Gestational diabetes (GDM) is a common complication characterized by glucose intolerance that develops during pregnancy. Diagnosis is made in the second or third trimester when insulin resistance increases secondary to diabetogenic placental hormones.1 Of all pregnancies, 2% to 10% are affected and rates have increased.2 People who are older, have a higher body mass index (BMI), or are carrying multiple gestations are more likely to be affected.3 The diagnosis increases the risk of developing a hypertensive disorder of pregnancy and often recurs. Individuals with a history of GDM are more likely to develop cardiovascular disease and type 2 diabetes later in life.4
Gestational diabetes increases the risk of preterm birth, stillbirth, macrosomia, shoulder dystocia, operative delivery, and birth trauma.5,6 Intensive management that starts with lifestyle changes can potentially decrease the need to initiate insulin therapy and maintain a relatively normal pregnancy course. Well-controlled GDM reduces incidence of macrosomia, rate of shoulder dystocia, bone fracture and nerve palsy, premature birth, neonatal intensive care unit admissions, and neonatal deaths.3 Patients with GDM express that they are given insufficient information and support as they face a steep learning curve to manage this diagnosis.7–9 Despite GDM’s significant impact on both maternal and fetal outcomes, consensus is lacking around optimal screening methods, diagnosis, and management. The purpose of this article is to provide practicing clinicians with an up-to-date overview of current screening and diagnosis guidelines and to describe nonpharmacologic and pharmacologic therapy best practices to improve health outcomes for patients with GDM.
Screening and diagnosis
Elevations in hemoglobin A1c higher than 6% to 6.5% during the first 10 weeks of pregnancy may increase the risk of diabetic embryopathy including congenital brain, heart, and renal anomalies.1,10 Prepregnancy and first-trimester screening for preexisting diabetes is made more difficult by lack of data and consensus on appropriate diagnostic thresholds and outcomes.1
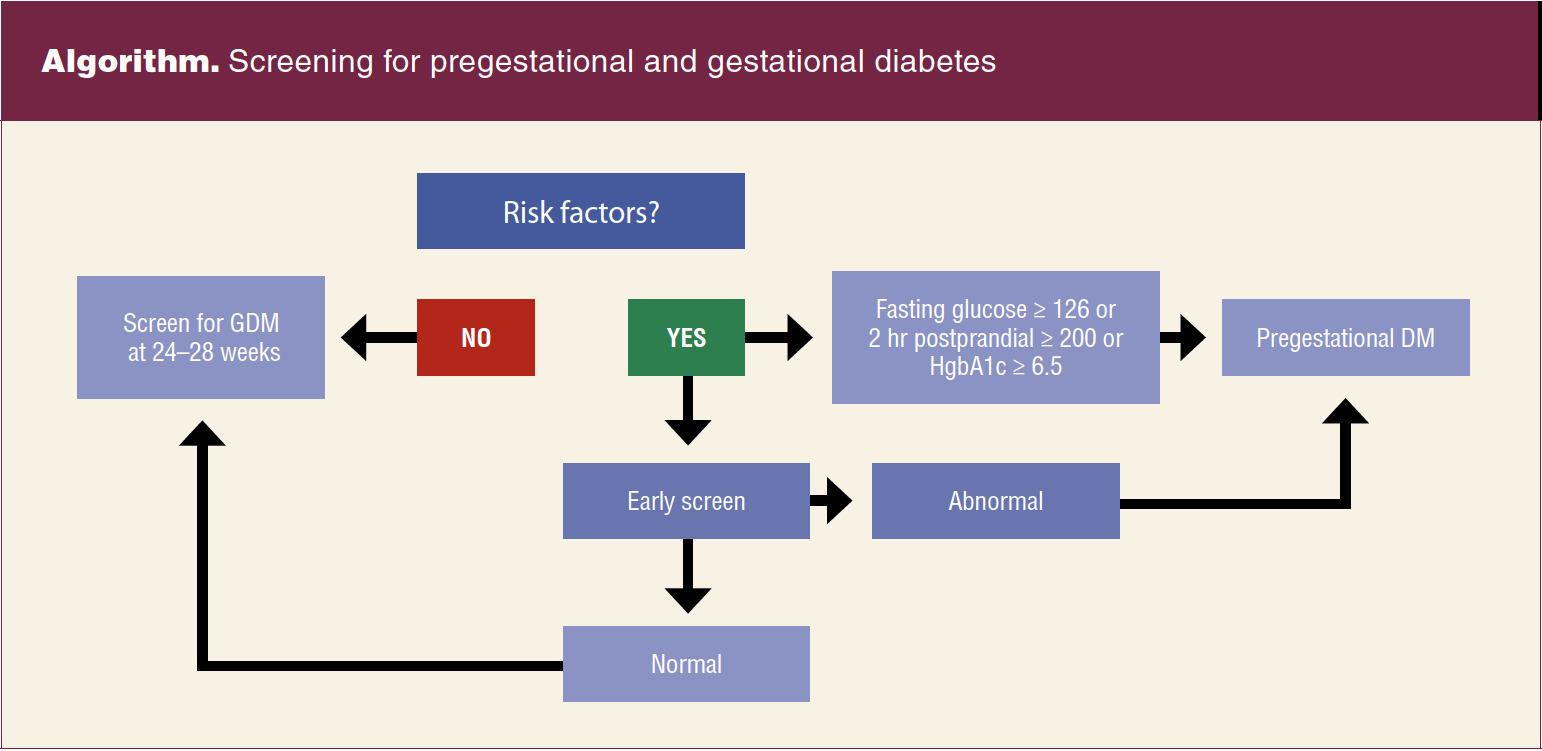
Consider screening for undiagnosed type 2 diabetes early in pregnancy for those with a BMI of 25 or more, or 23 or more in Asian Americans, with at least one additional risk factor (Box).1,5 The American College of Obstetricians and Gynecologists (ACOG) and the American Diabetes Association (ADA) recommend utilizing the same screening tests used to diagnose type 2 diabetes in early pregnancy as those used in nonpregnant populations including the 2-hour 75-g oral glucose tolerance test (OGTT) and/or hemoglobin A1c.1,5,11 Many providers continue to use the two-step screening method, a 50-g OGTT followed by 100-g 3-hour diagnostic OGTT if elevated, for early and third trimester screening.5
