The pelvic floor is a broad group of muscles, primarily composed of the levator ani muscle group. These muscles maintain a constant low level of contraction to support the pelvic organs and tighten the urethral and anal sphincters to maintain continence. Pelvic organ prolapse (POP) occurs when the pelvic floor muscles relax or are damaged and fail to support various pelvic organs. As a result, one or more of the vaginal walls or cervix descend. Cystocele and/or urethrocele occur with anterior vaginal wall prolapse. Rectocele and/or enterocele occur with posterior vaginal wall prolapse. Uterine/cervical prolapse or vaginal vault prolapse after a hysterectomy may also occur.
POP affects approximately 25% of women in the United States.1 The prevalence of POP increases with age, obesity, chronic constipation, and in those with hysterectomies and greater parity.1–3 Spontaneous vaginal delivery and trauma of the levator ani muscles associated with operative vaginal delivery increases the risk of POP.1,4,5
Many women with POP are asymptomatic and do not need treatment. For those who are symptomatic, pessaries are a treatment option and can often provide relief without surgical intervention. Nurse practitioners (NPs) providing women’s healthcare have an important opportunity to evaluate for POP, fit pessaries, provide instructions on use and care, and follow up to promote successful use and prevent any complications. Here, we review the evaluation of POP and management for symptomatic women with a focus on pessaries.
Clinical evaluation
A thorough history and pelvic exam are often sufficient to diagnose POP and pave the way for an appropriate treatment plan. When symptomatic, women commonly report sensations of vaginal pressure and seeing or feeling a vaginal bulge.1 Other symptoms such as painful sexual intercourse, urinary incontinence, sensation of incomplete bladder emptying, difficulty with defecating, fecal incontinence, or vaginal bleeding need careful evaluation as they may or may not be attributable to the prolapse and can be due to independent urinary, gynecologic, or gastrointestinal conditions. The NP should also ask the patient how symptoms are affecting her quality of life, sexual health, and ability to go about her usual daily routine. This information can be helpful in establishing the individual patient’s priorities and goals for treatment.
The physical examination should include assessment of the abdomen, external and internal genitalia, perianal area, and pelvic floor muscle tone. During the pelvic exam in semi-Fowler lithotomy position, the NP can assess muscle strength with vaginal palpation while the patient contracts her pelvic floor muscles. To assess for visible descent of vaginal walls, the patient can bear down in a Valsalva maneuver. If prolapse is not evident in supine position, then the patient can stand and valsalva while the NP assesses for visible or palpable prolapse. The NP should assess the integrity of the vaginal epithelium with attention to any atrophy, erosions, or ulcerations. It is beneficial to use a split speculum to evaluate each aspect of the vaginal wall and determine which compartments — anterior, posterior, apical — are involved.
Two systems commonly used to quantify the severity of prolapse are the Baden-Walker halfway scoring system and pelvic organ prolapse (POP) quantification system. Both methods assess the descent of the three pelvic compartments: anterior, posterior, and apical. The Baden-Walker halfway scoring system describes the degree of prolapse findings in relation to the hymen (Table).
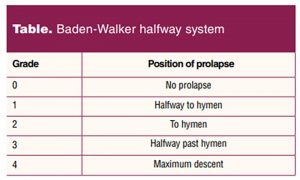
The POP quantification system is a more detailed assessment of each pelvic compartment in relation to the hymen.6 Quantification of the extent of prolapse, the severity of symptoms, and the impact on the patient’s quality of life are essential in making treatment decisions.
Pessaries for the treatment of symptomatic pelvic organ prolapse
It has been reported that women with symptomatic and bothersome pelvic organ prolapse are often satisfied with a pessary for management.7,8 Pessaries support the pelvic organs so they are in the proper anatomic position to relieve symptoms. They are available in a variety of shapes and sizes. The NP will want to become familiar with and have available more than one pessary type to allow for individual patient needs.
