Continuing education approval: This activity has been evaluated and approved by the Continuing Education Approval Program of the National Association of Nurse Practitioners in Women’s Health (NPWH) for 1.0 continuing education contact hours including 0.33 contact hours of pharmacology content now through October 31, 2024.
Intended audience: This continuing education (CE) activity has been designed to meet the educational needs of nurse practitioners and other clinicians who provide healthcare for reproductive-age women.
Goal statement: Nurse practitioners and other clinicians who provide healthcare for reproductive-age women will increase their knowledge about the diagnosis and management of polycystic ovary syndrome (PCOS).
Needs assessment: Polycystic ovary syndrome is the most common endocrine–metabolic disorder among women of reproductive age and may begin as early as puberty. It impacts a woman’s physical health, mental health, and quality of life from onset of symptoms through menopause. Identifying PCOS early provides opportunities to initiate counseling and management during the reproductive years. An understanding of recommended assessment, diagnostic criteria, and management options for PCOS is essential to provide care that is appropriate and attentive to individual needs and desires.
Educational objectives: At the conclusion of this educational activity, participants should be able to:
1. Identify clinical indicators, laboratory tests, and imaging studies helpful in the diagnosis of PCOS.
2. Discuss common goals of management for the individual with PCOS.
3. Describe indications, mechanism of action, efficacy, adverse events, and contraindication for pharmacologic options in treating PCOS.
Terrie H. Platt, DNP, RN, WHNP-BC, NCMP, has no actual or potential conflicts of interest in relation to this article.
Disclaimer: Participating faculty members determine the editorial content of the CE activity; this content does not necessarily represent the views of NPWH. This content has undergone a blinded peer review process for validation of clinical content. Although every effort has been made to ensure that the information is accurate, clinicians are responsible for evaluating this information in relation to generally accepted standards of care and integrating the information in this activity with that of established recommendations of other authorities, national guidelines, and individual patient characteristics.
Commercial support: This activity did not receive any commercial support.
Successful completion of the activity: Successful completion of this activity, J-22-05, requires participants to do the following:
1. “Sign in” at the top right-hand corner of the page if you have an NPWH account. You must be signed in to receive credit for this course. If you do not remember your username or password, please follow the “Forgot Password” link and instructions on the sign-in page. If you do not have an account, please click on the “Create an Account.”*
2. Read the learning objectives, disclosures, and disclaimers.
3. Study the material in the learning activity during the approval period (now through October 31, 2024).
4. Complete the post-test and evaluation. You must earn a score of 70% or higher on the post-test to receive CE credit.
5. Print out the CE certificate after you have successfully passed the post-test and completed the evaluation.
*If you are an NPWH member, were once a member, or have completed CE activities with NPWH in the past, you have a username and password in our system. Please do not create a new account. Creation of multiple accounts could result in loss of CE credits as well as other NPWH services. If you do not remember your username or password, either click on the “Forgot Username” or “Forgot Password” link or call the NPWH office at (202) 543-9693, ext. 1.
Polycystic ovary syndrome (PCOS) is the most common endocrine–metabolic disorder among women of reproductive age and may begin as early as puberty. PCOS impacts a woman’s physical health, mental health, and quality of life from onset of symptoms through menopause. The goal is to identify PCOS early and provide opportunities to initiate counseling and management during the reproductive years. This article provides an overview of the etiology and pathogenesis of PCOS and examines recommended assessment, diagnostic criteria, patient counseling, and management options.
Polycystic ovary syndrome (PCOS) with a clinical presentation of amenorrhea, hirsutism, obesity, and an associated finding of enlarged, polycystic ovaries was first described in 1935.1 Subsequently, diagnostic criteria and guidelines for assessment and treatment have been developed, but currently PCOS remains underdiagnosed and misunderstood by most providers.2,3 The comorbidities associated with PCOS include diabetes mellitus, dyslipidemia, obesity, hypertension, metabolic syndrome, depression, anxiety, obstructive sleep apnea, nonalcoholic fatty liver disease, endometrial cancer, and cardiovascular disease.4 Among women with PCOS, subfertility, a delay in conceiving, is common and the incidence of complications in pregnancy is high. Associated pregnancy complications include gestational diabetes, preeclampsia, and hypertension as well as increased risk of preterm birth, neonatal hypoglycemia, and neonatal hyperbilirubinemia.5 The clinical features of PCOS, particularly hirsutism and weight gain, can significantly impact quality of life (QOL).6 The subfertility experience reflects negatively on QOL due to resultant psychological and emotional distress. Nurse practitioners (NPs) who provide women’s healthcare must be aware of the long-term health risks associated with PCOS and prepared to provide appropriate assessment, counseling, and management options. The goal is to identify PCOS early and provide opportunities to initiate counseling and management during the reproductive years.4 This article addresses prevalence, pathophysiology, assessment and diagnostic considerations, and treatment options to aid NPs in recognizing PCOS and providing evidence-based patient counseling and management.
