Faculty: Nancy R. Berman, MSN, ANP-BC, NCMP, FAANP, is an adult nurse practitioner and certified menopause practitioner at Millennium Affiliated Physicians, Division of Michigan Healthcare Professionals, in Farmington Hills, Michigan.
Intended audience: This continuing education (CE) activity has been designed to meet the educational needs of nurse practitioners and other healthcare providers who provide primary care for women.
CE approval period: Now through June 30, 2023
Estimated time to complete this activity: 1 hour
CE approval hours: 1.0 contact hour of CE credit including 0.50 contact hours of pharmacology content
Goal statement: Nurse practitioners and other healthcare providers who provide primary care for women will increase their knowledge about screening and assessment for fracture risk and diagnosis and treatment for postmenopausal osteoporosis.
Needs assessment: Fragility fractures related to bone loss occur most often in older postmenopausal women and cause significant morbidity and mortality. Screening to assess fracture risk is underutilized, and effective pharmacologic therapy is under prescribed. Knowledge about screening and risk assessment recommendations, how to interpret findings, and how to individualize pharmacologic treatment for postmenopausal osteoporosis is needed to help older women avoid fractures that significantly affect quality of life.
Educational objectives: At the conclusion of this educational activity, participants should be able to:
- Describe recommended screening and fracture risk assessment for postmenopausal women.
- Identify the criteria for the diagnosis of osteoporosis in postmenopausal women.
- Discuss mechanism of action, factors that influence specific drug choice, patient education on use, adverse effects, and contraindications for pharmacologic options in treating postmenopausal osteoporosis.
Accreditation statement: This activity has been evaluated and approved by the Continuing Education Approval Program of the National Association of Nurse Practitioners in Women’s Health (NPWH) and has been approved for 1 contact hour CE credit, including 0.50 hours of pharmacology credit.
Faculty disclosures: NPWH policy requires all faculty to disclose any affiliation or relationship with a commercial interest that may cause potential, real, or apparent conflict of interest with the content of a CE program. NPWH does not imply that affiliation or relationship will affect the content of the CE program. Disclosure provides participants with information that may be important to their evaluation of an activity.
Nancy R. Berman, MSN, ANP-BC, NCMP, FAANP, has no actual or potential conflicts of interest in relation to the contents of this article.
Disclosure of unlabeled/unapproved use: NPWH policy requires authors to disclose to participants when they are presenting information about unlabeled use of a commercial product or device or an investigational use of a drug or device not yet approved for any use.
Disclaimer: Participating faculty members determine the editorial content of the CE activity; this content does not necessarily represent the views of NPWH. This content has undergone a blinded peer review process for validation of clinical content. Although every effort has been made to ensure that the information is accurate, clinicians are responsible for evaluating this information in relation to generally accepted standards in their own communities and integrating the information in this activity with that of established recommendations of other authorities, national guidelines, FDA-approved package inserts, and individual patient characteristics.
Successful completion of the activity: Successful completion of this activity, J-21-03, requires participants to do the following:
- “Sign In” at the top right-hand corner of this page if you have an NPWH account. You must be signed in to receive credit for this course. If you do not remember your username or password, please follow the “Forgot Password” link and instructions on the sign in page. If you do not have an account, please click on “Create an Account.”*
- Read the learning objectives, disclosures, and disclaimers on the next page and then click on the “Continue” button.
- Study the material in the learning activity during the approval period (now through June 30, 2023).
- Complete the post-test and evaluation. You must earn a score of 70% or higher on the post-test to receive CE credit.
- Print out the CE certificate after you have successfully passed the post-test and completed the evaluation.
*If you are an NPWH member, were once a member, or have completed CE activities with NPWH in the past, you have a username and password in our system. Please do not create a new account. Creation of multiple accounts could result in loss of CE credits as well as other NPWH services. If you do not remember your username or password, either click on the “Forgot Username” or “Forgot Password” link or call the NPWH office at (202) 543-9693, ext. 1.
