Progesterone has been called the “mother hormone.” It is created from pregnenolone along with the estrogens, androgens, mineralocorticoids and glucocorticoids. And pregnenolone is made from cholesterol.
This begs the question: What happens when cholesterol levels are extremely low? Could this affect hormone production? But that’s a potential subject for a future article – today I want to focus on progesterone and its actions. To help your understanding, first I’d like to explain a bit about general hormone metabolism. Here’s a diagram that might make my explanations below clearer:
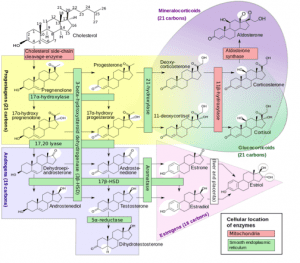 Source: Wikiversity
Source: Wikiversity
All these steroid hormones have a similar 4-ring structure with differences in the side chains. It’s easy to see how the body creates one hormone from another by making small changes in the side groups of the molecule or double bonds of the rings. If one hormone is deficient, this can affect the production of others, although some hormones can be produced by more than one pathway.
Of course, pharmaceutical hormones are not metabolized in the same manner, and supporting this balancing metabolism is one clear advantage of using hormones that are identical to those produced by the human body (sometimes referred to as biologically identical or “bioidentical” hormones). Supplementing progesterone, the “mother hormone,” provides a starting molecule the body can use to create other hormones that may be missing.
The chart also helps us understand why we see interactions between hormones. For example, when under high stress with increased demand for the stress hormone, cortisol, metabolism would be diverted from creating reproductive hormones in favor of stress hormones that are needed more urgently at that moment. This could help explain why some women may experience infertility or increased menopausal symptoms during periods of chronic stress.
Additionally, with the similarities in structure, large amounts of one hormone could potentially occupy receptors of another but without stimulating the receptor in the same way. This could result in a reduced hormone effect even though the hormone is present in the usual amount. A sudden stressful event bringing on a hot flash would be an example of this with a spike of cortisol interfering with estrogen action by blocking its receptor.
Metabolism
We know that progesterone is not well absorbed in the gastrointestinal tract. Additionally, what little progesterone that is absorbed is essentially metabolized completely on the first pass through the liver, making standard oral tablets/capsules ineffective. Manufacturers have worked around this by dissolving/suspending progesterone in peanut oil, to which it binds very strongly. This enables some of the swallowed dose—estimated at 10% or less—to escape first-pass metabolism by entering the lymphatic system. However, the other 90% is changed into metabolites that bind with benzodiazepine receptors in the brain, creating the classic drowsiness associated with oral progesterone in peanut oil.
Although we shouldn’t assume 100% absorption through the skin, transdermal progesterone is generally dosed at about 1/10th of the oral dose, suggesting a range of 10 to 30mg daily. The body is known to produce 30mg of progesterone daily starting at ovulation and continuing until just before menstruation and this is often used as a guide for a starting dose: 30mg transdermally on days 15-28. With these more physiological doses we do not see drowsiness as a side effect, suggesting (at least in my humble opinion) that the transdermal route should be preferred.
The problem with transdermal progesterone, however, is that it has not been well tested when used in combination with estrogen replacement. After years of experience working with this form of progesterone, however, I found a simple way for women to monitor if they have sufficient progesterone to balance the estrogen in their system.
Hyper-stimulated ovary syndrome
But first, let me take a step back and talk about hyper-stimulated ovary syndrome to explain the origin of my monitoring method. This syndrome was studied in detail by physician researcher, Dr. Jerilynn Prior, at University of British Columbia in Canada. It is a perimenstrual variation where women have anovulatory cycles with an inability to produce an ovum or progesterone but continued ability to produce ovarian estrogen. This contrasts with the description of ovulation ceasing when the ovaries run out of eggs with estrogen and progesterone both diminishing as a result, as described in most texts on menopause. Dr. Prior’s research showed this occurs in about 40% of women, but is a phenomenon not included in many medical texts. Animal studies—notably those done on alligators in Lake Apopka, Florida, which experienced severe contamination with endocrine disrupting chemicals—suggest that this change could be due to low level exposure to environmental contaminants with hormonal activity, commonly known as endocrine disruptors.