Epidemiology
Polycystic ovary syndrome is the most common endocrine– metabolic disorder among women of reproductive age and may begin as early as puberty.2,3,7 Classic features are manifested in 3% to 7% of the population and in up to 20% of women under the broadest definition.1,8 It is an inherited, complex polygenic, and multifactorial disorder.2 Environmental factors, including diet and lifestyle, interact with genetic variants and lead to PCOS.9 Epigenetic initiators may include fetal exposure to excess androgens, intrauterine growth restriction due to hypertension, diabetes, or other pregnancy complications, environmental chemicals, certain drugs, the aging process, and diet.9,10
Key diagnostic features of PCOS are hyperandrogenism and ovulatory dysfunction.2,11,12 Subfertility as a result of oligo- or anovulation is common among reproductive-age women with PCOS.2 Overweight, obesity, metabolic dysfunction, vascular dysfunction, malignancy, subfertility, increased risk of pregnancy complications, and mood disorders are features and morbidities associated with PCOS.9,12,13 Among women with PCOS, an increased risk of multiple mental health conditions exists including anxiety, depression, and bulimia.7 This disorder impacts a woman’s physical health, mental health, and QOL from onset of symptoms through menopause. Perimenopausal and menopausal women with PCOS have increased rates of obesity, hyperinsulinemia, hypertension, and hyperlipidemia as well as an increased risk of endometrial and ovarian cancer.13
Pathogenesis
The pathophysiology of PCOS is complex and not yet fully understood. Insulin resistance is the most prevalent metabolic disturbance with PCOS among women with both normal-range body mass index (BMI) and obesity.11,12 Insulin resistance and resultant hyperinsulinemia are central components to the pathogenesis of PCOS.9,11,12 Weight gain and obesity contribute to the degree of insulin resistance and metabolic disturbances. Visceral obesity has a greater impact on insulin resistance than overall weight gain and may be present in women with PCOS who present with a normal-range BMI.11 Women with PCOS are at increased risk of impaired glucose tolerance and type 2 diabetes.
Another key component of PCOS is hyperandrogenism. Biochemical androgen levels show a positive correlation to the degree of insulin resistance among women with PCOS, indicating there is a relationship between the two in its pathogenesis.11 Insulin resistance and the resultant hyperinsulinemia play a pathogenic role in the androgen excess and oligo- or anovulation recognized among women with PCOS.11 Hyperinsulinemia decreases production of sex hormone-binding globulin (SHBG) by the liver and increases production of androgens by the ovary. A decrease in SHBG levels leads to an increased level of free testosterone in the blood and promotion of androgenic activity.9
Neuroendocrine contributors to the clinical presentation of PCOS include increased frequency of the pulsatile secretion of gonadotropin-releasing hormone (GnRH), which results in increased luteinizing hormone (LH) secretion and an increased LH/follicle-stimulating hormone (FSH) ratio.1,2,11 The increased levels of LH stimulate androgen secretion from the ovarian theca cells. At the ovarian level, follicles are resistant to the stimulating effect of FSH. Elevated levels of LH throughout the cycle and resistance of the follicles to FSH results in the polycystic ovarian morphology (PCOM) common to PCOS with the presence of multiple immature follicles. The “cystic” in PCOS refers to these arrested or immature follicles. Disturbances in the hypothalamic-pituitary axis combined with hyperinsulinemia result in the typical manifestations of PCOS, which are ovulatory dysfunction, hyperandrogenism, and PCOM.2
Ovulatory dysfunction manifests as menstrual irregularity or oligomenorrhea and subfertility. Hyperandrogenism contributes to skin manifestations, insulin resistance, metabolic dysfunction, and abdominal/visceral adiposity. Metabolic dysfunction increases the risk for dyslipidemia, hyperglycemia, and hypertension.2,12 Oligo- or anovulation and hyperinsulinemia increase the risk for endometrial hyperplasia and endometrial carcinoma. An increased risk of anxiety and depression is recognized among women diagnosed with PCOS, and the risk is correlated with the degree of androgen excess and hyperinsulinemia.2
Clinical presentation and assessment
Clinical indicators of PCOS include hirsutism, acne, menstrual irregularity, subfertility , male pattern hair loss, acanthosis nigricans, and acrochordon or skin tags, which are consistent with the typical presenting concerns.1,2,8 This disorder manifests in women with BMIs across all ranges.11 The presence of overweight/obesity is not a reliable clinical indicator of PCOS, and the prevalence of overweight and obesity among women without PCOS is similar to the prevalence of overweight and obesity among women with PCOS.2
The two key clinical indicators are excess male-like body or facial hair and a history of menstrual irregularity or oligomenorrhea.2 A postpubescent female presenting with clinical indicators and/or concern(s) consistent with PCOS requires a comprehensive assessment. Initial assessment should include exploration of the chief concern(s); review of personal medical, surgical, and social history as well as family history; complete review of systems; and complete physical examination.