Commercial support: This activity did not receive any commercial support.
In older postmenopausal women, fragility fractures related to bone loss occur most often and can result in significant morbidity and mortality. This article examines current recommendations for screening and risk assessment, diagnosis, and treatment of postmenopausal osteoporosis, the latter including nonpharmacologic therapy and FDA-approved pharmacologic therapy. Factors to consider in individualizing treatment and the importance of patient education are also discussed.
Key words: postmenopausal osteoporosis, fracture, T score
Fractures related to bone loss cause significant morbidity and mortality. Many of these fragility fractures that occur most often in older postmenopausal women can be prevented. Screening to determine an individual’s risk for fracture is underutilized, and effective pharmacologic therapy is underprescribed.1 Even when patients are diagnosed with increased fracture risk, many choose not to take medication because of concerns about rare risks that are often sensationalized in the media.2 The goal of screening, diagnosis, and treatment is to prevent fractures and particularly the first fracture, because one fracture significantly increases the risk of a subsequent fracture. The purpose of this article is to describe screening and risk assessment recommendations, interpretation of findings, and treatment of postmenopausal osteoporosis. Patient education and factors to consider in individualizing treatment are discussed.
Screening for and assessment of fracture risk
All women age 65 years and older should be screened for bone mineral density (BMD) with dual-energy x-ray absorptiometry (DXA). Postmenopausal women should have BMD testing earlier if they have clinical risk factors for fracture including use of medications associated with bone loss (eg, systemic glucocorticoid therapy > 3 months, aromatase inhibitors), low body mass index, history of a fragility or low-trauma fracture, history of hip fracture in a parent, low calcium intake, current smoking, or excess alcohol or caffeine consumption. Other risk factors for fracture are medical conditions known to cause bone loss including chronic renal disease, estrogen deficiency (early menopause, anorexia), hyperparathyroidism, systemic lupus erythematosus, rheumatoid arthritis, chronic obstructive pulmonary disorders, and conditions associated with malabsorption such as celiac disease and inflammatory bowel disease.3–6 BMD testing is not appropriate in premenopausal women and women younger than age 65 unless there are risk factors such as a significant history of fracture, early menopause, or specific risk factors for bone loss, such as long-term glucocorticoid therapy.4
The gold standard for measuring BMD is DXA of the total hip, femoral neck, and lumbar vertebrae. If the lumbar spine or hips cannot be evaluated due to hardware or arthroplasty, the distal one-third of the forearm may be used. The T score represents the number of standard deviation (SDs) from young adult mean values at each site (Figure 1).5 It is useful to perform subsequent BMD tests on the same device for the most accurate assessment of stability or loss. Stable or increasing BMD with no evidence of new fractures is considered a response to therapy.4 Most insurance covers BMD after 2 years of therapy, although a 1-year follow-up may be considered to assess treatment efficacy. The most useful measurement for assessing response to pharmacologic therapy is the total hip T score.4
Diagnosing osteoporosis based on BMD test alone will miss a significant number of patients at high fracture risk who are not yet osteoporotic by T score. Multiple fracture risk assessment tools are available, but the most commonly used is FRAX.7 FRAX provides a calculation of 10-year fracture risk based on demographics including race, country of origin, age, height, weight, parental hip fracture, adult fracture, current smoking, rheumatoid arthritis, glucocorticoid therapy, and the BMD at the femoral neck, if available. FRAX is appropriate for fracture risk calculation in postmenopausal women age 40 years and older. The FRAX calculator is available online at the University of Sheffield.B FRAX does not include spine data and thus may underestimate an individual’s risk (Figure 2).7–10
Diagnosing osteoporosis
On the bone density test, the diagnosis of osteoporosis is made when a patient has a T score of -2.5 or lower at the lumbar spine (L1–L4), total hip, femoral neck, or distal one-third of the forearm. A FRAX score is not needed for further confirmation. If a patient has a T score of -1.0 to -2.5 at any site, it is important to calculate the FRAX score. A T score between -1.0 and -2.5 and a FRAX score indicating a 10-year risk of ≥ 20% for major osteoporotic fracture or ≥ 3% risk of hip fracture meets the criteria for a diagnosis of osteoporosis.4 The Box lists four accepted criteria for the diagnosis of osteoporosis in postmenopausal women. Pharmacologic therapy should be considered for patients who meet any of these criteria.