A ring pessary is a popular first-line choice. This style of pessary is available with or without a central support diaphragm and can be used for any type of prolapse. The ring pessary is flexible and can usually be easily removed and reinserted by the patient, an important characteristic if the patient wishes to manage the pessary and is sexually active. If the pessary is difficult to remove, threading a strand of plain, waxed dental floss through the ring can allow the patient to apply gentle traction to facilitate removal.
Although 90% of patients can be successfully fitted with a pessary, there are some vaginal configurations that make it more difficult to retain the pessary such as shortened vaginal length, wide introitus diameter, and more extensive degrees of prolapse.9 In these scenarios, a space-occupying pessary such as a Gellhorn or donut may be better retained and provide the most relief (Figure).

A Gellhorn pessary has a wide base that creates suction to the vaginal wall that must be broken prior to its removal. The donut pessary can be inflated and deflated for improved retention and removal, simply with a needle and syringe, and is especially effective for advanced prolapse and a wide introitus. These pessaries are rigid, giving more support. It is uncommon for the patient to be able to routinely remove and reinsert these on her own. The size of space-occupying pessaries and difficulty in removing can preclude sexual intercourse. Regular office visits are required for removal, cleaning, evaluation of the vagina, and reinsertion.
Fitting a pessary
Most pessaries come with printed instructions regarding fitting, insertion, removal, and care of the specific device. Pessary fitting kits are available to be able to evaluate more than one size for the best fit. The fitting should start with measuring the vaginal width with two fingers in the vagina (much like an intrapartum cervical exam). Then, the vaginal length can be determined by palpating the posterior fornix (or vaginal vault if post-hysterectomy) with the index or middle finger estimating the distance to the pubic symphysis. The combination of these two measurements can give the NP an idea for what type and size of pessary to trial first. A water-based lubricant should be applied to the leading edge of the pessary to make insertion easier. This is followed by separating the labia and inserting the pessary at the introitus and, with gentle pressure, sliding it up the posterior vaginal wall. The pessary can then be eased into position in the upper part of the vagina and behind the pubic symphysis.
A pessary should comfortably fit without undue tension. The NP should be able to easily insert one digit between the pessary and vaginal wall. Once a pessary is inserted, assess if it remains in place with the Valsalva maneuver and when voiding. If the pessary is not expelled, and the patient is able to void without difficulty, this is a good indicator that there is an appropriate fit.
Moisturizers and local vaginal estrogen
Moisturizers can be applied intravaginally frequently as needed to reduce irritation from the pessary. Over-the-counter options include commercial vaginal moisturizers, coconut oil, vitamin E oil, and aloe products.
Women with vaginal atrophy as a result of decreased estrogen levels can benefit from local vaginal estrogen to bolster, moisturize, and normalize the microbiome of the vaginal epithelium. If vaginal atrophy is especially symptomatic, intiating vaginal estrogen cream prior to pessary fitting can reduce discomfort. Regular use leads to more robust tissue and reduction of irritation from pessary placement. Because systemic estrogen concentrations are negligible with twice-weekly application of vaginal estrogen, there is no need to prescribe progesterone in women with a uterus.8
Care of the pessary
Instructions for the care and use of the pessary can be individualized according to the patient’s level of comfort with self-management and the health of her vaginal epithelium. Use of moisturizers or local vaginal estrogen reduce irritation, which decreases discharge and odor. Douching can alter the vaginal microbiome and increase risk of infections, so it is discouraged.10
For the patient who will self-manage, the pessary may be removed at least once weekly for cleaning with water and gentle soap. If symptoms are episodically bothersome, such as only during exercise, then the pessary may be removed as needed. With the sexually active patient, it is recommended to remove the pessary for penetrative intercourse for both their comfort and their partner’s. For self-managed pessaries without issues, patients may present for surveillance examinations at 12-month intervals.
If the pessary is not self-managed, the patient should return every 3 to 6 months for pessary removal, cleaning, examination of vaginal tissue health, and reinsertion of pessary.6 Using the diagnosis code of vaginal pessary in situ (Z96.0) alerts the NP to monitor the status of the vagina and pessary whenever the patient is seen for care. This can be especially important for patients with cognitive impairment.