In the presence of an irregular menstrual cycle, PCOS should be considered. The 2018 International Evidence-Based Guideline for the Assessment and Management of Polycystic Ovary Syndrome defined irregular menstrual cycles with variations based on time since menarche. During the first-year post menarche, irregular menstrual cycles are considered normal. A less than 21-day cycle or a greater than 45-day cycle is considered irregular greater than 1 year post menarche to less than 3 years post menarche. Greater than 3 years post menarche to perimenopause, a cycle less than 21 days or greater than 35 days (< 8 cycles per year) is defined as an irregular menstrual cycle. A single cycle greater than 90 days after 1 year post menarche meets the guideline’s criteria for an irregular menstrual cycle. In addition, the presence of primary amenorrhea at age 15 years or when more than 3 years’ post breast development meets the definition for an irregular menstrual cycle.
Hirsutism, excess male-like body or facial hair, is the most common physical finding associated with hyperandrogenism.2 The degree of hirsutism is a predictor of the degree of metabolic dysfunction. The modified Ferriman-Gallwey (mFG) scoring system is recommended for assessing the degree of hirsutism.2,3 Terminal hair growth in areas that males typically grow terminal hair is determined with the use of this visual scale. Terminal hairs grow greater than 5 mm in length and have both shape and pigment. The mFG includes the scoring of nine body areas: upper lip, chin, chest, arm, upper abdomen, lower abdomen, upper back, lower back, and thighs. Scores range from 1, minimal terminal hair growth, to 4, equivalent to a well-developed male. The availability of hair removal options necessitates inquiry about hair removal practices, and scoring should be completed prior to hair removal.
The purpose of diagnostic laboratory tests is to identify alternate or concurrent pathologies and to screen for comorbidities. Table 1 provides an overview of the recommended laboratory tests. A thyroid-stimulating hormone level is indicated to screen for thyroid dysfunction. The indication for a prolactin level is to exclude hyperprolactinemia. In the presence of signs of hyperandrogenism, a 17-hydroxyprogesterone level is recommended to rule out classic adrenal hyperplasia.2 In consideration of the increased risk of impaired glucose tolerance and type 2 diabetes among women with PCOS, a 2-hour 75-g oral glucose tolerance test (OGTT) is recommended at the time of diagnosis and every 1 to 3 years thereafter.2,3,7 A 2-hour 75-g OGTT also is recommended prepregnancy or at less than 20 weeks’ gestation.3 A baseline fasting lipid profile is recommended at diagnosis regardless of age and every 2 to 3 years thereafter.2,3 In the presence of obesity and/or marked hyperinsulinemia, baseline liver function testing should be completed.2