Box. Criteria for diagnosis of osteoporosis in postmenopausal women
- T score -2.5 or below in the lumbar spine, femoral neck, total proximal femur, or distal 1/3 radius
- Low-trauma spine or hip fracture (regardless of bone mineral density)
- T score between -1.0 and -2.5 and fragility fracture of proximal humerus, pelvis, or distal forearm
- T score between -1.0 and -2.5 and high FRAX (or if available, TBS-adjusted probability based on country-specific thresholds) defined as: ≥ 20% for major osteoporotic fracture or ≥ 3% for hip fracture
Initial work-up for patients with a diagnosis of osteoporosis includes health history, physical examination, and laboratory tests to assess the severity of osteoporosis, identify any secondary causes, and determine if there are any contraindications to specific therapies. Routine laboratory tests include a complete blood count, comprehensive metabolic panel (include calcium, creatinine, alkaline phosphatase, albumin, and phosphate), 25-hydroxyvitamin D, and 24-hour urine collection for calcium, sodium, and creatinine. Other laboratory tests may be considered based on patient history, physical examination, and routine laboratory test results including an intact serum parathyroid hormone concentration and thyroid-stimulating hormone.
A lateral thoracic and lumbar spine radiograph should be considered if the patient has an historical height loss of > 4 cm (> 1.5 inches). Two thirds of vertebral fractures are asymptomatic yet are a significant risk factor for future fracture and may impact choice of pharmacologic agent.4,5
Treatment
After a diagnosis of osteoporosis, both nonpharmacologic and pharmacologic therapies should be discussed with the patient. Patients may be reluctant to start pharmacologic therapy and will often ask if they can just increase their intake of calcium. Patient education involves a discussion of the role of nonpharmacologic therapy but that it is inadequate by itself to decrease fracture risk in the osteoporotic patient.
Nonpharmacologic therapy
Nonpharmacologic therapy includes adequate calcium and vitamin D intake. Vitamin D status is assessed by measurement of serum 25(OH)D. The recommended level by the Endocrine Society is ≥ 30 ng/mL. There is controversy regarding the upper limit for serum 25(OH)D, although a reasonable upper limit is 50 ng/mL. The society also recommends daily dosing of up to 5,000 IU vitamin D3 daily for 8 to 12 weeks in vitamin D deficiency and notes that only in uncommon clinical situations is there a need to prescribe the high dose of 50,000 IU weekly. It is hypothesized that daily dosing more closely replicates the physiology of its metabolism in the skin. Clinical factors such as obesity and a history of malabsorption may require increasing the dose necessary to achieve adequate levels.4
Many published reports recommend an intake of at least 1,000 IU vitamin D daily for adults age 50 years and older and 4,000 IU as a safe upper limit.11 The need for and amount of calcium supplementation should be based on a patient’s dietary history. The current daily calcium recommendation is for 1,200 mg total between diet and supplement if indicated. The most common calcium supplement is carbonate, which is easy to obtain but may cause gastrointestinal (GI) distress with gas and constipation. The most easily absorbed calcium is citrate, and the best tolerated is phosphate. Patients should be encouraged to have regular weight-bearing exercise such as walking for 30 to 40 minutes a day and strength and balance training. Fall prevention strategies should be implemented.12–14 Smoking cessation and alcohol intake reduction should be advised.