Possible complications
Pessary complications are rare but include vaginal ulcerations, abrasions, bleeding, vaginal discharge and odor, urinary retention, and occult stress urinary incontinence.3,7 Serious and rare complications include fistula development and pessary migration, which are often due to long-term pessary neglect.6 If a woman develops evidence of a vaginal erosion, a “pessary holiday” (leaving the pessary out for 2–4 weeks) is advisable to allow healing while reinforcing vaginal estrogen use during this time.3
Other treatments for POP
Women with POP may also benefit from pelvic floor muscle strengthening exercises and pelvic floor physical therapy. If conservative therapy does not achieve adequate symptom control or the patient does not tolerate or declines conservative therapy, then a surgical referral to a urogynecologist is indicated. Other indications for referral include recurrent vaginal ulcerations from pessaries, obstruction of the urinary tract, or development of new urinary incontinence.
Conclusion
Pessaries are a safe and potentially long-term intervention for women with symptomatic pelvic organ prolapse. Severity of prolapse, vaginal atrophy, and preservation of sexual function influence pessary shape and frequency of follow-up visits. With the aging of the population, the number of women with POP will likely increase. NP knowledge about pessary use provides patients with an effective nonsurgical option for POP treatment.
Cassandra Shaw is a women’s health nurse practitioner (WHNP) in the Urogynecology Division of the Department of Obstetrics & Gynecology at Duke University in Durham, North Carolina. Michele O’Shea is a fellow in Female Pelvic Medicine & Reconstructive Surgery in the Department of Obstetrics & Gynecology at Duke University. Kathryn Trotter is an Associate Professor at Duke University School of Nursing and the lead faculty for the WHNP program. The authors have no actual or potential conflicts of interest in relation to the contents of this article.
Resources:
For the patient:
voicesforpfd.org/resources/fact-sheets-and-downloads/
yourpelvicfloor.org/leaflets/
voicesforpfd.org/assets/2/6/Vaginal_Pessaries.pdf
For healthcare providers:
The AUGs site specifically for healthcare providers is augs.org/patient-services/healthcare-providers/
For both patients and providers:
voicesforpfd.org
References
- Blomquist JL, Muñoz A, Carroll M, Handa VL. Association of delivery mode with pelvic floor disorders after childbirth. JAMA. 2018;320(23):2438-2447.
- Lukacz ES, Santiago-Lastra Y, Albo ME, Brubaker L. Urinary incontinence in women: a review. JAMA. 2017;318(16):1592-1604.
- Pelvic organ prolapse: ACOG Practice Bulletin Summary, Number 214. Obstet Gynecol. 2019;134(5):1124-1127.
- Milsom I, Gyhagen M. Breaking news in the prediction of pelvic floor disorders. Best Pract Res Clin Obstet Gynaecol. 2019;54:41-48.
- Tähtinen RM, Cartwright R, Vernooij RWM, et al. Long-term risks of stress and urgency urinary incontinence after different vaginal delivery modes. Am J Obstet Gynecol. 2019;220(2):181.e1-181.e8.
- Propst K, Mellen C, O’Sullivan DM, Tulikangas PK. Timing of office-based pessary care: a randomized controlled trial. Obstet Gynecol. 2020;135(1):100-105.
- Coolen AWM, Troost S, Mol BWJ, et al. Primary treatment of pelvic organ prolapse: pessary use versus prolapse surgery. Int Urogynecol J. 2018;29(1):99-107.
- Naumova I, Castelo-Branco C. Current treatment options for postmenopausal vaginal atrophy. Int J Womens Health. 2018;10:387-395.
- Mao M, Ai F, Zhang Y, et al. Predictors for unsuccessful pessary fitting in women with symptomatic pelvic organ prolapse: a prospective study. BJOG. 2018;125(11):1434-1440.
- Vaginitis in nonpregnant patients: ACOG Practice Bulletin, Number 215. Obstet Gynecol. 2020;135(1):e1-e17.
- Lamers BHC, Broekman BMW, Milani AL. Pessary treatment for pelvic organ prolapse and health-related quality of life: a review. Int Urogynecol J. 2011;22(6):637-644.

