In this article, the author discusses how the PALM-COEIN classification system helps clinicians group various causes of abnormal uterine bleeding into structural and nonstructural etiologies, allowing assessment and management to be more specifically employed by nurse practitioners to offer women the most effective treatments.
Women’s health nurse practitioners frequently provide care for reproductive-aged women who have abnormal uterine bleeding (AUB), which comprises heavy periods, intermittent bleeding, and prolonged bleeding. It is estimated that 1.4 million women will experience AUB in the United States each year. This includes an estimated 50% of women in perimenopause who have an increased incidence of polyps, fibroids, and anovulation, leading to AUB.1 AUB accounts for as much as one-third of all gynecologic visits.2
The impact of AUB on a woman’s quality of life can be significant. In fact, over half of women with heavy and/or prolonged menstrual bleeding report that their periods interfere with their lives as compared to the response by women with lighter cycles.3 Work and school absences as well as loss of productivity can occur. AUB can interfere with sexual relationships. Many women lose sleep from frequent trips to the bathroom during the night to change their menstrual protection. Others experience more severe blood loss resulting in anemia. Most AUB can be managed medically or with minimally invasive treatments. When bleeding is particularly heavy or prolonged, a blood transfusion may be required to correct severe anemia. AUB may be a symptom of endometrial hyperplasia and endometrial carcinoma. Hysterectomy may be needed when significant abnormal bleeding persists, despite medical and minimally invasive treatments, and when cancer is diagnosed.
Terminology and classifications for abnormal uterine bleeding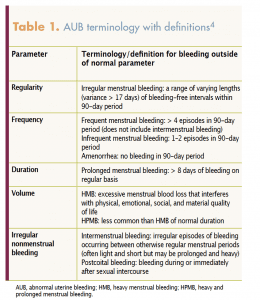
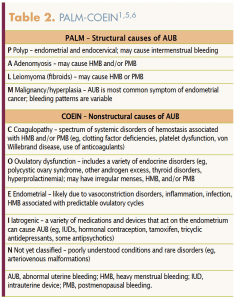
Two systems created by the International Federation of Gynecology and Obstetrics (FIGO) standardize terminology and provide a systematic approach to the evaluation of AUB in reproductive-aged women. The FIGO System 1 includes four parameters of menstrual bleeding with terminology and definitions for bleeding that fall outside of the normal for these parameters, which are regularity, frequency, duration, and volume (Table 1).4 FIGO System 2, the PALM-COEIN classification system, was created by FIGO to classify and separate the causes of AUB into structural and nonstructural etiologies.5 The first part of the acronym, PALM, succinctly defines the common structural causes as polyps, adenomyosis, leiomyoma (fibroids), and malignancy/hyperplasia. The second part of the acronym, COEIN, defines the nonstructural causes as coagulopathy, ovulatory dysfunction, endometrial, iatrogenic, and not yet classified. PALM-COEIN terminology helps healthcare providers base differential diagnoses and treatment options on systematic assessment using knowledge about the etiologies and common signs and symptoms for each potential one (Table 2).1,5,6
Assessment of abnormal uterine bleeding
The clinician begins the workup of AUB with a complete history, including a detailed focus on menstrual history, sexual history, current uterine bleeding pattern, any associated pain, contraception, medication use (including herbal remedies), other excessive bleeding, family history of AUB or other bleeding problems, and risk factors for cancer of reproductive tract organs. Any description of a change in menses that leads to a disruption in a woman’s usual lifestyle and interferes with her quality of life is relevant.