Diagnosis of pregestational diabetes is made when the fasting glucose is 126 mg/dL or higher, the 2-hour postprandial level is 200 mg/dL or higher, or the hemoglobin A1c is 6.5% or higher (Algorithm).10 Patients with hyperglycemia in the first half of pregnancy (< 20 weeks) are diagnosed with pregestational diabetes. Those with glucose intolerance any time after 20 weeks of gestation are classified as having GDM.11
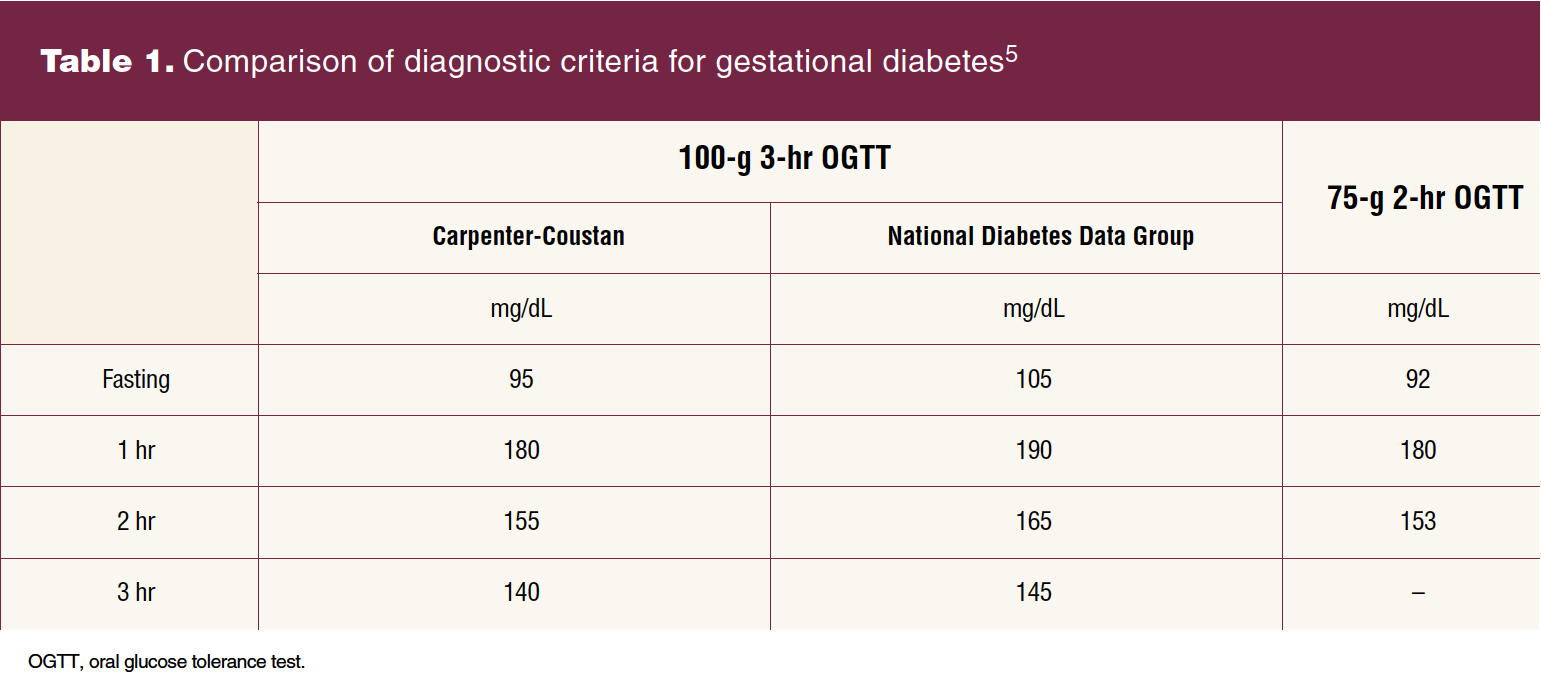
Given that GDM carries significant maternal, fetal, and neonatal risks, clinical practice guidelines recommend universal screening between 24 and 28 weeks.5,12,13 Although ACOG endorses the 2-step approach to screen and/or rescreen individuals at 24 to 28 weeks, variability in diagnostic ranges leads to confusion and inconsistent diagnosis. Evidence supports a cutoff range between 130 mg/dL and 140 mg/dL for the 1-hour OGTT and variable cutoff ranges for the 3-hour OGTT (Table 1).5 Utilizing the lower range increases diagnosis but does not support improved outcomes. Typically, diagnosis is made when two of the four values are elevated, but adverse outcomes have been demonstrated with one elevated value.5 If an individual has an initial abnormal 1-hour OGTT but normal 3-hour OGTT values, they should be retested in the third trimester with the 3-hour test bypassing the first step.
The International Association of Diabetes and Pregnancy Study Group and the ADA endorse the universal use of a one-step approach with a 75-g 2-hour OGTT. Diagnosis is made when one of the three values is elevated (Table 1).5 This method increases diagnosis, healthcare costs, and maternal stress without clearly improving outcomes. Although some populations may benefit from this approach, more evidence is needed to endorse universal change.5
The variation in screening recommendations and diagnostic thresholds is confusing. Recognizing that there are many paths to screening and diagnosis, the ultimate goal is to identify insulin resistance and GDM. Institutions and individual practices must establish and consistently use standardized screening and diagnostic criteria based on the current literature within the context of their own patient population and available resources.1,5,10
Once diagnosed, patients should monitor their capillary glucose levels daily, fasting and 1 to 2 hours postprandially to maintain strict glycemic control.3,5,7,10,11 Fasting levels should be lower than 95 mg/dL and postprandial values lower than 140 mg/dL at 1 hour or 120 mg/dL at 2 hours.1,2 There is insufficient evidence defining the optimal frequency of monitoring, but once levels are well controlled, frequency can be modified. Despite the disruptive and time-consuming practice of glucose monitoring, even those with optimal control should continue to measure levels at least twice daily.5