FDA-approved pharmacologic therapy
The most frequently used drugs for treatment of postmenopausal osteoporosis are the bisphosphonates with oral and intravenous (IV) infusion administration options. This class of drugs inhibits osteoclastic bone resorption by attaching to hydroxyapatite binding sites on bony surfaces, especially those which are undergoing active resorption.4 These are well-studied agents, have a long-term safety record, and are effective in decreasing the risk of vertebral, nonvertebral, and hip fractures. The exception is ibandronate, with data to support its ability to decrease the risk of vertebral but not nonvertebral or hip fracture.4,5 The most common oral bisphosphonate is alendronate, which is taken once a week with a full glass of water and a 30-minute waiting period before eating, drinking, or laying down. This strategy decreases the risk of esophageal irritation or ulceration, and not laying down encourages passage into the stomach. Drinking coffee or juice before 30 minutes can reduce absorption by as much as 60%.4
For the patient who experiences acid reflux on oral bisphosphonates or who has gastroesophageal reflux disorder (GERD), once yearly IV infusion of the bisphosphonate, zoledronic acid, can be considered. Many insurance companies require preauthorization with documentation that the patient has tried and failed oral therapy, but a case can be made for anticipated intolerance or noncompliance. Some patients may do well initially but, with cognitive decline, may lose the ability to follow the special directions for taking the drug correctly.15 IV infusion of zoledronic acid should take approximately 15 minutes. The patient should be well hydrated with 2 glasses of water on the morning of the infusion. Patients should take prophylactic ibuprofen or acetaminophen before the infusion to avoid risk of flulike symptoms, although if these occur, they are usually transient over a few days. IV infusion of zoledronic acid provides greater bioavailability than the oral agents as it bypasses the GI tract and has yearlong efficacy due to its high-binding affinity to bone.4,15,16
Bisphosphonates should not be used in patients with significantly decreased renal function (glomerular filtration rate < 35 mL/min) or hypocalcemia. Rare risks of bisphosphonates include osteonecrosis of the jaw (ONJ) and atypical subtrochanteric femur fractures that may occur more often with long-term use. It is best to avoid initiating bisphosphonates in patients having tooth extractions, or tooth implants until the jawbone has healed. There is no evidence that holding the drug for those procedures in patients already on bisphosphonates will decrease the risk, due to the drug’s long half-life.17 Drug holidays from bisphosphonates of 1 to 2 years may be considered after 5 years for patients on oral therapy and after 3 years of IV therapy to reduce the risks for ONJ and atypical femur fracture. The decision to initiate a drug holiday should be based on patient’s current fracture risk as determined by BMD.18–25
The rank ligand inhibitor denosumab is a fully human monoclonal antibody that works through a different pathway than the bisphosphonates. It specifically targets a ligand called RANKL that binds to a receptor called RANK, a key mediator of osteoclast formation, function, and survival.26 Denosumab reduces how many bone-removing cells are activated, improving cortical and trabecular bone density, volume, and strength.
Indications for denosumab include treatment of postmenopausal women with osteoporosis and high risk of fracture, treatment of glucocorticoid-induced osteoporosis, and treatment of postmenopausal women who are receiving adjuvant aromatase inhibitor therapy for breast cancer due to decreasing estrogen production and the subsequent loss of estrogen’s bone protection. Denosumab is given twice yearly by subcutaneous injection administered by a healthcare provider. It should never be discontinued without starting an alternate antiresorptive medication.There is a rapid decrease in BMD and increased risk of vertebral fractures within a few months after stopping this medication.27,28 Patients should be advised that if they are unable to get their denosumab injection within 4 to 5 weeks of their every 6 months schedule they should take a bisphosphonate until they can resume the denosumab. There is safety data for 10 years of continuous denosumab therapy, and ongoing trials will provide additional data over time.29
It is important to evaluate calcium and vitamin D levels before initiating treatment with denosumab as it may cause hypocalcemia with a greater risk in patients with significantly impaired renal function. Any hypocalcemia should be corrected prior to starting treatment. All patients on denosumab should be taking adequate calcium and vitamin D. Denosumab is not contraindicated in patients with renal insufficiency, and no dose adjustment is required.4 ONJ is a rare risk for patients on denosumab.
