Continuing education approval: This activity has been evaluated and approved by the Continuing Education Approval Program of the National Association of Nurse Practitioners in Women’s Health (NPWH) for 1.0 continuing education contact hours including 0.35 contact hours of pharmacology content now through October 31, 2025.
Educational objectives: At the conclusion of this educational activity, participants should be able to:
1. Discuss potential causes of heavy menstrual bleeding in adolescents.
2. Describe components of an initial evaluation of heavy menstrual bleeding in adolescents.
3. Discuss the management of heavy menstrual bleeding in adolescents with hormonal and nonhormonal medications.
The authors have no actual or potential conflicts of interest in relation to the contents of this article.
Disclaimer: Participating faculty members determine the editorial content of the CE activity; this content does not necessarily represent the views of NPWH. This content has undergone a blinded peer review process for validation of clinical content. Although every effort has been made to ensure that the information is accurate, clinicians are responsible for evaluating this information in relation to generally accepted standards of care and integrating the information in this activity with that of established recommendations of other authorities, national guidelines, and individual patient characteristics.
Commercial support: This activity did not receive any commercial support.
Successful completion of the activity: Successful completion of this activity, J-23-05, requires participants to do the following:
1. “Sign in” at the top right-hand corner of the page pathlms.com/npwh/courses/57405 if you have an NPWH account. You must be signed in to receive credit for this course. If you do not remember your username or password, please follow the “Forgot Password” link and instructions on the sign-in page. If you do not have an account, please click on the “Create an Account.”*
2. Read the learning objectives, disclosures, and disclaimers.
3. Study the material in the learning activity during the approval period (now through October 31, 2025).
4. Complete the post-test and evaluation. You must earn a score of 70% or higher on the post-test to receive CE credit.
5. Print out the CE certificate after you have successfully passed the post-test and completed the evaluation.
*If you are an NPWH member, were once a member, or have completed CE activities with NPWH in the past, you have a username and password in our system. Please do not create a new account. Creation of multiple accounts could result in loss of CE credits as well as other NPWH services. If you do not remember your username or password, either click on the “Forgot Username” or “Forgot Password” link or call the NPWH office at (202) 543-9693, ext. 1.
The majority of adolescents have cycles every 21 to 45 days and have 2 to 7 days of bleeding at a time. Average blood loss ranges between 30 and 80 mL per cycle.1 Abnormal uterine bleeding (AUB) refers to uterine bleeding that is abnormal in volume, frequency, or length.2 Adolescents have a higher incidence of AUB than adults, with the most frequent presentation being heavy menstrual bleeding (HMB).1 Per the American College of Obstetricians and Gynecologists, HMB is excessive menstrual blood loss that hinders physical, social, emotional, or material quality of life.1
Heavy menstrual bleeding causes hundreds of adolescents to be hospitalized every year. Some present with severe anemia, up to 5% of whom require intensive-care admissions, whereas others present with anemia and/or iron deficiency.3 Adolescents experiencing HMB may have school absenteeism or decline in their physical or social activities that can lead to impacts on their mental health.3
Women’s health nurse practitioners in primary care settings are frontline in the initial assessment of adolescents with HMB and are often able to manage bleeding with pharmacologic therapies individualized to the patient’s particular needs. They are key in initiating a consultation or referral to a hematology specialist when a coagulopathy is suspected. This article describes potential causes of HMB in adolescents, initial assessment, and management that may include consultation or referral with hematology specialists.
Pathophysiology
The most common cause of AUB in the first few years after menarche is anovulation because of an immature hypothalamic-pituitary-ovarian axis.2 This occurs in up to 95% of adolescents with AUB and may manifest as irregular menses and/or HMB.4 Other potential causes of AUB in adolescents include pregnancy complications, sexually transmitted infections, and pelvic inflammatory disease. Conditions such as polycystic ovary syndrome and thyroid disorders can cause AUB that may include HMB.4
However, up to 20% of adolescents with HMB may have an underlying coagulopathy or bleeding disorder according to some studies.5,6 The true prevalence remains unknown due to the lack of prospective studies with objective menstrual flow assessment, standardized hemostatic testing, and standardized lab definitions of von Willebrand disease and platelet function defects.3
The most common diagnosed bleeding disorder causing HMB is von Willebrand disease, affecting 1% of the population.7 It is the most common bleeding disorder identified in adolescents with HMB, followed by platelet function defects. Less common bleeding disorders that may contribute to HMB include hemophilia carrier status, other clotting factor deficiencies, fibrinolytic disorders, joint hypermobility disorders with bleeding tendencies, and immune thrombocytopenia purpura.