 The clinician conducts a comprehensive physical examination to include assessment for significant weight change, signs of androgen excess (eg, hirsutism, acne, alopecia), thyroid enlargement/nodules, and signs that may suggest a bleeding disorder (eg, petechiae, ecchymosis). A pelvic examination with speculum and bimanual examination is included for all adult women with AUB. Clinical judgment should be used regarding the extent of pelvic examination for younger adolescents who have never had sexual intercourse and may be limited to an external inspection. It is critical in reproductive-aged women with AUB to rule out pregnancy with a sensitive urine HCG [human chorionic gonadotropin] test. Other common initial laboratory testing dependent on characteristics of the AUB, age of the woman, and history and physical examination findings are a complete blood count, thyroid-stimulating hormone test, and cervical cancer screening per current recommendation.1,2,6 When anemia is identified, a ferritin level provides an indirect measurement of iron stores.1 If indicated by history or physical examination findings, a test for Chlamydia trachomatis should also be considered.1,2,6 When ovulatory dysfunction is suspected, additional hormonal tests such as follicle-stimulating hormone, estrogen, and prolactin levels may be helpful.1,6
The clinician conducts a comprehensive physical examination to include assessment for significant weight change, signs of androgen excess (eg, hirsutism, acne, alopecia), thyroid enlargement/nodules, and signs that may suggest a bleeding disorder (eg, petechiae, ecchymosis). A pelvic examination with speculum and bimanual examination is included for all adult women with AUB. Clinical judgment should be used regarding the extent of pelvic examination for younger adolescents who have never had sexual intercourse and may be limited to an external inspection. It is critical in reproductive-aged women with AUB to rule out pregnancy with a sensitive urine HCG [human chorionic gonadotropin] test. Other common initial laboratory testing dependent on characteristics of the AUB, age of the woman, and history and physical examination findings are a complete blood count, thyroid-stimulating hormone test, and cervical cancer screening per current recommendation.1,2,6 When anemia is identified, a ferritin level provides an indirect measurement of iron stores.1 If indicated by history or physical examination findings, a test for Chlamydia trachomatis should also be considered.1,2,6 When ovulatory dysfunction is suspected, additional hormonal tests such as follicle-stimulating hormone, estrogen, and prolactin levels may be helpful.1,6
Tests for a coagulopathy are indicated for all adolescents with heavy menstrual bleeding (HMB) and for women with frequent epistaxis or gum bleeding, easy bruising, postpartum hemorrhage, and bleeding associated with dental work.7 Approximately 5% to 24% of women with a long history of HMB since adolescence will have a coagulopathy such as von Willebrand disease. Initial screening when a coagulopathy is suspected should include complete blood count with platelets, prothrombin time, an activated partial thromboplastin time, and fibrinogen. Depending on the results of these initial tests, specific tests for von Willebrand disease and factor VIII may be indicated.7,8
Imaging
Several imaging modalities are available to assist with diagnosis depending on history and physical examination findings. These include transvaginal ultrasound, sonohysterography, and magnetic resonance imaging (MRI). Transvaginal ultrasound is useful to evaluate abnormal findings on bimanual examination or if an adequate examination cannot be completed due to obesity or pain. Uterus size and shape and ovaries can be assessed, the latter for size, masses, and presence of multiple follicular cysts consistent with polycystic ovary syndrome (PCOS). Endometrial thickness can also be assessed, but it is not considered helpful in the evaluation of premenopausal women with AUB because thickness normally varies throughout the menstrual cycle.2 Use of saline-infused ultrasound and sonohysterography separates the lateral walls of the uterus to better detect intracavitary lesions such as fibroids or polyps. Routine use of MRI is not recommended but may be helpful to guide treatment decisions when there are multiple myomas.2
Hysteroscopy allows a clinician to directly visualize the lining of the uterus using a small scope. Until recently, hysteroscopy was only performed in outpatient or inpatient surgery centers, but newer devices can be used in an office setting.9 With hysteroscopy, clinicians can evaluate the uterine cavity for polyps, features of adenomyosis, and the presence of submucosal fibroids. Hysteroscopes with multiple channels can be utilized to pass instruments through to eradicate and remove polyps and fibroids, as well as to obtain samples of the endometrium to assess for endometrial glands and stroma within the myometrium, which may signal adenomyosis.
Endometrial tissue sampling
Although infrequent, especially in reproductive-aged women, the possibility of endometrial hyperplasia and carcinoma must be considered. Endometrial tissue sampling is indicated for all women with AUB older than age 45 years, younger women with a history of unopposed or increased estrogen exposure such as may be seen with obesity, PCOS, or any condition causing anovulation, and for women who continue to have AUB despite medical treatment.10
Women younger than age 45 years, with new onset of AUB and an endometrial stripe that measures over 8 mm or with persistent AUB for 6 months or more and an endometrial stripe greater than 3 mm, should have an endometrial biopsy to rule out hyperplasia and carcinoma.10,11
Management of AUB
The goal of treatment for acute or chronic AUB is to reduce blood loss at the time and in the future and to correct anemia. Acute AUB is defined as an episode of bleeding in a woman of reproductive age, who is not pregnant, that is of sufficient quantity to require immediate intervention to prevent further blood loss.5 Patients with hemodynamic instability, defined as hypotension, orthostatic vital signs, and tachycardia with a hemoglobin level of 10 g/dL or less or those with hemoglobin levels below 7 g/dL, should be hospitalized.12 Acute AUB management usually includes high-dose estrogen either intravenously or orally or tranexamic acid intravenously or orally. Intrauterine tamponade with a catheter infused with 30 mL of fluid may also be used.1 Iron supplementation should be initiated for identified anemia with follow-up hemoglobin/hematocrit and ferritin levels to monitor return of appropriate iron stores.