Continuous glucose monitoring (CGM) allows for the monitoring of blood glucose levels over the course of the day. It is typically used in those with type 1 and type 2 diabetes.14 In patients with GDM treated with insulin, CGM versus standard monitoring can be considered, but evidence is limited that this reduces adverse outcomes.10,15
Nonpharmacologic therapy
Management of GDM should take a multidisciplinary approach.16 The primary goal is to prevent fetal overgrowth and pregnancy complications.6 Lifestyle modifications including medical nutrition therapy (MNT) and the promotion of physical activity are the cornerstone of treatment and should be initiated with diagnosis. Dietary changes aim to encourage a healthful diet that achieves and maintains normal glycemia, minimizes postprandial hyperglycemia, prevents ketosis, and promotes appropriate pregnancy weight gain and adequate fetal growth. Regular physical activity aims to increase glucose uptake and insulin sensitivity in skeletal muscle and reduces postpartum weight retention.6
Medical nutrition therapy is ideally managed by a registered dietician (RD) with expertise in GDM. Dietary recommendations should be individualized and include patient input.1,10 Registered dietitians typically complete a comprehensive dietary assessment that includes medical, sociocultural, psychosocial, and economic factors.3 Regular and frequent MNT visits that are personalized effect greater change.3,17 The plan should be based on the comprehensive assessment and guidance from the Dietary Reference Intakes. It should include a minimum of 175 g of carbohydrates, 71 g of protein, and 28 g of fiber while limiting saturated fats.1 This diet includes mainly low glycemic index carbohydrates and divides daily caloric intake into three main meals and two to four snacks, distributing carbohydrate intake and reducing postprandial glucose fluctuations. Eating complex over simple carbohydrates promotes slower digestion and is less likely to cause postprandial hyperglycemia while reducing insulin resistance ensuring sufficient fetal growth and supporting cerebral development and function.5,6 In resource-poor settings in which RDs are not available, clinicians should guide patients with high-quality resources.5,7 Telehealth technologies can be used to improve pregnancy care by increasing patient engagement and provider communication.17 Patient education is the foundation of GDM management and virtual visits are an effective and efficient way for diabetic educators to provide nutrition counseling and self-management education including insulin teaching. Virtual technologies can improve and enhance the management of GDM when local resources are not available. Remote patient monitoring technologies are effective, allowing glucose logs to be reviewed and managed remotely by clinicians and educators.18