The estrogen agonist/antagonist raloxifene is FDA approved for the prevention and treatment of postmenopausal osteoporosis and for the prevention of invasive breast cancer in high-risk women.30 It reduces the risk of vertebral fractures but there are no data on nonvertebral or hip fracture. This drug is a reasonable choice for younger postmenopausal women who want prevention of both vertebral fractures and breast cancer. The drug exerts estrogen-like effects on the skeleton but is a weak antiresorptive agent. A small percentage of women may have hot flashes or leg cramps on the medication.4 There is a small increased risk for thromboembolic events, and the drug should be held for 1 to 2 weeks before periods of immobilization. If a woman on raloxifene loses bone mass at the hip over time and her T score reaches -2.5 or lower, she needs to start a more robust antiresorptive therapy. If she prefers to continue to have the benefit of breast cancer prevention, she may add the antiresorptive and stay on the raloxifene.
Anabolic agents are unique from other treatments because they are bone building through increased osteoblast activity. The FDA-approved anabolic agents are teriparatide and abaloparatide, both analogs of recombinant human parathyroid hormone. Both agents require a daily subcutaneous injection from a preloaded pen. Use is not recommended beyond 18 to 24 months’ duration. Because their effects diminish rapidly after discontinuing therapy, the course of treatment must be followed by maintenance with a bisphosphonate or a rank ligand inhibitor.4
The single most important reason for choosing an anabolic agent is a history of previous hip or spine fracture.31 Use of anabolic agents may also be indicated for patients with multiple fractures, very low BMD (T score below -3.0), failure of other medications, or other factors such as advanced age and increased fall risk.32 If an anabolic therapy is appropriate, some data suggest that the sequence of treatment is important and anabolic therapy first, followed by an antiresorptive agent, provides the best fracture prevention.4,33
Anabolic agents carry a boxed warning because of the occurrence of osteosarcomas in rats treated with very high doses. These agents should not be used in patients at increased risk of osteosarcoma and not in patients with untreated or unresolved hyperparathyroidism.4,32–35
Romosozumab is the newest agent for management of osteoporosis. It is a humanized monoclonal antibody that binds and inhibits sclerostin, a regulatory factor in bone metabolism. Romosozumab has a dual effect that increases bone formation and decreases bone resorption to a lesser extent. Indications for use of romosozumab are the same as those for the anabolic agents. It is administered as two separate subcutaneous injections once a month. The course of therapy is limited to 12 months and should be followed with another osteoporosis drug for maintenance. In studies that led to FDA approval, there were cardiovascular events that led to a recommendation to not start the drug within the first year of a myocardial infarction or stroke. Romosozumab has the same precautions regarding hypocalcemia, ONJ, and atypical femur fracture as the other osteoporosis drugs.36,37
Individualizing pharmacologic therapy
It is important to approach osteoporosis as a disease state that requires ongoing evaluation and continued treatment. Once a patient is diagnosed with osteoporosis the diagnosis remains even if their T score improves to better than -2.5.4 Many factors impact the decision for initial drug choice and should include the patient’s age, severity of bone loss and fracture risk, history of GERD or trouble swallowing, renal function, mental competence to follow a specific direction for taking the oral medication, and insurance coverage. It is also important to assess the newly diagnosed patient for secondary causes of bone loss and for prior vertebral or hip fracture as this may change the choice of drug.30
Patients with osteoporosis can be categorized as very high risk with a recent fracture (within the last 12 months), fractures while on approved drug therapy, multiple fractures while on drugs causing skeletal harm such as glucocorticoids, very low T score (< -3.0), very high fall risk and very high FRAX score: more than 30% for major osteoporotic fracture and 4.5% for hip fracture.31 High-risk patients with a diagnosis of osteoporosis include postmenopausal women over age 50 with a prior hip or spine fracture, T score of -2.5 or lower at the hip or spine, and postmenopausal women with a T score between -1 and -2.5 at the femoral neck, total hip, or spine if: the 10-year risk by FRAX is 20% or greater for major osteoporotic fracture or 3% or greater for hip fracture.4,38 The Table provides examples of initial treatment choices based on these risk categories.