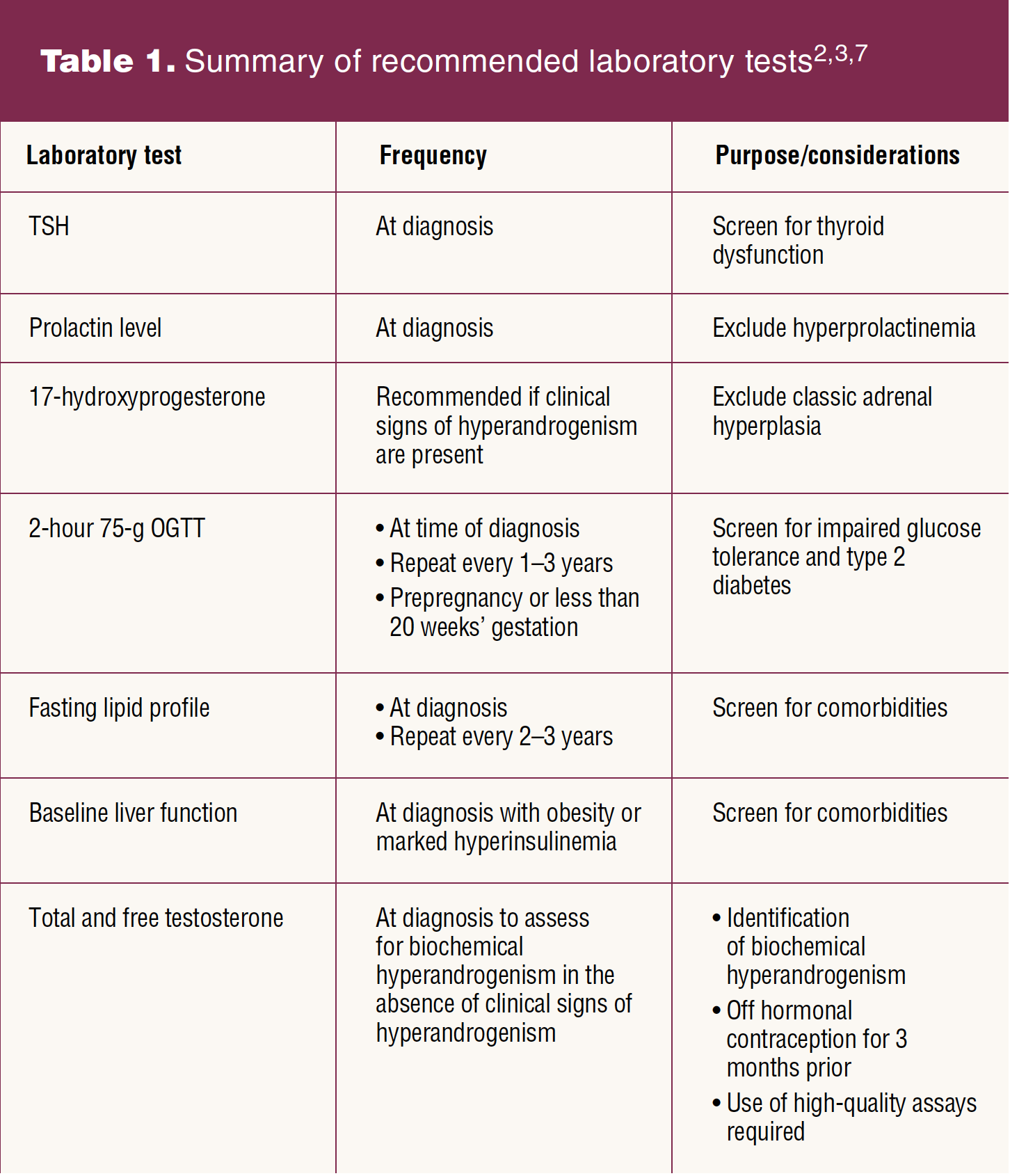
Laboratory assessment of biochemical hyperandrogenism has diagnostic usefulness when the clinical signs of hyperandrogenism are not evident.2,3 Discontinue hormonal contraception for a minimum of 3 months prior to assessment of biochemical hyperandrogenism.3 Hormonal contraception affects SHBG and alters gonadotrophin- dependent androgen production. Biochemical assessment of hyperandrogenism requires the use of high-quality assays to determine both a total and free testosterone level. One abnormal value is required for a diagnosis of hyperandrogenemia. Normative ranges for the specific laboratory must be defined and utilized by the provider for accurate interpretation of results.2,3
If the individual has had vaginal sexual intercourse and consents, transvaginal ultrasonography is the preferred method of evaluation for PCOM.1–3 Parameters measured are total follicle count in each ovary and ovarian volume. The 2018 evidence-based guidelines on PCOS suggest that with use of a transvaginal ultrasound transducer with a frequency bandwidth that includes 8 MHz, the diagnostic threshold for PCOM should be a follicle number per ovary greater than or equal to 20 on either ovary and/or an ovarian volume greater than or equal to 10 mL in the absence of corpora lutea, cysts, or dominant follicles.3 With the use of older ultrasound technologies and/or the transabdominal approach, the threshold is an ovarian volume of greater than or equal to 10 mL on either ovary. In the presence of irregular menstrual cycles and hyperandrogenism, ultrasound is not necessary in diagnosis. In the United States, 10% to 25% of the reproductive-age population of females will exhibit polycystic-appearing ovaries without other signs or symptoms of PCOS. Ultrasound to assess for PCOM is not recommended within 8 years of menarche as many adolescents will exhibit PCOM.3
Serum anti-mullerian hormone (AMH) level is controversial as a marker for PCOM in PCOS. AMH is produced in the granulosa cells of early developing follicles and correlates with the number of ovarian preantral follicles.1 The correlation between AMH level and the antral follicle count with ultrasound has been documented. But studies also have demonstrated an overlap between AMH levels among women with PCOS who have evidence of PCOM on ultrasound and those who do not. Current evidence does not support the use of serum AMH as a marker for the detection of PCOM or as a single test for the diagnosis of PCOS.3