Analyzing the effects of exercise alone is difficult. Most clinical interventions use a combination of diet and exercise. Timing and duration of physical activity are not well established. Regular aerobic exercise for 30 minutes, five times a week, or 150 minutes is recommended.10 Multiple mobile apps targeted at lifestyle modifications, exercise, and specifically diabetes including GDM are easily accessible and may be beneficial. Few have been validated and it remains unclear how effective they are and how best to integrate them into pregnancy care.19 The majority of patients (70%–85%) can manage GDM with lifestyle modifications.1,16,20 Patients who can effectively manage their diagnosis feel empowered to integrate recommended treatments into their daily routines.3,5,7,10 These interventions can potentially prevent the development of recurrent GDM or delay the onset of type 2 diabetes.20 Patients who can achieve glucose control with lifestyle modifications alone are referred to as A1GDM. Those who continue to have hyperglycemia and require medical therapy to achieve euglycemia receive the A2GDM diagnosis.11
Pharmacologic therapy
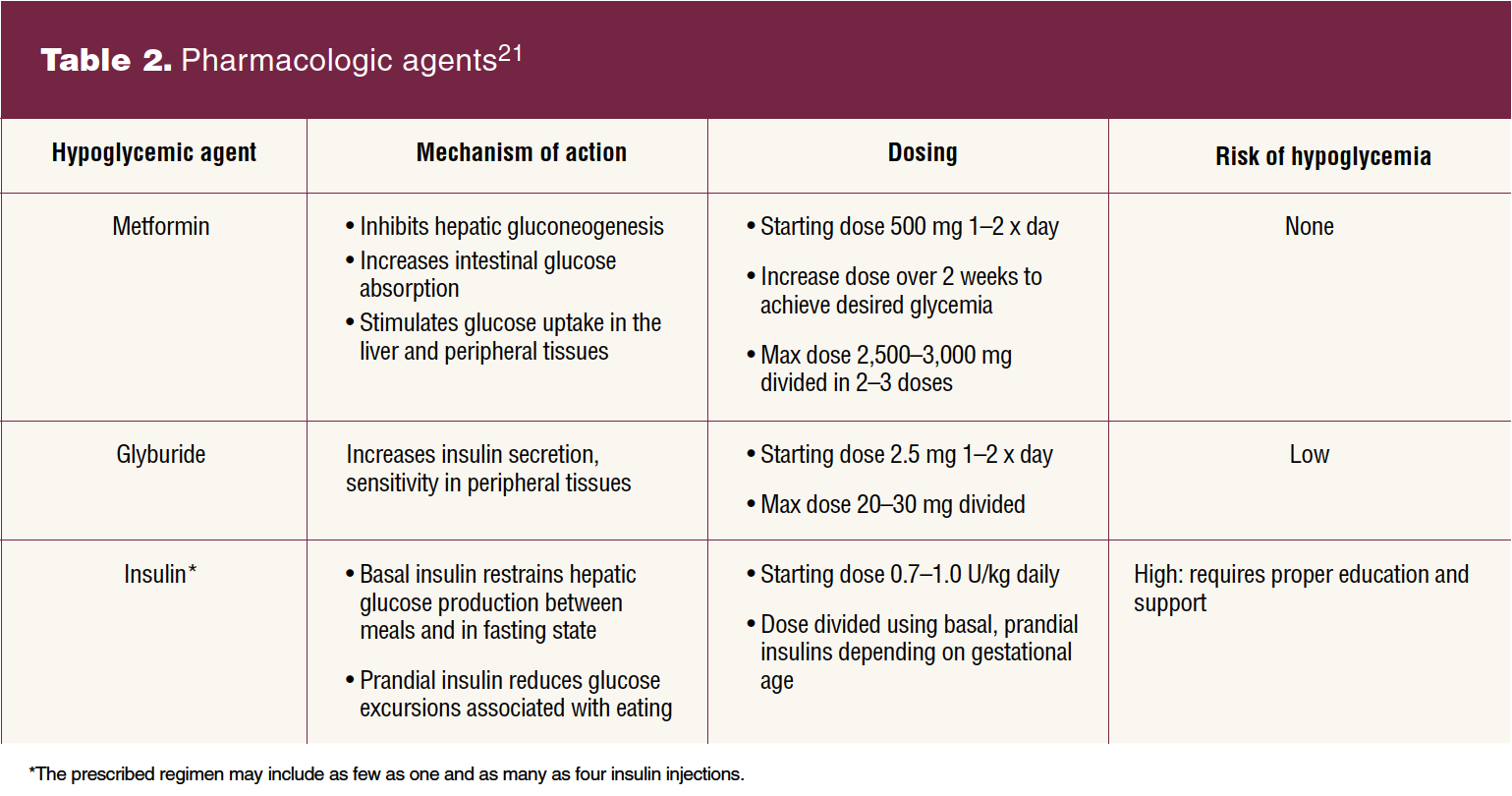
If target glucose levels cannot be achieved and greater than 30% of values remain elevated after 1 to 2 weeks, pharmacologic treatment is recommended.10,21 Insulin, metformin, and glyburide are used (Table 2).21 Insulin therapy is recognized by ACOG and the ADA as the first-line agent. The Society for Maternal Fetal Medicine continues to support oral hypoglycemic agents as first-line therapy because they are less expensive and more accepted, potentially increasing adherence and satisfaction.22 Patients report that they receive inadequate information about these options. Their needs and preferences should be taken into account when initiating medication.8
Insulin does not cross the placenta and has been safely used for decades.8 Insulin therapy requires significant education. Patients need to learn to manage daily injections while closely monitoring glucose levels and titrating insulin to maintain euglycemia. As maternal insulin resistance increases with advancing gestational age, a commonly used protocol uses maternal weight and gestational age to calculate starting daily insulin requirements.11 If fasting and postprandial hyperglycemia are present, long- and intermediate-acting insulin are used.5 Short-acting