Table. Initiating pharmacologic therapy: Case examples4,31
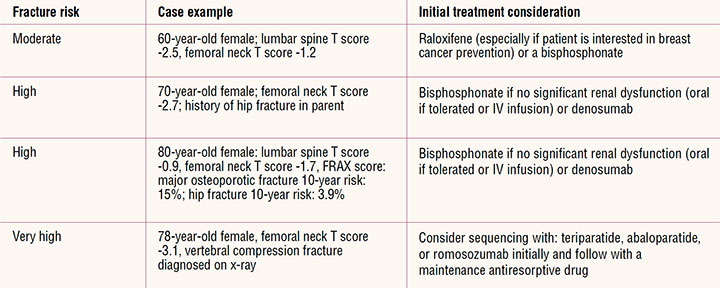
The most common initial therapy is an oral bisphosphonate. When the patient is not able to take the oral bisphosphonate, it is reasonable to recommend IV infusion.4 Denosumab is also considered an appropriate initial drug for some patients and is best covered by Medicare but once started should not be stopped without continuing with another therapy. Patients at very high risk may be candidates for abaloparatide, denosumab, romosozumab, teriparatide, and zoledronic acid.
Long-term follow-up
Follow-up of patients with osteoporosis must be ongoing so that decisions can be made regarding continuation of therapy or change in therapeutic agent. A DXA may be repeated every 1 to 2 years until findings are stable. Stable or increasing bone density and no evidence of new fractures or vertebral fracture progression is a sign of successful therapy. One new fracture may not necessarily be evidence of treatment failure, but two or more fragility fractures is.4,19,24,38
With an oral bisphosphonate consider a drug holiday after 5 years and after 3 years with IV infusion if fracture risk is no longer high (T score better than -2.5 and no fractures). Continue oral therapy up to 10 years and IV infusion therapy for 6 years if fracture risk remains high. Drug holidays should not be used for non-bisphosphonate antiresorptive drugs. If denosumab is discontinued, the patient should be transitioned to another antiresorptive.20 The treatment course of anabolic agents and the antisclerostin agent are followed with a drug intended for long-term use.
Individualized patient counseling to include explanations about BMD T scores, FRAX scores, and treatment options is needed for effective management of osteoporosis. Patients may not adhere to treatment regimens if they do not understand the association between low bone mass and increased fracture risk when they do not have symptoms. Patients may have strong aversion to taking medication for osteoporosis due to things they have read, heard in the media, or presented by well-meaning family or friends. The risk/benefit discussion is important and requires addressing the morbidity and mortality related to fractures versus the rare complications of osteonecrosis of the jaw and atypical subtrochanteric femur fractures. Shared decision making regarding nonpharmacologic and pharmacologic treatment should be based on individual patient needs and desires. Instructions on the use of medications are important to prevent and/or manage side effects and potential adverse reactions. Adherence to medication regimens should be evaluated at routine healthcare visits. Patients should know that not all fractures can be prevented with pharmacologic therapy and that T scores may not always improve even when the drug is effective.
