Faculty: Kendra L. Roloff, DNP, APRN, WHNP-C, CUNP, MBA, is a certified women’s health and urology nurse practitioner at Pelican Health in Bismarck, North Dakota.
Intended audience: This continuing education (CE) activity has been designed to meet the educational needs of nurse practitioners and other healthcare providers who provide primary care for women.
CE approval period: Now through October 31, 2023
Estimated time to complete this activity: 1 hour
CE approval hours: 1.0 contact hour of CE credit including 0.25 contact hours of pharmacology content
Goal statement: Nurse practitioners and other healthcare providers who provide primary care for women will increase their knowledge about diagnosis and both nonpharmacologic and pharmacologic management of interstitial cystitis/bladder pain syndrome (IC/BPS).
Needs assessment: IC/BPS affects up to 3% to 6% of adult women in the United States and is poorly understood. Diagnosis and proper management are often delayed by 3 to 6 years. IC/BPS is often chronic and is associated with multiple comorbid conditions including fibromyalgia, depression, irritable bowel syndrome, and chronic fatigue syndrome. Dyspareunia is commonly reported. The pain and chronicity associated with IC/BPS can significantly impact quality of life (QOL). Knowledge about signs/symptoms and diagnostic tools for IC/BPS is needed for earlier diagnosis so that evidence-based management can be implemented to improve QOL for women suffering from this condition.
Educational objectives: At the conclusion of this educational activity, participants should be able to:
- Identify signs/symptoms and appropriate evaluation for the diagnosis of IC/BPS.
- Discuss recommended first-line nonpharmacologic therapies for IC/BPS.
- Describe indications, mechanism of action, efficacy, adverse effects, and contraindications for pharmacologic options in treating IC/BPS.
Accreditation statement: This activity has been evaluated and approved by the Continuing Education Approval Program of the National Association of Nurse Practitioners in Women’s Health (NPWH) and has been approved for 1 contact hour CE credit, including 0.25 hours of pharmacology credit.
Faculty disclosures: NPWH policy requires all faculty to disclose any affiliation or relationship with a commercial interest that may cause potential, real, or apparent conflict of interest with the content of a CE program. NPWH does not imply that affiliation or relationship will affect the content of the CE program. Disclosure provides participants with information that may be important to their evaluation of an activity.
Kendra L. Roloff, DNP, APRN, WHNP-C, CUNP, MBA, has no actual or potential conflicts of interest in relation to the contents of this article.
Disclosure of unlabeled/unapproved use: NPWH policy requires authors to disclose to participants when they are presenting information about unlabeled use of a commercial product or device or an investigational use of a drug or device not yet approved for any use.
Disclaimer: Participating faculty members determine the editorial content of the CE activity; this content does not necessarily represent the views of NPWH. This content has undergone a blinded peer review process for validation of clinical content. Although every effort has been made to ensure that the information is accurate, clinicians are responsible for evaluating this information in relation to generally accepted standards in their own communities and integrating the information in this activity with that of established recommendations of other authorities, national guidelines, FDA-approved package inserts, and individual patient characteristics.
Successful completion of the activity: Successful completion of this activity, J-21-05, requires participants to do the following:
- “Sign In” at the top right-hand corner of the page if you have an NPWH account. You must be signed in to receive credit for this course. If you do not remember your username or password, please follow the “Forgot Password” link and instructions on the sign in page. If you do not have an account, please click on “Create an Account.”*
- Read the learning objectives, disclosures, and disclaimers on the next page and then click on the “Continue” button.
- Study the material in the learning activity during the approval period (now through October 31, 2023).
- Complete the post-test and evaluation. You must earn a score of 70% or higher on the post-test to receive CE credit.
- Print out the CE certificate after you have successfully passed the post-test and completed the evaluation.
*If you are an NPWH member, were once a member, or have completed CE activities with NPWH in the past, you have a username and password in our system. Please do not create a new account. Creation of multiple accounts could result in loss of CE credits as well as other NPWH services. If you do not remember your username or password, either click on the “Forgot Username” or “Forgot Password” link or call the NPWH office at (202) 543-9693, ext. 1.
Commercial support: This activity did not receive any commercial support.
Interstitial cystitis/bladder pain syndrome is defined as an unpleasant sensation of pain, pressure, or discomfort perceived to be related to the urinary bladder, associated with lower urinary tract symptoms of more than 6 weeks’ duration, in the absence of infection or other identifiable causes. Interstitial cystitis/bladder pain syndrome affects up to 3% to 6% of adult women in the United States and is poorly understood. Diagnosis and proper management are often delayed by 3 to 6 years. When managing interstitial cystitis/bladder pain syndrome, a multidisciplinary approach to care must be taken. The women’s health nurse practitioner is well positioned to identify the problem and initiate care.