Initial evaluation
In adolescents presenting to the primary care setting with AUB, baseline exclusions include sexually transmitted infections, pregnancy complications (eg, ectopic pregnancies, spontaneous abortion), and pelvic inflammatory disease. Once these have been excluded, the AUB PALM-COEIN [Polyp, Adenomyosis, Leiomyoma, Malignancy and hyperplasia, Coagulopathy, Ovulatory dysfunction, Endometrial, Iatrogenic, Not yet classified] system workup can be initiated.7 The PALM categories of AUB, which include structural causes, are rare in adolescents, making up only 1.3% of AUB in this population.8 Nonstructural causes, or COEIN [Coagulopathies, Ovulatory disorders, Endometrial disorders, Iatrogenic, Not yet classified], are the most common causes of AUB in adolescents, with ovulatory disorders being the most common.8
For adolescents presenting with HMB, the first step is to complete a detailed history. The provider should ask about menstrual history including when the patient obtained menarche, when HMB began, regularity of periods, any intermenstrual bleeding, passing clots, flooding, previous hormonal agents tried, failure to respond to a hormonal agent (if taken properly), number and type of products changed (particularly on heavy days), and number of heavy days per cycle. The utility of bleeding assessment tools has not been well studied in adolescents, but can be used with this in mind to help determine the need for further screening for bleeding disorders.7 The Pictorial Blood Assessment Chart (PBAC) and Questionnaire is a semi-quantitative method that allows women to track the number of pads or tampons used during a menstrual period and the degree of soiling. The score is based on that information. Scores of 100 or higher correlate with 80 mL or more of menstrual blood loss, as scientific consensus defines HMB as blood loss more than 80 mL per cycle. For women presenting with a PBAC score of greater than 100, this tool had a sensitivity of 89%, but in adolescents age 13 to 19 years the sensitivity was only 62%.3 PBAC can be found online at hemophilia.ca/files/Menstrual%20Assessment%20Chart.pdf. The International Society on Thrombosis and Haemostasis Bleeding Assessment Tool (ISTH-BAT) is a standardized method of reporting bleeding symptoms in both adults and children.3 This tool was designed to extend the utility of earlier BATs by including pediatric specific and symptom frequency and severity. An abnormal bleeding score for the ISTH-BAT for an adolescent, because they have not had as many bleeding challenges as adult women, remains unknown but likely falls between 2 and 5. The authors propose the use of ISTH-BAT prospectively in a comprehensive management protocol for adolescents with HMB under evaluation for a bleeding disorder.3 ISTH-BAT can be found on the ISTH reference tools page at https://www.isth.org/page/reference_tools.
It is important to ask the adolescent with HMB about symptoms of bleeding such as epistaxis, gum bleeding, hematuria, hematemesis, hematochezia, abnormally large bruising for the type of trauma, hematomas, spontaneous bruising, prolonged bleeding with injury, muscle or joint bleeding, and joint hypermobility. Ask about history of significant bleeding with minor procedures like dental work or ear tube placement, unexpected bleeding during or after surgical procedures or injuries, hemorrhage from ovarian cyst, hemorrhage requiring blood transfusion, and postpartum hemorrhage (especially after 24 hours).5
Family history may provide additional information about the potential for an inherited coagulation disorder. This includes asking about bleeding history in the family, such as members with HMB, those requiring blood transfusions after childbirth, postpartum hemorrhage or early hysterectomies, or family members with excessive bleeding after surgeries, members with frequent nose bleeds, or any other known bleeding concerns. As structural causes of HMB are uncommon in adolescents, imaging such as pelvic ultrasound may not be useful. In patients for whom initial medical management is ineffective, pelvic ultra-sonography may be considered.8 A speculum and/or bimanual exam is not needed as part of the evaluation.