As women with PCOS age, improvements in menstrual irregularity, biochemical hyperandrogenism, and ovarian morphology are recognized. Perimenopausal women with PCOS will often experience improved menstrual cycle regularity and ovarian volume and the number of follicles decreases. Androgen levels remain higher among women with PCOS as they age than among similarly aged women without PCOS. Criteria for a postmenopausal diagnosis of PCOS are a history of premenopausal menstrual dysfunction and hyperandrogenism during the reproductive years.4,14
Diagnosis
The European Society for Human Reproduction and Embryology and the American Society of Reproductive Medicine met in Rotterdam in 2003 and established the Rotterdam criteria for the diagnosis of PCOS. The diagnostic criteria include oligo- and/or anovulation, clinical and/or biochemical indicators of hyperandrogenism, and polycystic ovaries, and two of these must be present for a diagnosis of PCOS.8 Both the 2012 NIH Evidence-Based Methodology Workshop on PCOS and the 2018 International Evidence-Based Guideline for the Assessment and Management of Polycystic Ovary Syndrome recommend use of the Rotterdam criteria for the diagnosis of PCOS with the additional identification of the specific PCOS phenotype.3,15
The four recognized phenotypes of PCOS are: phenotype A (hyperandrogenism plus oligo- or anovulation plus PCOM), phenotype B (hyperandrogenism plus oligo- or anovulation), phenotype C (hyperandrogenism plus PCOM), and phenotype D (oligo- or anovulation plus PCOM).15 Phenotype A and B are referred to as classic PCOS phenotypes and constitute approximately two-thirds of the total number of women with PCOS. The NIH recommends reporting the specific phenotype in research and in clinical care.15 The specific phenotypes have differing long-term health and metabolic implications. For example, the classic phenotypes of PCOS, A and B, are associated with a higher incidence of insulin resistance and metabolic dysfunction.8
Management
Among women diagnosed with PCOS, the patient’s phenotype and personal concerns and goals guide therapeutic choices. The goals of management include suppressing androgen secretion, protecting the endometrium, improving menstrual dysfunction, improving metabolic status, and improving ovulatory fertility.2,3 In addition, consideration should be given to QOL and mental health concerns. A QOL screening tool specific to PCOS, the Polycystic Ovary Health Related Quality of Life