Interstitial cystitis/bladder pain syndrome (IC/BPS) is defined by the American Urologic Association (AUA) and the Society for Urodynamics, Female Medicine & Urogenital Reconstruction as “An unpleasant sensation (pain, pressure, discomfort) perceived to be related to the urinary bladder, associated with lower urinary tract symptoms of more than 6 weeks’ duration, in the absence of infection or other identifiable causes.”1,2 This often chronic condition occurs in approximately 3% to 6% of women over the age of 18 in the United States.3 Multiple comorbid conditions exist with IC/BPS including fibromyalgia, depression, temporomandibular joint disorder, irritable bowel syndrome, constipation, chronic fatigue syndrome, and endometriosis. Pain with sexual intercourse is also commonly reported, as IC/BPS is believed to be related to an underlying pelvic floor dysfunction (PFD).1
Interstitial cystitis/bladder pain syndrome has been estimated to cost over $100 million annually in this country.4 The pain and chronicity associated with IC/BPS can significantly impact quality of life (QOL).2 This complex condition usually requires a multidisciplinary team approach for effective management of symptoms. The women’s health nurse practitioner (WHNP) is well positioned to be a valuable member of this team to improve QOL for individuals with IC/BPS. The purpose of this article is to describe current understanding regarding the pathology of IC/BPS, review recommended assessment and diagnostic techniques, and provide details regarding strategies for management.
Pathogenesis
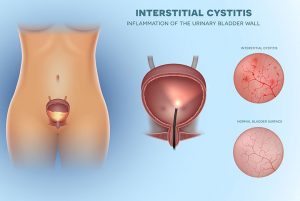 Interstitial cystitis/bladder pain syndrome remains poorly understood, and researchers speculate the etiology to be multifactorial. The detrusor smooth muscle of the urinary bladder is covered with a mucosal lining called the urothelium. The layer of the urothelium that protects the epithelial cells from irritants is called the glycosaminoglycan (GAG) layer. If the GAG layer is altered in such a way that it allows irritants to come into contact with the epithelial cells, pain and inflammation can occur.5 It is rare for alterations in the GAG layer to be visible via cystoscopicexam, but, in 10% of cases, Hunner’s lesions are identified. These lesions are characterized as distinctive reddened areas on the mucosal lining. The lesions rupture with bladder distention causing glomerulations.6 When the GAG layer is altered, mast cells are activated by both cytokines and an excess of potassium that diffuses past the altered urothelium. The activation of mast cells contributes to inflammation of the bladder wall causing chronic pain, urinary urgency, and urinary frequency, all of which are common symptoms experienced by individuals with IC/BPS.5
Interstitial cystitis/bladder pain syndrome remains poorly understood, and researchers speculate the etiology to be multifactorial. The detrusor smooth muscle of the urinary bladder is covered with a mucosal lining called the urothelium. The layer of the urothelium that protects the epithelial cells from irritants is called the glycosaminoglycan (GAG) layer. If the GAG layer is altered in such a way that it allows irritants to come into contact with the epithelial cells, pain and inflammation can occur.5 It is rare for alterations in the GAG layer to be visible via cystoscopicexam, but, in 10% of cases, Hunner’s lesions are identified. These lesions are characterized as distinctive reddened areas on the mucosal lining. The lesions rupture with bladder distention causing glomerulations.6 When the GAG layer is altered, mast cells are activated by both cytokines and an excess of potassium that diffuses past the altered urothelium. The activation of mast cells contributes to inflammation of the bladder wall causing chronic pain, urinary urgency, and urinary frequency, all of which are common symptoms experienced by individuals with IC/BPS.5
Some researchers speculate that the immune system is involved with IC/BPS. Compared to the general population, individuals with IC/BPS have a higher prevalence of autoimmune conditions such as systemic lupus erythematosus, inflammatory bowel disorders, Sjogren’s syndrome, and rheumatoid arthritis. Autoantibodies have been detected in the serum and urine of patients with IC/BPS, which is consistent with immune involvement.7 The role of the nervous system is another area of IC/BPS research. When the nerves that carry bladder sensations dysfunction, pain can be caused by events that are not normally painful.7 Pelvic floor dysfunction is yet another facet of IC/BPS. It is well documented that individuals with IC/BPS experience pelvic pain related to pelvic floor muscle spasm. In fact, up to 85% of individuals with IC/BPS experience PFD.8 When the bladder wall is inflamed and causing bladder pain, it is believed to activate the pelvic floor muscles in such a way that a hypertonic contracted state results. This is essentially a state of chronic muscle tightness or spasm that contributes to pelvic pain.8
Evaluation
Interstitial cystitis/bladder pain syndrome can easily be overlooked for conditions such as overactive bladder, urinary tract infection (UTI), vaginitis, pelvic floor disorders, or endometriosis. The most common symptom of IC/BPS is suprapubic discomfort related to bladder filling and relieved with bladder emptying.3 Other common symptoms of IC/BPS include urinary urgency, urinary frequency, nocturia, and dysuria. It is primarily a diagnosis of exclusion. Thus, a thorough evaluation requires a detailed history and focused physical exam. The history should include questions about symptom type, severity, and duration as well as any previous evaluation findings and treatments.9
A physical exam is important to detail the location of pain and to determine if anatomy is within normal limits. The exam should focus on anterior vaginal wall tenderness, suprapubic tenderness, presence of pelvic floor hypertonicity, integrity of the vaginal tissue, and signs of pelvic organ prolapse.9 Table 1 compares history and exam findings for differential diagnosis.