The treatment of osteoporosis has continued to evolve as new evidence comes forward. Clinicians have opportunities to reduce gaps in care for postmenopausal women by engaging in evidence-based screening, risk assessment, and treatment to reduce fracture risk. Clinicians should be able to recognize patients meeting the criteria for very high risk for fracture and make appropriate referrals to specialists for further assessment and initial management. The clinician can also reduce gaps in care by recognizing secondary causes of bone loss that increase fracture risk and providing or referring for treatment. Ongoing dialogue between patient and clinician is critical for effective fracture prevention and a subsequent reduction in morbidity and mortality.
References
Miller PD. Underdiagnosis and undertreatment of osteoporosis: the battle to be won. J Clin Endocrinol Metab. 2016;101(3):852-859.
Cipriani C, Pepe J, Minisola S, Lewiecki EM. Adverse effects of media reports on the treatment of osteoporosis. J Endocrinol Invest. 2018;41(12):1359-1364.
Management of osteoporosis in postmenopausal women: 2010 position statement of The North American Menopause Society. Menopause (New York, NY). 2010;17(1):25-54; quiz 55-26.
Camacho PM, Petak SM, Binkley N, et al. American Association of Clinical Endocrinologists/American College of Endocrinology clinical practice guidelines for the diagnosis and treatment of postmenopausal osteoporosis–2020 update executive summary. Endocr Pract. 2020;26(5):564-570.
Cosman F, de Beur SJ, LeBoff MS, et al. Clinician’s guide to prevention and treatment of osteoporosis. Osteoporos Int. 2014;25(10):2359-2381.
Curry SJ, Krist AH, Owens DK, et al. Screening for osteoporosis to prevent fractures: US Preventive Services Task Force recommendation statement. JAMA. 2018;319(24):2521-2531.
Kanis JA, Johansson H, Harvey NC, McCloskey EV. A brief history of FRAX. Arch Osteoporos. 2018;13(1):118.
Edwards BJ. Osteoporosis risk calculators. J Clin Densitom. 2017;20(3):379-388.
Kanis JA, Harvey NC, Johansson H, et al. FRAX update. J Clin Densitom. 2017;20(3):360-367.
Watts NB, Ettinger B, LeBoff MS. FRAX facts. J Bone Miner Res. 2009;24(6):975-979.
Kahwati LC, Weber RP, Pan H, et al. Vitamin D, calcium, or combined supplementation for the primary prevention of fractures in community-dwelling adults: evidence report and systematic review for the US Preventive Services Task Force. JAMA. 2018;319(15):1600-1612.
Ross AC, Manson JE, Abrams SA, et al. The 2011 report on dietary reference intakes for calcium and vitamin D from the Institute of Medicine: what clinicians need to know. J Clin Endocrinol Metab. 2011;96(1):53-58.
Weaver CM, Alexander DD, Boushey CJ, et al. Calcium plus vitamin D supplementation and risk of fractures: an updated meta-analysis from the National Osteoporosis Foundation. Osteoporos Int. 2016;27(1):367-376.
Grossman DC, Curry SJ, Owens DK, et al. Interventions to prevent falls in community-dwelling older adults: US Preventive Services Task Force recommendation statement. JAMA. 2018;319(16):1696-1704.
Black DM, Delmas PD, Eastell R, et al. Once-yearly zoledronic acid for treatment of postmenopausal osteoporosis. N Engl J Med. 2007;356(18):1809-1822.
Eastell R, Lang T, Boonen S, et al. Effect of once-yearly zoledronic acid on the spine and hip as measured by quantitative computed tomography: results of the HORIZON Pivotal Fracture trial. Osteoporos Int. 2010;21(7):1277-1285.
Abed HH, Al-Sahafi EN. The role of dental care providers in the management of patients prescribed bisphosphonates: brief clinical guidance. Gen Dent. 2018;66(3):18-24.
Adams AL, Adams JL, Raebel MA, et al. Bisphosphonate drug holiday and fracture risk: a population-based cohort study. J Bone Miner Res. 2018;33(7):1252-1259.