One function of progesterone is to shrink blood vessels in the endometrium after ovulation so less blood is lost during menstruation. Without the presence of sufficient progesterone to balance (or oppose) the amount of estrogen being produced, blood vessels continue to grow in the endometrium, leading to increased amounts of blood and clots in menstrual fluid and a much heavier period. Additionally, without the feedback of progesterone to signal the hypothalamus that ovulation has occurred, the menstrual control center increases production of FSH and LH, further stimulating estrogen production by the ovaries.
This results in higher than normal blood estrogen levels—sometimes as much as 6 or 7 times normal—with little or no progesterone to oppose it. Driven by high estrogen and little or no progesterone, the woman experiences heavy flooding menstrual periods containing noticeable blood clots. Of course, supplementing estrogen in this case, as is often done when increased FSH and LH are measured in the blood, does not make sense and would have the potential to worsen the woman’s symptoms of fluid retention, breast soreness and heavy flow. She just needs progesterone to balance and oppose the increased estrogen she is producing.
After menstruation, vaginal mucous production gradually increases in response to rising levels of estrogen. This mucous is clear and “stretchy” (forms gel-like strings when stretched between fingers or 2 pieces of toilet paper), much like an egg white would. At ovulation, the mucous changes to a white or yellowish colour and creamy texture with reduced volume under the influence of progesterone. Vaginal mucous is often used by women to indicate their level of fertility since the peak amount of mucous production occurs with peak estrogen levels and fertility the day before ovulation, and the change in colour and texture of the mucous indicates that ovulation has occurred and progesterone is being produced.
However, women with this anovulatory hyper-stimulated ovary syndrome would often tell me they produced the clear, stretchy mucous from the end of one period until the start of the next, and so much wetness they needed to wear a mini-pad to absorb it.
So, I used this mucous production caused by unopposed estrogen to help women determine whether they were supplementing sufficient progesterone to balance the estrogen they were producing. In hyper-stimulated ovary syndrome, I advised them to apply 30mg initially, then to increase the dose incrementally until the clear egg white-like mucous was no longer present. Some women needed as much as 60mg daily and needed to use it from the end of one period to the start of the next to control their naturally occurring endometrial hyperplasia.
Hormone replacement therapy
Let’s fast-forward to how this might apply to hormone replacement therapy. When supplementing with estrogen, compared to balancing endogenous estrogen production as above, the same principle could be used. However, I do wish to state that I am not aware of any studies that have been performed using this technique. It is based on my experience helping women who wanted to use low-dose hormone replacement therapy using transdermal creams. In my experience, most clinicians with patients using progesterone transdermal cream to oppose replaced estrogen would opt to confirm the absence of endometrial hyperplasia via biopsy.
The dosage conversion from oral progesterone is simple: if an estradiol 25mcg patch is sufficiently balanced by oral progesterone 200mg, then applying a measured dose of progesterone cream 20mg (assuming 90% metabolism of an oral dose) should be the correct dose. However, progesterone, being produced in high amounts during pregnancy, is considered a safe hormone so I would usually err on the generous side of dosing and recommend 30mg daily, paralleling natural production. There will also be some individual variation. For example, women with higher amounts of 5-alpha-reductase enzyme in the skin (identified by increased body hair growth) will metabolize more progesterone as it passes through the skin, requiring a slightly higher dose. The presence of increased amounts of clear vaginal mucous would signal an increased dose is needed, and an endometrial biopsy after 3 to 4 months would confirm a sufficient dose.
However, some women, knowing that progesterone is a precursor to other hormones and that women produce 300 to 400mg daily during pregnancy (and that pregnancy is associated with lower rates of breast cancer, not higher), opted to supplement only with progesterone to ease the symptoms of the menopausal transition. Some, who suffered from heavy periods (hyper-stimulated ovary syndrome) began using progesterone before menopause and simply continued, eventually decreasing the dose gradually before stopping when it was no longer needed. Others, with more damaged ovaries or with a history of exposure to estrogen-like environmental chemicals, opted to continue with a low dose of 5 to 20mg daily to oppose the estrogen activity of these chemicals that are known to be stored in the body long-term (for example, DDT).
Overall, these women on progesterone alone did amazingly well—some even commented that they noticed improved memory and clearer thinking. One client called progesterone cream her “memory cream.” Again, I have been unable to find research supporting a role of progesterone in memory, but so many women commented that their recall of words and names was noticeably improved that I have become convinced it must play a role, at least in some types of memory.
Tissues responsive to progesterone
Progesterone receptors have been identified in many body tissues besides the uterus, including the breast, brain, bone and myelin. Studies to detect the presence of a receptor have been used to confirm action of the hormone in those tissues.