Pharmacologic treatment options for chronic AUB
Chronic AUB is defined as uterine bleeding that is abnormal in duration, volume, and/or frequency and has been present for the majority of the last 6 months.5 This does not mean, however, that treatment should not begin as soon as the woman identifies a change that is significant to her.
When considering the treatment options for chronic AUB, it’s imperative to assess the woman’s desire for fertility at the current time and in the future. Treatment options that also provide contraception include the 52-mg levonorgestrel-releasing intrauterine system, combination oral contraceptives that may be taken cyclically or in extended or continuous regimens, depot medroxyprogesterone acetate, and the etonogestrel subdermal implant. These methods have been shown to be effective at reducing bleeding in women with adenomyosis, leiomyomas, coagulopathies, ovulatory dysfunction such as PCOS, and endometrial disorders.1,13 Other noncontraceptive progestins such as cyclic medroxyprogesterone acetate and oral micronized progesterone can be used for treatment of anovulatory bleeding.1,14 All of these methods also help to protect women with unopposed estrogen exposure from endometrial hyperplasia. None of these methods resolve any underlying structural or hormonal causes for HMB. Women with chronic AUB related to ovulatory dysfunction who desire a pregnancy in the near future should be referred to a fertility specialist.
For women with ovulatory heavy and/or prolonged menstrual bleeding who prefer not to take hormones or who are considering a pregnancy, over-the-counter nonsteroidal anti-inflammatory drugs (NSAIDs) and tranexamic acid are treatment options.1,6,14 NSAIDs started up to 3 days prior to menses or at the onset of menses may reduce the amount of bleeding and any discomfort and cramping. Tranexamic acid is an antifibrinolytic medication taken 3 times each day for 5 days during menses.14 Tranexamic acid should not be used in combination with estrogen-containing contraception and is contraindicated in women with a history of or risk for thrombosis.
A GnRH agonist, such as leuprolide acetate, nafarelin acetate, or goserelin acetate may be used short term, especially for women with HMB related to leiomyomas, to suppress ovarian function and induce amenorrhea, promoting a rise in hemoglobin and reduction in the size of the leiomyomas prior to surgical intervention. Treatment with GnRH agonists is limited to 6 months because of an increased risk for osteoporosis with estrogen suppression. If treatment is longer than 6 months, concomitant therapy with low doses of estrogen and progesterone are recommended to prevent bone loss.15
Minimally invasive treatment options
Women who have AUB from endometrial polyps and/or leiomyoma who also wish to preserve their fertility and avoid hysterectomy may be candidates for minimally invasive tissue removal. Hysteroscopic techniques have evolved and are known as “see-and-treat,” where the surgeon can look inside the uterus and remove abnormal tissue at the same time.
To perform hysteroscopic polypectomy and myomectomy, instruments are inserted into a hysteroscope and utilize tiny rotary or reciprocating cutters to shave off segments of submucosal fibroids while an integrated suction device removes the tissue fragments. These have become the gold standard of treatment, with success rates in the range of 85% to 95%.16
Endometrial ablation
For women who have completed childbearing, endometrial ablation removes a thin layer of the endometrium, utilizing instruments that are passed through the cervix into the uterine cavity. The procedure is low risk and efficient, with a short recovery time and no incision, and has a 98% reduction in bleeding at 12 months and 65% amenorrhea rates at 3 years.17
Although endometrial ablation may cause infertility, it’s recommended that reproductive-aged women who are sexually active continue to use contraception after an ablation because pregnancies that do occur can have serious consequences. Some of these consequences are: miscarriage, ectopic pregnancy, placenta accreta, preterm delivery, intrauterine growth restriction, intrauterine fetal demise, and uterine rupture.18
Uterine artery embolization
Uterine artery embolization is a minimally invasive procedure that shrinks uterine fibroids. During the procedure, an interventional radiologist identifies the blood vessels that lead to the specific fibroid and injects clot-forming particles into them. Over time, the fibroid, deprived of its blood supply, shrinks in size.19
Dilation and curettage
While dilation and curettage may be employed with other modalities to help evacuate the tissue and blood within the uterus, when utilized by itself, it is inadequate and will only provide temporary relief.11
Hysterectomy
In the past, hysterectomy was one of the most common surgical procedures in the United States.19 As more minimally invasive treatment options have been utilized to treat AUB, the number of open hysterectomies has declined and robotic-assisted laparoscopic hysterectomies have become more common due to reduced postoperative side effects. Although hysterectomy remains the most effective treatment, it also carries the highest risks, requires the longest recovery time, and can impact a woman’s mood, self-image, and sexual response, which is why this option should be reserved for women who have failed other options and who have been appropriately counseled.20
Implications for practice
The clinician who understands normal and abnormal parameters for menstrual bleeding can utilize PALM-COEIN to guide assessment that includes history, physical examination, laboratory tests, imaging studies, and other diagnostic procedures to work through differential diagnoses and determine the cause for AUB. Shared decision making between clinician and patient for management of AUB revolves around diagnosis, the impact of bleeding on her quality of life, extent of anemia, her fertility desire, and any risks for a cancerous or precancerous cause. =
Barbara A. Dehn is a women’s health nurse practitioner with the El Camino Women’s Medical Group in Mountain View, California. She is a member of our Editorial Advisory Board. The author states that she does not have a financial interest in or other relationship with any commercial product named in this article.