Lab tests for iron deficiency should be obtained. It is important to assess both hemoglobin and ferritin because iron deficiency without anemia has been associated with increased muscle fatigue and decreased verbal learning and memory in adolescents.3,9 Anemia is defined as hemoglobin less than 12 g/dL in females 12 years and older. There are differing definitions of iron deficiency, but serum ferritin lower than 15 to 20 ng/L is a common cutoff.
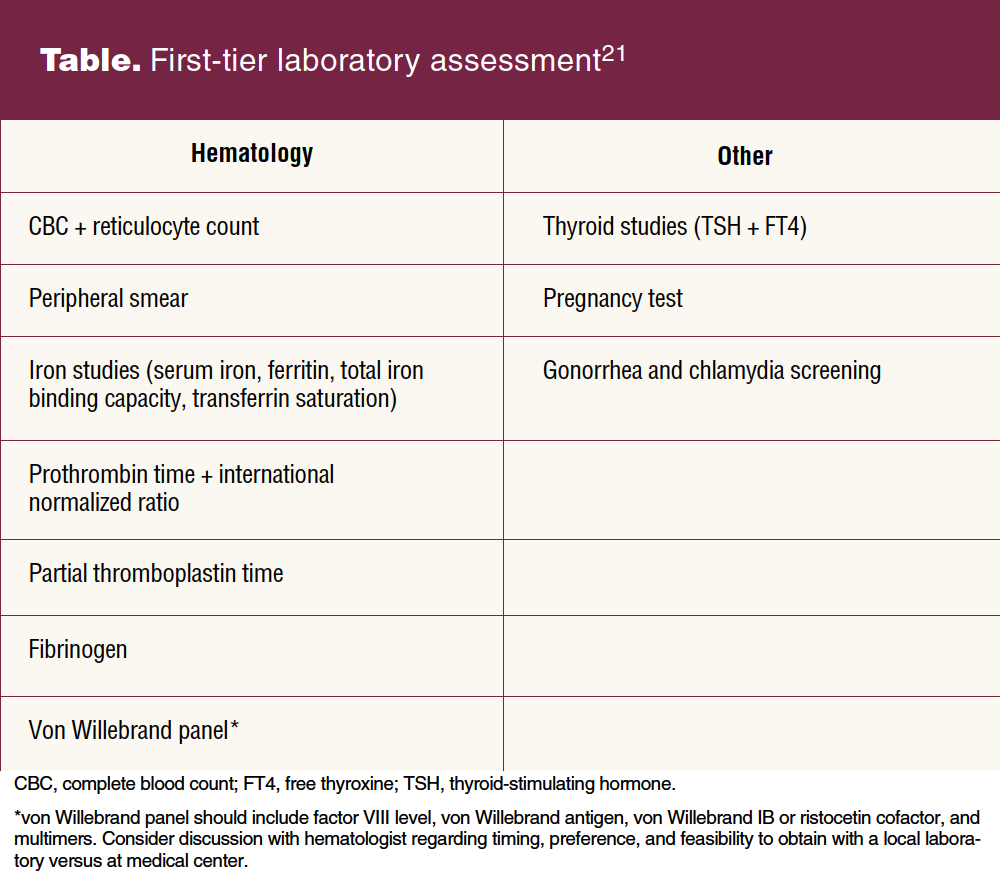
The Table lists first-tier laboratory tests that may be considered when there is a concern about a potential bleeding disorder. Coagulation studies should be collected on site with immediate processing because delays can lead to artificially low coagulation levels due to variables like temperature or sample stagnation that the reference laboratory cannot control. High-dose estrogen therapy elevates von Willebrand factor (vW) levels, but testing while on oral contraceptives with 30 to 35 mg of estrogen is unlikely to affect the results.3 It is recommended to obtain vW testing while the adolescent is having a period to capture the lowest baseline, although this has not been conclusively proved. It is not recommended to delay or withhold hormonal treatment to complete testing for von Willebrand disease.