Questionnaire (PCOSQ) or the modified PCOSQ, may be used to help determine patient concerns and priorities.3,6,8,16 The modified PCOSQ includes an acne domain. Screening for anxiety and depression is recommended in both adolescent and adult women at diagnosis.3 The increased tendency for weight gain among women with PCOS as well as an increased risk for negative body image, psychosexual dysfunction, and eating disorders necessitates ongoing screening and counseling.3 Lifestyle modifications are considered the first line of treatment for PCOS.3 Adherence to healthy eating principles and regular physical activity will help maintain weight, optimize hormonal outcomes, and improve general health and QOL across the lifespan. Changes to diet or physical activity should be recommended to all women with PCOS in an effort to improve cardiometabolic risk profile.8 A 5% to 10% weight loss among women with PCOS who are overweight or obese has been shown to produce significant results with improvement in ovulatory and menstrual function, hyperandrogenism, and insulin resistance.2,3,9,12 No one specific diet plan has been identified to be superior to another, but a reduction in caloric intake and increased activity level are recommended with a goal of achieving and maintaining a healthy weight.3 Prepregnancy optimization of blood glucose, weight, diet, exercise, sleep, mental health, and emotional health is recommended to improve reproductive and obstetric outcomes.3,17
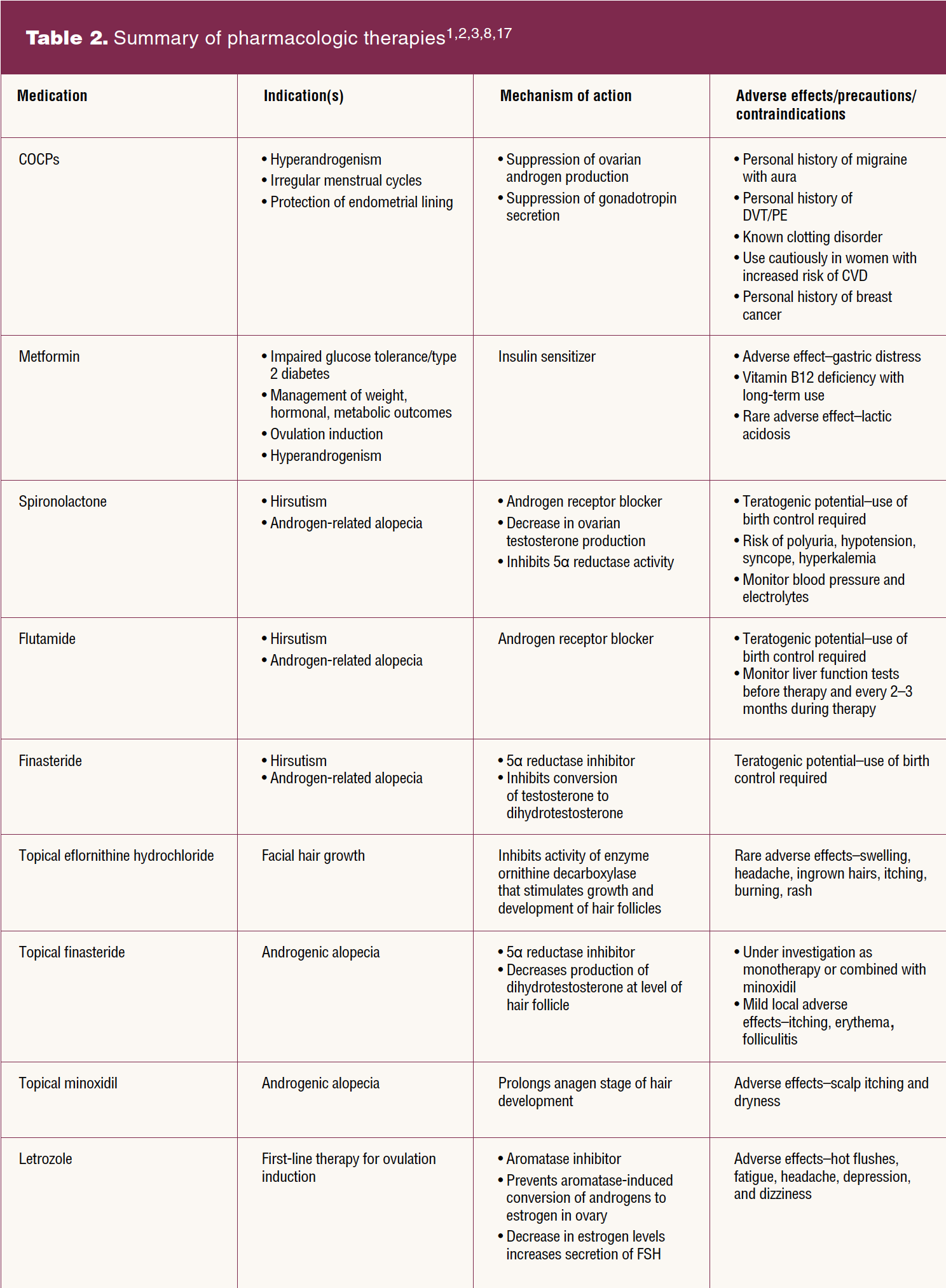
Pharmacologic treatment of PCOS should be individualized, symptom oriented, and combined with continued lifestyle counseling. Specific targets of pharmacologic therapies include androgen excess, ovulatory dysfunction, and insulin resistance. No drugs have been approved specifically for the treatment of PCOS.3,8 Table 2 provides a summary of pharmacologic therapies. Use of combined oral contraceptive pills (COCPs), metformin, antiandrogens, and all other treatments are off-label use in PCOS. Current evidence supports the use of COCPs to decrease androgen production by the ovary, suppress gonadotropin secretion, regulate menses, and protect the endometrial lining. The use of COCPs is recommended for both adolescents and adults with PCOS who are not currently seeking pregnancy.2,3,17 Consensus on a specific type or dose of COCPs has not been reached. Transdermal combination contraceptives or progestin only contraceptives may be considered but have a modest effect on androgen levels.2 Cyclic progestogen use will help protect the endometrium but will not achieve the goal of suppressing androgen production.
