;7:32-37.
28 Siris ES, Baim S, Nattiv A. Primary care use of FRAX: absolute fracture risk assessment in postmenopausal women and older men. Postgrad Med. 2010;122(1):82-90.
29 Bailey S, Lin J. The association of osteoporosis knowledge and beliefs with preventive behaviors in postmenopausal breast cancer survivors. BMC Womens Health. 2021;21(1):297.
30 Shapiro CL. Osteoporosis: a long-term and late-effect of breast cancer treatments. Cancers (Basel). 2020;12(11):3094.
31 Kling JM, Clarke BL, Sandhu NP. Osteoporosis prevention, screening, and treatment: a review. J Womens Health (Larchmt). 2014;23(7):563-572.
32 Wu H, Pang Q. The effect of vitamin D and calcium supplementation on falls in older adults: a systematic review and meta-analysis. [German]. Orthopade. 2017;46(9):729-736.
33 Dhaliwal R, Aloia JF. Effect of vitamin D on falls and physical performance. Endocrinol Metab Clin North Am. 2017;46(4):919-933.
34 Hadji P, Aapro MS, Body JJ, et al. Management of aromatase inhibitor-associated bone loss (AIBL) in postmenopausal women with hormone sensitive breast cancer: joint position statement of the IOF, CABS, ECTS, IEG, ESCEO IMS, and SIOG. J Bone Oncol. 2017;7:1-12.
35 Grant M, De Rossi S, Sussman J. Supporting models to transition breast cancer survivors to primary care: formative evaluation of a cancer care Ontario initiative. J Oncol Pract. 2015;11(3):e288-e295.
Key words: osteoporosis, aromatase inhibitor, ERP, breast cancer, primary care, guidelines
Womens Healthcare. 2022;10(5):19-24. doi: 10.51256/WHC102219