Adler RA, El-Hajj Fuleihan G, Bauer DC, et al. Managing osteoporosis in patients on long-term bisphosphonate treatment: report of a task force of the American Society for Bone and Mineral Research. J Bone Miner Res. 2016;31(1):16-35.
Anagnostis P, Paschou SA, Mintziori G, et al. Drug holidays from bisphosphonates and denosumab in postmenopausal osteoporosis: EMAS position statement. Maturitas. 2017;101:23-30.
Bindon B, Adams W, Balasubramanian N, et al. Osteoporotic fractures during bisphosphonate drug holiday. Endocr Pract. 2018;24(2):163-169.
Kuroshima S, Sasaki M, Sawase T. Medication-related osteonecrosis of the jaw: a literature review. J Oral Biosci. 2019;61(2):99-104.
Ro C, Cooper O. Bisphosphonate drug holiday: choosing appropriate candidates. Curr Osteoporos Rep. 2013;11(1):45-51.
Watts NB, Diab DL. Long-term use of bisphosphonates in osteoporosis. J Clin Endocrinol Metab. 2010;95(4):1555-1565.
Ottesen C, Schiodt M, Gotfredsen K. Efficacy of a high-dose antiresorptive drug holiday to reduce the risk of medication-related osteonecrosis of the jaw (MRONJ): a systematic review. Heliyon. 2020;6(4):e03795.
Cummings SR, San Martin J, McClung MR, et al. Denosumab for prevention of fractures in postmenopausal women with osteoporosis. N Engl J Med. 2009;361(8):756-765.
Cummings SR, Ferrari S, Eastell R, et al. Vertebral fractures after discontinuation of denosumab: a post hoc analysis of the randomized placebo-controlled FREEDOM trial and its extension. J Bone Miner Res. 2018;33(2):190-198.
Tsourdi E, Langdahl B, Cohen- Solal M, et al. Discontinuation of denosumab therapy for osteoporosis: a systematic review and position statement by ECTS. Bone. 2017;105:11-17.
Bone HG, Wagman RB, Brandi ML, et al. 10 years of denosumab treatment in postmenopausal women with osteoporosis: results from the phase 3 randomised FREEDOM trial and open-label extension. Lancet Diabetes Endocrinol. 2017;5(7):513-523.
Ettinger B, Black DM, Mitlak BH, et al. Reduction of vertebral fracture risk in postmenopausal women with osteoporosis treated with raloxifene: results from a 3-year randomized clinical trial. Multiple Outcomes of Raloxifene Evaluation (MORE) investigators. JAMA. 1999;282(7):637-645.
Kanis JA, Harvey NC, McCloskey E, et al. Algorithm for the management of patients at low, high and very high risk of osteoporotic fractures. Osteoporos Int. 2020;31(1):1-12.
Cosman F. The evolving role of anabolic therapy in the treatment of osteoporosis. Curr Opin Rheumatol. 2019;31(4):376-380.
Cosman F, Nieves JW, Dempster DW. Treatment sequence matters: anabolic and antiresorptive therapy for osteoporosis. J Bone Miner Res. 2017;32(2):198-202.
Sleeman A, Clements JN. Abaloparatide: a new pharmacological option for osteoporosis. Am J Health Syst Pharm. 2019;76(3):130-135.
Harper KD, Krege JH, Marcus R, Mitlak BH. Osteosarcoma and teriparatide? J Bone Miner Res. 2007;22(2):334.
Cosman F, Crittenden DB, Ferrari S, et al. FRAME study: the foundation effect of building bone with 1 year of romosozumab leads to continued lower fracture risk after transition to denosumab. J Bone Miner Res. 2018;33(7):1219-1226.
Cosman F, Crittenden DB, Grauer A. Romosozumab treatment in postmenopausal osteoporosis. N Engl J Med. 2017;376(4):396-397.
Cosman F. Long-term treatment strategies for postmenopausal osteoporosis. Curr Opin Rheumatol. 2018;30(4):420-426.