 A 24-hour frequency-volume chart can be used to detail important bladder habits including number of voids per day, daily fluid intake, and daily urine output. A normal voiding chart is represented by less than 8 voids per day, nocturia 0 to2 times, and voided volumes of 300 to 400 mL.9
A 24-hour frequency-volume chart can be used to detail important bladder habits including number of voids per day, daily fluid intake, and daily urine output. A normal voiding chart is represented by less than 8 voids per day, nocturia 0 to2 times, and voided volumes of 300 to 400 mL.9
A urine dipstick or microscopic urinalysis is recommended to rule out UTI. If the urinalysis is abnormal, a urine culture is necessary. Cystoscopy is not typically required for a diagnosis of IC/BPS, but it is useful to rule out other conditions including bladder cancer when the diagnosis is uncertain. The presence of Hunner’s lesions seen on cystoscopy indicates advanced disease.10
 Potassium sensitivity testing was previously utilized as a diagnostic tool for IC/BPS. The potassium sensitivity test was believed to test the permeability of the GAG layer of the bladder, which is impaired in some patients with IC/BPS. The test is no longer recommended because testing in this manner was painful and induced unnecessary flares for patients.11 A more widely accepted evaluation tool is the anesthetic bladder challenge. A solution of lidocaine, sodium bicarbonate, and sometimes heparin is instilled into the bladder. Individuals with IC/BPS and in a state of pain may experience temporary relief from the instillation given its anesthetic effect on the bladder wall. The challenge is useful to determine if pain is stemming from the bladder or another source such as the pelvic floor muscle.11
Potassium sensitivity testing was previously utilized as a diagnostic tool for IC/BPS. The potassium sensitivity test was believed to test the permeability of the GAG layer of the bladder, which is impaired in some patients with IC/BPS. The test is no longer recommended because testing in this manner was painful and induced unnecessary flares for patients.11 A more widely accepted evaluation tool is the anesthetic bladder challenge. A solution of lidocaine, sodium bicarbonate, and sometimes heparin is instilled into the bladder. Individuals with IC/BPS and in a state of pain may experience temporary relief from the instillation given its anesthetic effect on the bladder wall. The challenge is useful to determine if pain is stemming from the bladder or another source such as the pelvic floor muscle.11
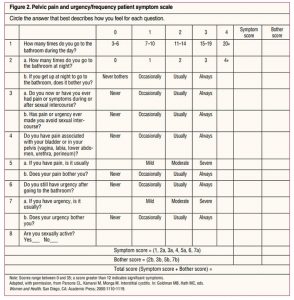 The O’Leary-Sant symptom index and pelvic pain and urgency/frequency (PUF) questionnaire are both tools that can be useful to determine baseline symptoms and to follow a patient’s response to treatment. The O’Leary-Sant symptom index assesses QOL, whereas the PUF questionnaire can help determine if the patient’s pain is related to the bladder or reproductive tract. Neither of these tools has been validated for establishing a diagnosis.9 See Figure 1 for the O’Leary-Sant symptom index and Figure 2 for the PUF questionnaire.
The O’Leary-Sant symptom index and pelvic pain and urgency/frequency (PUF) questionnaire are both tools that can be useful to determine baseline symptoms and to follow a patient’s response to treatment. The O’Leary-Sant symptom index assesses QOL, whereas the PUF questionnaire can help determine if the patient’s pain is related to the bladder or reproductive tract. Neither of these tools has been validated for establishing a diagnosis.9 See Figure 1 for the O’Leary-Sant symptom index and Figure 2 for the PUF questionnaire.