Metformin with or without a COCP is an option in addition to lifestyle modifications in adults and adolescents for the management of weight and the hormonal and metabolic features of PCOS.3 The first-line treatment for hirsutism is a COCP combined with cosmetic therapy. If the results are unsatisfactory following 6 months of therapy, use of an antiandrogen can be considered in combination with a COCP.3 Available antiandrogens include androgen receptor blockers such as spironolactone and flutamide and the 5α-reductase inhibitor finasteride.2 Combined therapy with an antiandrogen and COCP also is an option for the treatment of alopecia. Use of an antiandrogen alone for the management of hirsutism and/or alopecia can be considered if COCPs are not tolerated.3 Use of an antiandrogen without a COCP necessitates continued use of an alternative form of effective contraception due to the teratogenic potential for feminization of a male fetus. For patients with hirsutism, cosmetic and topical therapies are available. Topical eflornithine hydrochloride as well as bleaching, chemical epilation, shaving, electrology for destruction of the hair follicle, or laser epilation can effectively remove unwanted hair.2 Minoxidil as well as newer therapies such as topical finasteride, growth factors from platelet-rich plasma, or stem-cell-based therapies are available options for the treatment of alopecia.2,8,17
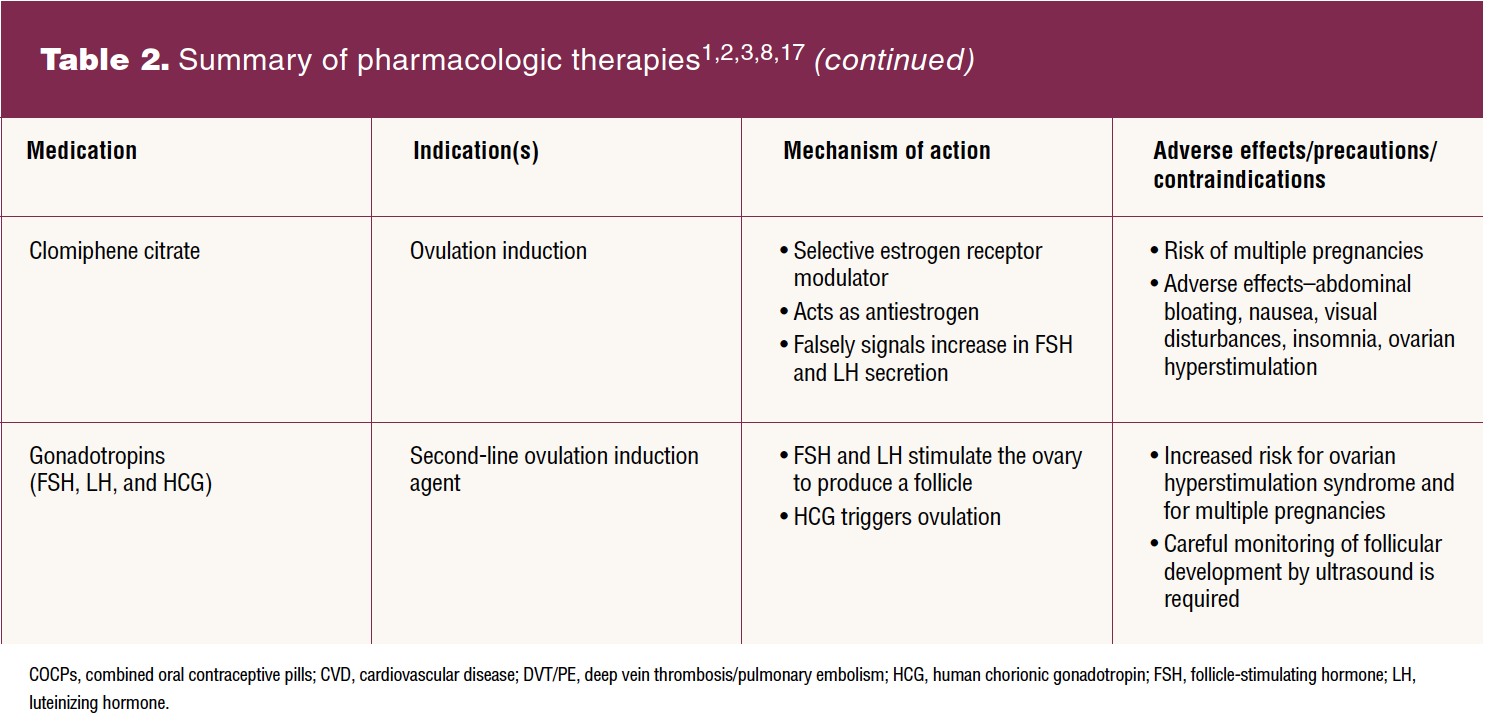
Among patients with anovulatory infertility and no other infertility factors, the aromatase inhibitor letrozole is the first-line pharmacologic treatment for ovulation induction.3,17 Clomiphene citrate and metformin, alone or together, are additional pharmacologic options for improving ovulation and pregnancy rates. Live birth rate is higher with letrozole use than with use of clomiphene citrate.17 Metformin alone is an affordable option but is less effective than other ovulation induction agents and has mild gastrointestinal adverse effects. Gonadotropins are second-line pharmacologic agents to be considered for women who fail to conceive with the first-line oral ovulation induction therapy.3,17 Women with PCOS are at increased risk of ovarian hyperstimulation syndrome and require careful monitoring. Low-dose therapy with gonadotropins is recommended to achieve a higher ovulation rate, development of a single follicle, and to lower the risk of ovarian hyperstimulation syndrome.17 Assisted reproductive technology therapies including in-vitro fertilization and intracytoplasmic sperm injection for women with PCOS and anovulatory infertility are available as third-line therapies.3 Laparoscopic ovarian surgery is a second-line therapy for PCOS-associated anovulatory infertility or as a first-line treatment if laparoscopic surgery is scheduled for another reason.3,17 The risks associated with laparoscopic ovarian surgery include reduced ovarian reserve and adhesion formation as well as the risks associated with any surgical intervention.