insulin is used to target isolated and specific abnormal levels. Ideally, patients requiring insulin therapy are managed by a specialized multidisciplinary team, which includes a certified diabetes care and education specialist, clinician(s) with expertise in diabetes care, and often a maternal-fetal medicine provider. If clinicians do not have the expertise to manage insulin, providing appropriate resources and referrals is imperative.1 Telehealth provides an opportunity to extend resources that are not readily available.17,18
Oral hypoglycemics used in pregnancy include metformin and glyburide. Metformin, an oral biguanide, crosses the placenta. Data regarding long-term outcomes in offspring are limited but reassuring. Gastrointestinal side effects are common, but discontinuation rates are low. The sulfonylurea glyburide also crosses the placenta, but studies evaluating long-term effects in offspring are lacking. Monotherapy with oral agents effectively controls hyperglycemia in over half of pregnancies. A meta-analysis of studies comparing oral hypoglycemics and insulin suggests that glyburide is inferior to metformin, insulin, and metformin with insulin. In two Cochrane reviews, the authors concluded that there was not sufficient evidence to assess whether one oral hypoglycemic agent is superior to another or to insulin. They noted that the choice among these medications may reasonably be based on clinician and patient preference, availability, or the severity of GDM.22
Antenatal surveillance and birth planning
Patients with A1GDM, unlike patients with A2GDM, regardless of treatment regimen, are at low risk for complications and do not require antepartum surveillance.5,11 The type and frequency of surveillance utilized in the setting of A2GDM varies by community standards. Recommendations include serial growth ultrasounds every 4 weeks, typically from 28 weeks until term.10 Either weekly biophysical profiles or twice-weekly nonstress tests with weekly amniotic fluid index (AFI) are recommended after 32 weeks. Serial AFI measurements are incorporated to assess for polyhydramnios that can result from fetal hyperglycemia.5,10,11
Poorly controlled GDM is not clearly defined, but earlier birth timing is supported in patients with persistent hyperglycemia despite interventions. Timing is guided by the tradeoff between the risk of prematurity and the ongoing risks of stillbirth.5 Similar to low-risk pregnancies, timing of birth for patients with well-controlled A1GDM should not occur before 39 weeks and can be expectantly managed up to 40 6/7 weeks barring additional comorbidities. Initiating antenatal testing after 40 weeks in this population can be considered.11 For those with well-controlled A2GDM, birth between 39 0/7 and 39 6/7 weeks’ gestation is recommended. Gestational diabetes is not a contraindication to vaginal birth after cesarean. Mode of birth should be discussed when estimated fetal weight is greater than 4,500 g.5,10