Management
Management should focus on the most conservative therapies first, followed by less conservative options, and surgical intervention should be considered only when all conservative options have failed.2 The American Urologic Association provides an IC/BPS treatment algorithm. The algorithm separates management options into specific treatment line groups. Multiple simultaneous treatment options from various line groups may be warranted in order to achieve symptom control. If a treatment option is not improving an individual’s symptoms, it is recommended to discontinue the treatment.2,6
Education and self-care are considered first-line therapy.2 Self-care consists of dietary changes, bladder retraining, and stress management techniques. Acidic foods and fluids such as coffee, soda, other carbonated beverages, artificial sweeteners, citrus fruits, and alcohol can irritate the bladder and exacerbate symptoms.9 Bladder retraining is a method used to gradually increase the time between voids so that the bladder will hold larger volumes of urine for longer periods of time. Instead of allowing oneself to void with every urge, bladder retraining encourages an individual to be still once an urge is felt, perform pelvic floor muscle exercises, and use distraction methods such as deep breathing until the urge lessens or ceases. This method trains the bladder muscle to hold more urine so that the individual will experience less urges with and without urine loss.12 Bladder retraining may take several weeks to experience the desired outcomes.12 Stress management includes breathing techniques, meditation, exercise, or any other activity that helps an individual relax.9 When conservative first-line therapy fails, or if it has been determined an individual may benefit from first-and second-line therapies simultaneously, second-line therapy is offered. Second-line therapy includes physical therapy (PT) to address PFD, vaginal and/or oral medications, and intravesical instillations.2 Physical therapists with expertise in treating PFD use manual therapy to release myofascial trigger points and connective tissue of the lumbosacral region in an effort to reduce hypertonicity.8 It is important to understand that pelvic floor therapy aimed at strengthening the pelvic floor muscles (eg, Kegel exercises) is not recommended. Intravaginal muscle relaxants are also commonly used for symptom relief when pelvic floor hypertonicity is contributing to an individual’s pain. Baclofen and diazepam can be prescribed as a vaginal suppository and are often recommended prior to sexual intercourse or pelvic floor PT to decrease discomfort.13
Oral medications can be used alone or in combination with other therapies. Pentosan polysulfate is the only US Food and Drug Administration (FDA)-approved medication for IC/BPS. Pentosan polysulfate works by rebuilding the GAG layer and reducing bladder permeability.14 Clinical trials report 38% of individuals who utilized pentosan polysulfate for 3 months reported greater than 50% improvement in bladder pain.15
Recommended dosing of pentosan polysulfate is 100 mg orally three times per day at least 1 hour before a meal or 2 hours after a meal. Pentosan polysulfate may take 3 to 6 months for symptom improvement to occur. If symptoms do not improve after 6 months, the medication should be discontinued. Recent studies have linked long-term use (> 3 years) of pentosan polysulfate with vision-threatening side effects.16 Pentosan polysulfate is classified as a pregnancy category B medication by the FDA meaning animal reproduction studies have failed to demonstrate a risk to the fetus and no adequate and well-controlled studies have been performed in pregnant women. Despite the potential side effects, the medication remains on IC/BPS guidelines.14
Tricyclic antidepressants such as amitriptyline block the reabsorption of the neurotransmitter’s serotonin and norepinephrine. Increased levels of serotonin and norepinephrine decrease pain and in turn can reduce urinary urgency and frequency.14 Controlled and noncontrolled studies have demonstrated efficacy of amitriptyline to range between 50% and 70%. Recommended dosing of amitriptyline is 10 mg to 25 mg daily, titrated weekly to a target dose of 75 mg to 100 mg daily for up to 23 months.16 Amitriptyline is classified as a pregnancy category C medication, which means animal studies have demonstrated adverse effects, but there are no adequate and well-controlled studies available in humans.17 Side effects associated with tricyclic antidepressants include constipation, dry mouth, vision problems, and sedation. Nearly 80% of individuals develop side effects and are not able to tolerate therapeutic doses of tricyclic antidepressants.14
Antihistamines such as hydroxyzine and cimetidine have also been used to help manage IC/BPS. The bladder wall has mastcells similar to those that generate allergy symptoms. The release of histamine has been demonstrated to evoke an inflammatory response by the bladder, which causes pain. The recommended dose of cimetidine is 400 mg orally twice daily and the recommended dose of hydroxyzine is 10 mg to 50 mg orally at bedtime.14 Observational studies using hydroxyzine and cimetidine have demonstrated up to 90% and 57% reduction in IC/BPS symptoms, respectively.1,14 The side-effect profile of antihistamines is minimal, with sedation being reported most frequently. Cimetidine is classified as pregnancy category B and hydroxyzine is classified as pregnancy category C.14
Intravesical instillations have been reported to exhibit anti-inflammatory and analgesic effects on the bladder by reconstituting the GAG layer.1 The AUA guidelines list dimethyl sulfoxide (DMSO), lidocaine, and heparin as instillation agents of choice.2 Dimethyl sulfoxide is the only FDA-approved medication for intravesical instillation and is used alone or in combination with other medications. A typical regimen consists of weekly instillations for 6 weeks. Dimethyl sulfoxide is classified as pregnancy level C.14 See Table 2 for a summary of second-line medication therapy.