References
- Marnach ML, Laughlin-Tommaso SK. Evaluation and management of abnormal uterine bleeding. Mayo Clinic Proc. 2019;94(2):326-335.
- Committee on Practice Bulletins—Gynecology. Practice bulletin no. 128: diagnosis of abnormal uterine bleeding in reproductive-aged women. Obstet Gynecol. 2012;120(1):197-206.
- Brito LG, Panobianco MS, Sabino-de-Freitas MM, et al. Uterine leiomyoma: understanding the impact of symptoms on womens’ lives. Reprod Health. 2014;11(1):10.
- Frasier IS, Critchley HO, Broder M, Munro MG. The FIGO recommendations on terminologies and definitions for normal and abnormal uterine bleeding. Semin Reprod Med. 2011;29(5):383-390.
- Munro MG, Critchley HO, Broder MS, Fraser IS; FIGO Working Group on Menstrual Disorders. FIGO classification system (PALM-COEIN) for causes of abnormal uterine bleeding in nongravid women of reproductive age. Int J Gynaecol Obstet. 2011;113(1):3-13.
- Wouk N, Helton M. Abnormal uterine bleeding in premenopausal women. Am Fam Physician. 2019;99(7):435-443.
- Elmaog˘ulları S, Aycan Z. Abnormal uterine bleeding in adolescents. J Clin Res Pediatr Endocrinol. 2018;10(3):191-197.
- Gray SH. Menstrual disorders. Pediatr Rev. 2013;34(1):6-17.
- Kolhe S. Management of abnormal uterine bleeding – focus on ambulatory hysteroscopy. Int J Womens Health. 2018;10:127-136.
- Shifren JL, Gass ML; NAMS Recommendations for Clinical Care of Midlife Women Working Group. The North American Menopause Society recommendations for clinical care of midlife women. Menopause. 2014;21(10):1038-1062.
- ACOG committee opinion no. 557: Management of acute abnormal uterine bleeding in nonpregnant reproductive-aged women. Obstet Gynecol. 2013;121(4):891-896.
- Armstrong AJ, Hurd WW, Elguero S, Barker NM, Zanotti KM. Diagnosis and management of endometrial hyperplasia. J Minim Invasive Gynecol. 2012;19(5):562-571.
- Lethaby A, Hussain M, Rishworth JR, Rees MC. Progesterone or progestogen-releasing intrauterine systems for heavy menstrual bleeding. Cochrane Database Syst Rev. 2015;(4):CD002126.
- Bitzer J, Heikinheimo O, Nelson AL, Calaf-Alsina J, Fraser IS. Medical management of heavy menstrual bleeding: a comprehensive review of the literature. Obstet Gynecol Surv. 2015;70(2):115-130.
- Lethaby A, Puscasiu L, Vollenhoven B. Preoperative medical therapy before surgery for uterine fibroids. Cochrane Database Syst Rev. 2017;11:CD000547.
- Tanos V, Berry KE, Frist M, Campo R, DeWilde RL. Prevention and management of complications in laparoscopic myomectomy. Biomed Res Int. 2018;2018:8250952.
- Gimpelson RJ. Ten-year literature review of global endometrial ablation with the NovaSure® device. Int J Womens Health. 2014;6:269-280.
- Kohn JR, Shamshirsaz AA, Popek E, et al. Pregnancy after endometrial ablation: a systematic review. BJOG. 2018;125(1):43-53.
- Kohi MP, Spies JB. Updates on uterine artery embolization. Semin Intervent Radiol. 2018;35(1):48-55.
- Rodrigues RC, Rodrigues MRK, Freitas NO, Rudge MVC, Lima SAM. Quality of life in patients who undergo conventional or robotic-assisted total laparoscopic hysterectomy: protocol for a systematic review of randomized controlled trials. Medicine (Baltimore). 2019;98(23):e15974.