Postpartum
Typically, carbohydrate intolerance resolves after giving birth and any pharmacologic treatment should be discontinued. Postpartum screening up to 12 weeks with a 75-g OGTT is recommended, as one in three will have persistent diabetes or impaired glucose tolerance.5,10,23 Diabetes, obesity, and long-term metabolic disorders are more prevalent in individuals who have had GDM.24 Breastfeeding may improve insulin resistance and glucose intolerance and lower the lifetime risk of developing diabetes.10,24
Despite the overwhelming demands associated with the postpartum period, it is the ideal time to promote patient self-efficacy over their future health and future pregnancies. Providers must stress to patients the importance of lifelong screening for type 2 diabetes. Patients need explicit information about their underlying risk and must be encouraged to prioritize follow-up with a primary care provider for continued diabetes prevention interventions and surveillance.4,9
Conclusion
The increasing prevalence of obesity predisposes more pregnant people to the development of diabetes in pregnancy. It is imperative for clinicians to understand the current screening and diagnostic guidelines as well as treatment modalities to support patients with GDM. An individualized, multidisciplinary approach that incorporates dietary and lifestyle changes and pharmacologic therapies if indicated will facilitate optimal care. Women’s health nurse practitioners can expertly support patients with GDM during and after pregnancy. They are well positioned to educate people about their higher lifetime risk of cardiometabolic disease while promoting risk reduction behaviors to optimize future health.
Genevieve M. Hofmann is Assistant Professor in the Department of Ob/Gyn at the University of Colorado School of Medicine in Denver, Colorado, and Dalisa Barquero is a women’s health nurse practitioner and clinical lead at Mountain Park Health Center in Phoenix, Arizona. The authors have no actual or potential conflicts of interest in relation to the contents of this article.
Womens Healthcare. 2023;11(4):22-27. doi: 10.51256/WHC082322
References
1 ElSayed NA, Aleppo G, Aroda VR, et al, on behalf of the American Diabetes Association. 2. Classification and diagnosis of diabetes: standards of care in diabetes–2023. Diabetes Care. 2023;46(suppl 1):S19-S40.
2 Gregory ECW, Ely DM. Trends and characteristics in gestational diabetes: United States, 2016–2020. Natl Vital Stat Rep. 2022;71(3):1-15.