If the provider suspects a patient has an underlying bleeding disorder, they should refer the patient to or collaborate with a hematologist for evaluation and management.1 Indications for referral to hematology would include identification of HMB with a PBAC score of more than 100, if menstrual bleeding is not controlled by pharmacologic therapies, if there are additional current or history of bleeding symptoms along with HMB or if there are abnormal laboratory findings including anemia, iron deficiency, or initial abnormal coagulation laboratory findings. Abnormal laboratory findings can include either mild or significant abnormalities. Abnormal initial laboratory findings are not a requirement for referral if there are bleeding concerns.
Hematology evaluation
Hematology specialist evaluation for HMB includes a systematic search for possible coagulation disorders. If first-tier testing as described in the Table is negative, the hematologist generally uses a stepwise strategy based on history and exam findings for additional testing to evaluate for platelet function defects and if indicated for other bleeding disorders. Genetic testing may be required to confirm a diagnosis of hemophilia carrier status if there is a suspicion.6 The role of the hematologist is to perform a comprehensive clinical and laboratory evaluation based on the degree of clinical suspicion; make a diagnosis; identify and manage concomitant iron deficiency; and provide input to the primary care provider regarding management of the HMB, especially for those with identified coagulation disorders.3 A hematologist consultation also is critical for evaluation of the safety of using medications such as aspirin or nonsteroidal anti-inflammatory drugs, presurgical evaluation for coagulation considerations, and for collaborative care if the patient is considering becoming or is pregnant.
Medical management
Management of HMB consists of both hormonal and nonhormonal therapies and can typically be managed in the outpatient setting. The treatment plan should be tailored for each individual patient. The customization process should consider the following aspects: the individual’s health history including previously tried hormonal therapies, individual preferences, availability, affordability, adherence, and the need for contraception.10
Hormonal therapy options
Progestin-only hormone therapy or combined estrogen-progestin contraceptives can be used for management of both acute and chronic HMB.5 Combined estrogen-progestin contraceptives are available through taking a daily pill, applying a weekly patch, or inserting a monthly vaginal ring.10 These combined methods are ideal for patients who have no contraindications to estrogen such as migraine with aura, systemic lupus erythematosus with antiphospholipid antibodies, or arterial or venous thromboembolic disease.11 A patient may consider extended or continuous cycling with any of these combined hormonal methods with a goal of having fewer bleeding episodes or amenorrhea.12 Cochrane reviews have demonstrated that combined hormonal contraceptive therapies, no matter the cycling method (monthly, extended, or continuous), led to an improvement in the bleeding pattern of the menstrual cycle.12
Progestin-only methods should be considered for patients who have a contraindication to estrogen, or who desire fewer menstrual cycles, or amenorrhea.5,11 These methods entail taking a daily pill, receiving an injection every 3 months, or placement of an implant in the arm or a progestin-releasing intrauterine device.12 A prospective study found that the highest rate of success for managing HMB among adolescent patients with an underlying bleeding disorder was with the levonorgestrel intrauterine device followed by norethindrone acetate taken orally.13
When bleeding is acute (deemed to be severely heavy), the primary goal is to stop the bleeding and ensure the patient is medically stable.10 Intravenous conjugated estrogen plus a regimen of tapered combined hormonal contraception pills is the primary hormonal therapy to initiate taking into account that the patient has no contraindications to estrogen.5 For patients who are not candidates for estrogen, high doses of a progestin taken orally or as an injection given intramuscularly or subcutaneously can be used to cease the bleeding.5 Depending on the severity, inpatient hospitalization and blood products may be required.10 After the patient has been stabilized and the initial treatment plan has been completed, the patient should continue to be seen for follow-up by the hematology specialist in the outpatient setting for further evaluation and to determine a long-term management plan.
Nonhormonal therapy options
For various reasons, some patients and families are not comfortable with using hormonal contraceptive therapy for medical indications such as HMB. Antifibrinolytics such as tranexamic acid or aminocaproic acid are nonhormonal therapy options. These medications can be taken once a month to decrease episodes of heavy bleeding associated with the menstrual cycle.14
Antifibrinolytics decrease HMB by binding to the lysine receptor on plasminogen. This binding prevents plasmin from joining and securing the fibrin matrix.