Conclusion
The most common endocrine–metabolic disorder among women of reproductive age, PCOS remains underdiagnosed and often misunderstood by providers. This disorder impacts QOL, mental health, reproductive health, and increases long-term health risks. Early diagnosis, screening for comorbidities, patient counseling, and evidence-based management are all key to improving outcomes among women with PCOS.
References
1 Lobo RA. Polycystic ovary syndrome. In: Lobo RA, Gershenson DM, Lentz GM, Valea FA, eds. Comprehensive Gynecology. 8th ed. Elsevier; 2022: 824-837.
2 Azziz R. Polycystic ovary syndrome. Obstet Gynecol. 2018;132(2):321-326.
3 Teede H, Misso M, Costello M, et al. International evidence-based guideline for the assessment and management of polycystic ovary syndrome. Monash University, 2018. https://www.monash.edu/medicine/sphpm/mchri/pcos.
4 Cooney LG, Dokras A. Beyond fertility: polycystic ovary and long-term health. Fertil Steril. 2018;110(5):794-809.
5 Li X, Liu X, Zuo Y, et al. The risk factors of gestational diabetes mellitus in patients with polycystic ovary syndrome: what should we care. Medicine. 2021;100(31):e26521.
6 Naumova I, Castelo-Branco C, Kasterina J, Casals G. Quality of life in infertile women with polycystic ovary syndrome: a comparative study. Reprod Sci. 2021;28(7):1901-1909.
7 Rollene NL, Lentscher JA, Hemphill AM, Decherney AH. Polycystic ovary syndrome and medical conditions. Clin Obstet Gynecol. 2020;64(1):110-118.
8 Escobar-Morreale HF. Polycystic ovary syndrome: definition, aetiology, diagnosis and treatment. Nat Rev. 2018;14(5):270-284.
9 Sanchez-Garrido MA, Tena-Sempere M. Metabolic dysfunction in polycystic ovary syndrome: pathogenic role of androgen excess and potential therapeutic strategies. Mol Metab. 2020;35:100937.
10 Barber TM. Why are women with polycystic ovary syndrome obese? Br Med Bull. 2022;ldac007.
11 Kamenov Z, Gateva A. Inositols in PCOS. Molecules. 2020;25(23):5566.
12 Jeanes YM, Reeves S. Metabolic consequences of obesity and insulin resistance in polycystic ovary syndrome: diagnostic and methodological challenges. Nutr Res Rev. 2017;30(1):97-105.
13 Matevossian K, Carpinello O. Polycystic ovary syndrome: menopause and malignancy. Clin Obstet Gynecol. 2020;64(1):102-109.
14 Lizneva D, Suturina L, Walker W, et al. Criteria, prevalence, and phenotypes of polycystic ovary syndrome. Fertil Steril. 2016;106(1):6-15.
15 National Institutes of Health. Evidence-based methodology workshop on polycystic ovary syndrome: executive summary. 2012. https://prevention.nih.gov/sites/default/files/2018-06/FinalReport.pdf.
16 Maghadam ZB, Fereidooni B, Saffari M, Montazere A. Measures of health-related quality of life in PCOS women: a systematic review. Int J Womens Health. 2018;10: 397-408.
17 American College of Obstetricians and Gynecologists. ACOG practice bulletin no. 194. Polycystic ovary syndrome. Obstet Gynecol. 2018;131(6):e157-e171.
Key words: polycystic ovary syndrome, insulin resistance, hyperandrogenism, ovarian dysfunction, infertility, irregular menstrual cycles
Womens Healthcare. 2022;10(5):8-14,42. doi: 10.51256/WHC102208