3 Duarte-Gardea MO, Gonzales-Pacheco DM, Reader DM, et al. Academy of Nutrition and Dietetics gestational diabetes evidence-based nutrition practice guideline. J Acad Nutr Diet. 2018;118(9):1719-1742.
4 Arabin B, Baschat AA. Pregnancy: an underutilized window of opportunity to improve long-term maternal and infant health—an appeal for continuous family care and interdisciplinary communication. Front Pediatr. 2017;5:69.
5 American College of Obstetricians and Gynecologists. ACOG Practice Bulletin no. 190. Gestational diabetes mellitus. Obstet Gynecol, 2018;131(2):e49-e64.
6 McIntyre HD, Catalano P, Zhang C, et al. Gestational diabetes mellitus. Nat Rev Dis Primers. 2019;5(1):47.
7 Draffin C, Alderdice FA, McCance DR, et al. Exploring the needs, concerns and knowledge of women diagnosed with gestational diabetes: a qualitative study. Midwifery. 2016;40:141-147.
8 Gray MF, Hsu C, Kiel L, Dublin S. “It’s a very big burden on me”: women’s experiences using insulin for gestational diabetes. Matern Child Health J. 2017;21(8):1678-1685.
9 Parsons J, Ismail K, Amiel S, Forbes A. Perceptions among women with gestational diabetes. Qual Health Res. 2014;24(4):575-585.
10 Tsakiridis I, Giouleka S, Mamopoulos A, et al. Diagnosis and management of gestational diabetes mellitus: an overview of national and international guidelines. Obstet Gynecol Surv. 2021;76(6):367-381
11 Denney JM, Quinn KH. Gestational diabetes: underpinning principles, surveillance, and management. Obstet Gynecol Clin North Am. 2018;45(2):299-314.
12 Zhang M, Zhou Y, Zhong J, et al. Current guidelines on the management of gestational diabetes mellitus: a content analysis and appraisal. BMC Pregnancy Childbirth. 2019;19(1):200.
13 US Preventive Services Task Force. Final Recommendation Statement. Gestational Diabetes: Screening. August 10, 2021. https://www.uspreventiveservicestaskforce.org/uspstf/recommendation/gestational-diabetes-screening.
14 Olczuk D, Priefer R. A history of continuous glucose monitors (CGMs) in self-monitoring of diabetes mellitus. Diabetes Metab Syndr. 2018;12(2):181-187.
15 Igwesi-Chidobe CN, Okechi PC, Emmanuel GN, Ozumba BC. Community-based non-pharmacological interventions for pregnant women with gestational diabetes mellitus: a systematic review. BMC Womens Health, (2022);22(1):482.
16 Johns EC, Denison FC, Norman JE, Reynolds RM. Gestational diabetes mellitus: mechanisms, treatment, and complications. Trends EndocrinolMetab. 2018;29(11),743-754.
17 Aziz A, Zork N, Aubey JJ, et al. Telehealth for high-risk pregnancies in the setting of the COVID-19 pandemic. Am J Perinatol. 2020;37(8):800-808.
18 Zork NM. Telehealth for the management of diabetes in pregnancy. Curr Diab Rep. 2022;22(8):365-369.
19 Tassone C, Keshavjee K, Paglialonga A, et al. Evaluation of mobile apps for treatment of patients at risk of developing gestational diabetes. HealthInformatics J. 2020;26(3):1983-1994.
20 Liney T, Shah NM, Singh N. Recurrent gestational diabetes: breaking the transgenerational cycle with lifestyle modification. Wien klin Wochenschr. 2022;134(21-22):788-798.
21 Hod M, Di Renzo GC, Jovanovic LG, et al, eds. Textbook of Diabetes and Pregnancy, 3rd ed. London, UK: CRC Press; 2016. https://doi.org/10.1201/9781315382104.
22 Society of Maternal-Fetal Medicine (SMFM) Publications Committee. SMFM statement: pharmacological treatment of gestational diabetes.Am J Obstet Gynecol. 2018;218(5):B2-B4.
23 Paul JC, Fitzpatrick JJ. Postpartum glucose screening among women with gestational diabetes. Appl Nurs Res. 2020;56:151341.
24 Choudhury AA, Rajeswari VD. Gestational diabetes mellitus – a metabolic and reproductive disorder. Biomed Pharmacother. 2021;143:112183.
Key words: pregnancy, gestational diabetes, prenatal care, postpartum care