Based on the open-label, single-arm, efficacy study of tranexamic acid in adolescents with HMB, the suggested regimen to decrease menstrual blood loss with tranexamic acid consists of taking 1,300 mg (2 pills) three times a day for up to 5 days a month.14 In situations of acute HMB, these nonhormonal medications can be given orally or intravenously, either independently or combined with hormonal therapy.12 Tranexamic acid can be problematic for some patients due to the pill’s large size and its inability to be crushed, at which point aminocaproic acid in liquid form should be considered. Adverse effects associated with taking these medications include but are not limited to a risk of venous thrombosis such as a pulmonary embolism or deep vein thrombosis, seizures, gastrointestinal side effects, and vision disturbances. This medication is an appropriate option for patients who desire nonhormonal therapy for treatment of HMB but is contradicted for select patient populations: patients who have a known allergy to it, intracranial bleeding, known defective color vision, patients with an active or history of venous or arterial thrombosis, and being more than 3 hours after a traumatic injury.15 See the Box for more detailed information about antifibrinolytics.16–18
Box. Antifibrinolytics16–18
- Tranexamic acid and aminocaproic acid
- Mechanism of action: prevention of fibrinolysis by reducing activity of plasmin, which is responsible for breaking up fibrin in blood clots
- Contraindications: personal or immediate family history of blood clots or stroke, personal history of color blindness or kidney impairment; use cautiously in patients concurrently taking hormonal therapies
- Common side effects (> 10%): abdominal pain, headache, back and musculoskeletal pain, nasal symptoms
- Serious side effects: hypersensitivity reactions both immediate and delayed, visual defects, seizures with high IV doses, and thrombosis
- Outpatient dosing: tranexamic acid 650 mg–1,300 mg 3 times daily for up to 5 days once a month only available in tablet form at outpatient pharmacies; aminocaproic acid 50–100 mg/kg every 6 hours (max 24 g/day) may be taken up to 2 weeks but recommend only taking during bleeding, available in both tablet and solution forms
Management of iron deficiency
Iron supplementation should be incorporated into the treatment regimen when iron deficiency or iron deficiency anemia is identified. Providing once-a-day 65 to 130 mg elemental iron, depending on the severity of anemia, is recommended to continue for a minimum of 3 months.
Re-evaluate the ferritin level, during a time when the individual is without illness, after 3 months of iron supplementation to determine response and potential need for further supplementation. Because adolescents often have iron-deficient diets, they should be educated about consuming iron-rich foods.9 For symptomatic iron deficiency anemia or difficulty tolerating oral iron formulations, intravenous iron replacement may be given. Red-blood-cell transfusions along with intravenous iron replacement may be needed for patients with severe anemia.3
Psychosocial considerations
Adolescents with HMB with and without bleeding disorders have been found to have lower health-related quality of life compared to those without it.19 Many can have emotional effects as they deal with a new diagnosis and managing their bleeding symptoms.19 One recent study compared adolescents with HMB to those without it, finding that depression occurred in 50.9% of those with HMB compared to 24.2% of those without HMB.20
Screening for and management of depression should be part of regular clinical care. Patients with bleeding disorders can be offered support through counseling resources and local and national resources such as the Foundation for Women and Girls with Blood Disorders.19
Conclusion
Although HMB in the adolescent population is most often due to anovulation because of an immature hypothalamic-pituitary-ovarian axis, it may be the first symptomatic presentation of a coagulation disorder. It is critical that the primary care provider remain cognizant of and careful to assess for this possibility. A hematologic evaluation is imperative when there is suspicion of such to identify common or rare bleeding disorders.
Ideally, the adolescent with a bleeding disorder should be referred to a multidisciplinary clinic where they can receive comprehensive care from hematology and gynecology providers with specialized knowledge in the care of adolescents as well as psychosocial support. When ongoing care in a specialty setting is not feasible, the primary care provider should collaborate with hematology specialists in ongoing evaluation and management of the adolescent with a bleeding disorder.
Womens Healthcare. 2023;11(5):4-9. doi: 10.51256/WHC102304
References
1 American College of Obstetricians and Gynecologists. Committee Opinion no. 651. Menstruation in girls and adolescents: using the menstrual cycle as a vital sign. Obstet Gynecol. 2015;126(6):e143-e146.