A landmark study by Chang et al back in 1995 not only demonstrated that progesterone is well absorbed through the skin, but that it opposes estrogen’s stimulation of breast cell division (reference below). Medroxyprogesterone, on the other hand, has shown conflicting results in studies that generally also included an exogenous source of estrogen. However, Depo-medroxyprogesterone, used alone as birth control, has clearly shown increased risk of breast cancer that diminishes on cessation of therapy. Certainly, this is an area that needs more research, but it suggests that progesterone may be a better choice than synthetic progestins, particularly for menopausal women with other risks for breast cancer.
Rates of bone growth and resorption change with the menstrual cycle, with increased activity during dominance of the progesterone cycle. Some have described progesterone as a “bone-trophic” hormone. Researchers have noted bone loss even in young women experiencing anovulatory cycles, and bone loss generally begins around age 35 when progesterone levels begin to diminish.
In the central nervous system, progesterone receptors can be detected in every neural cell type. Progesterone has many non-reproductive functions relating to cognition, mood, neurogenesis, myelination and more. It also aids in recovery from traumatic brain injury. Again, a synthetic progestin substitute would not have the same actions in the nervous system as our own hormone, progesterone, due to its different structure. This could have implications for menopausal women, affecting mood, cognition and memory. Anecdotally, I have had women report improved memory after changing from a synthetic progestin to progesterone. This is another area that deserves better research.
The indication for progestin replacement in standard hormone replacement therapy is to protect against endometrial hyperplasia due to exogenous estrogen. However, this may be because synthetic progestins are defined solely by their action on the uterus. However, we know that progesterone itself has actions throughout the body, many of which are not reproduced by synthetic progestins. Understanding the wide range of actions of progesterone, it makes sense (at least to me) to offer progesterone (but not a synthetic progestin) to women who do not have a uterus as well as to those who do.
Conclusion
I have several important learnings from my years of hormone studies:
- It can take many years for hormone therapy risks to manifest.
- Replacing hormones with the same molecules produced by the human body will maintain effects in tissues throughout the body, many of which are not monitored and may be unknown as yet.
- Using the lowest effective dose is likely to provide lower risk.
- Administering via the transdermal route avoids extensive liver metabolism and allows lower doses to be used, reducing the potential for side effects.
I suspect I may have given you some new food for thought and I’d encourage you to do additional reading to learn more. Most of what I learned was through conferences, networking with colleagues, and reading studies over the past 15 years. I have researched and added references for further reading below.
References:
Steroid Hormone (image) – Wikipedia https://en.wikipedia.org/wiki/Steroid_hormone
Natural Progesterone, The Multiple Roles of a Remarkable Hormone – John Lee, MD (an older text but with very interesting information)
Ovarian Aging and the Perimenopausal Transition: The Paradox of Endogenous Ovarian Hyperstimulation – NIH https://pubmed.ncbi.nlm.nih.gov/16034185/
Influences of Percutaneous Administration of Estradiol and Progesterone on Human Breast Epithelial Cell Cycle in Vivo (Chang, et al) – NIH https://pubmed.ncbi.nlm.nih.gov/7890063/
Effect of Depo-Medroxyprogesterone Acetate on Breast Cancer Risk – Cancer Research https://cancerre+s.aacrjournals.org/content/72/8/2028.long
Medroxyprogesterone Acetate-Driven Increase in Breast Cancer Risk… — Maturitas https://pubmed.ncbi.nlm.nih.gov/23856385/
Progesterone as a Bone-Trophic Hormone – Endocrine Review https://pubmed.ncbi.nlm.nih.gov/2194787/
Progesterone Receptors: Form and Function in Brain – NIH https://pubmed.ncbi.nlm.nih.gov/18374402/
Alligators and Endocrine Disrupting Contaminants: A Current Perspective – American Zoologist https://academic.oup.com/icb/article/40/3/438/379945

Jeannie Collins Beaudin is a recently retired Canadian community pharmacist with 40 years of experience, including specialization in compounding pharmacy, and hormone assessment and management. In addition to publishing her book, Can I Speak to the Hormone Lady? Managing Menopause and Hormone Imbalances in 2018, she has written articles and blogs for Pharmacy Practice + Business since 2006, peer reviewed educational programs for Canadian pharmacists since 1998 and publishes a weekly health blog found on her website.
Editor’s Note –The views expressed in the nurse influencer posts are those of the contributing authors. NPWH does not sanction the content of these posts.