2 Munro MG, Critchley HOD, Fraser IS; FIGO Menstrual Disorders Committee. The two FIGO systems for normal and abnormal uterine bleeding symptoms and classification of causes of abnormal uterine bleeding in the reproductive years: 2018 revisions. Int J Gynaecol Obstet. 2018;143(3):393-408.
3 O’Brien SH. Evaluation and management of heavy menstrual bleeding in adolescents: the role of the hematologist. Blood. 2018;132(20):2134-2142.
4 Deligeoroglou E, Karountzos V, Creatsas G. Abnormal uterine bleeding and dysfunctional uterine bleeding in pediatric and adolescent gynecology.
Gynecol Endocrinol. 2013;29(1):74-78.
5 Hernandez A, Dietrich JE. Abnormal uterine bleeding in the adolescent. Obstet Gynecol. 2020;135(3):615-621
6 Zia A, Rajpurkar M. Challenges of diagnosing and managing the adolescent with heavy menstrual bleeding. Thromb Res. 2016;143:91-100.
7 American College of Obstetricians and Gynecologists. Committee on Adolescent Health Care; Committee on Gynecologic Practice. Committee Opinion no. 580. von Willebrand disease in women. Obstet Gynecol. 2013;122(6):1368-1373.
8 Pecchioli Y, Oyewumi L, Allen LM, Kives S. The utility of routine ultrasound in the diagnosis and management of adolescents with abnormal uterine bleeding. J Pediatr Adolesc Gynecol. 2017;30(2):239-242.
9 Levy JH, Connors JM. Evaluation and management of oral anticoagulant bleeding: quid didicimus? (What have we learned?). J Thromb Haemost. 2023;21(3):460-462
10 Haamid F, Sass AE, Dietrich JE. Heavy menstrual bleeding in adolescents. J Pediatr Adolesc Gynecol. 2017;30(3):335-340
11 Lauring JR, Lehman EB, Deimling TA, et al. Combined hormonal contraception use in reproductive-age women with contraindications to estrogen use. Am J Obstet Gynecol. 2016;215(3):330.e1-330.e7.
12 Edelman A, Micks E, Gallo MF, et al. Continuous or extended cycle vs. cyclic use of combined hormonal contraceptives for contraception. Cochrane Database Syst Rev. 2014;2014(7):CD004695.
13 Alaqzam TS, Stanley AC, Simpson PM, et al. Treatment modalities in adolescents who present with heavy menstrual bleeding. J Pediatr Adolesc Gynecol. 2018;31(5):451-458.
14 O’Brien SH, Saini S, Ziegler H, et al. An open-label, single-arm, efficacy study of tranexamic acid in adolescents with heavy menstrual bleeding. J Pediatr Adolesc Gynecol. 2019;32(3):305-311.
15 Chauncey JM, Wieters JS. Tranexamic Acid. nih.gov. December 6, 2018. https://www.ncbi.nlm.nih.gov/books/NBK532909/.
16 Tranexamic acid. Hudson, OH: Lexicomp, 2023. http://online.lexi.com/. Updated May 11, 2023. Accessed May 13, 2023.
17 Aminocaproic acid. Hudson, OH: Lexicomp, 2023. http://online.lexi.com/. Updated May 11, 2023. Accessed May 13, 2023.
18 Bryant-Smith AC, Lethaby A, Farquhar C, Hickey M. Antifibrinolytics for heavy menstrual bleeding. Cochrane Database Syst Rev. 2018;4(4):CD000249.
19 American College of Obstetricians and Gynecologists. Committee Opinion no. 785. Screening and management of bleeding disorders in adolescents with heavy menstrual bleeding. Obstet Gynecol. 2019;134(3):e71-e83.
20 McGrath M, Quint EH, Weyand AC. Depression in adolescents and young adults with heavy menstrual bleeding in a referral clinic setting. Am J Hematol. 2021;96(4):E105-E108.
21 Zia A, Kouides P, Khodyakov D, et al. Standardizing care to manage bleeding disorders in adolescents with heavy menses–A joint project from the ISTH pediatric/neonatal and women’s health SSCs. J Thromb Haemost. 2020;18(10):2759-2774.
KEY WORDS: heavy menstrual bleeding, abnormal uterine bleeding, von Willebrand disease, adolescent, coagulopathies, bleeding disorders