 Per AUA guidelines, cystoscopy with hydrodistension is listed under third-line therapy.2 Hydrodistension stretches the bladder muscle, thereby breaking down fibrosis and causing ischemia to nerve fibers. The effects of hydrodistension include an increase in bladder capacity and a decrease inurinary urgency.18
Per AUA guidelines, cystoscopy with hydrodistension is listed under third-line therapy.2 Hydrodistension stretches the bladder muscle, thereby breaking down fibrosis and causing ischemia to nerve fibers. The effects of hydrodistension include an increase in bladder capacity and a decrease inurinary urgency.18
Fourth-line therapy for IC/BPS includes intravesical botulinum toxin A and sacral neuromodulation (SNM).2 Intravesical botulinum toxin A injections can be done in the office using intravesical lidocaine for pain control. The injection into the bladder muscle is performed via a cystoscope and acts by relaxing the smooth muscle and controlling inflammation.19 Potential side effects of intravesical botulinum toxin A include an elevated post-void residual, gross hematuria, difficult urination, and UTIs.19
The minimally invasive surgical procedure, SNM, is an option for refractory IC/BPS.2 Stimulation of the sacral nerves can improve pain associated with IC/BPS and decrease urinary urgency and frequency by inhibiting the urinary tract reflexes. In SNM, a neurostimulator device is surgically implanted that delivers stimulation to the sacral nerve via mild electrical pulses.20
Following AUA guidelines, fifth-line therapy includes cyclosporine A and sixth-line therapy includes surgical options such as urinary diversion or substitution cystoplasty.2 Cyclosporine A is an immunosuppressive agent that interferes with the activity and growth of cytokine secreting T cells that contribute to inflammation.21 A urinary diversion procedure involves removing the urinary bladder and constructing a new way for urine to exit the body.22 Substitution cystoplasty is a surgical procedure performed to make the urinary bladder larger.23
In addition to the treatment algorithm recommended by the AUA, providers must consider an individual’s mental health when managing IC/BPS. One study indicated women with IC/BPS are 23% more likely to have depression and anxiety than women without IC/BPS.8 Involving a counselor with the management of IC/BPS can optimize treatment outcomes.2
Practice implications
IC/BPS is a complex problem often requiring a multidisciplinary approach to care. The WHNP may likely be the first provider the patient has seen who can navigate her symptoms and get her on a path to proper management. Therefore, building a meaningful rapport is paramount, especially when the WHNP is the point of communication across multiple specialties including PT, urology, gynecology, colorectal surgeons, mental health, and integrative medicine.8
After years of failed treatments and chronic pain, patients with IC/BPS often give up hope.8 Through education, support, good communication, and recommendations for effective management techniques, the WHNP can restore the patient’s hope. The WHNP has the unique ability to recognize IC/BPS and transform the patient’s experience from ordinary to extraordinary.
References
- Han E, Nguyen L, Sirls L, Peters K. Current best practice management of interstitial cystitis/bladder pain syndrome. Ther Adv Urol. 2018;10(7):197-211.
- Hanno PM, Erickson D, Moldwin R, Faraday MM; American Urological Association. Diagnosis and treatment of interstitial cystitis/bladder pain syndrome: AUA guideline amendment. J Urol. 2015;193(5):1545-1553.
- International Continence Society. Interstitial cystitis/bladder pain syndrome (IC/BPS). Updated March 2018.
- Cardenas-Trowers O. Clinical trial comparing two bladder instillations for IC/BPS. Updated October 27, 2020.
- Hurst RE, Greenwood-Van Meerveld B, Wisniewski AB, et al. Increased bladder permeability in interstitial cystitis/painful bladder syndrome. Transl Androl Urol. 2015;4(5):563-571.
- Jerauld A, Wormuth L, Carlson B. New approaches in managing interstitial cystitis/bladder pain syndrome. US Pharm. 2016;41(9):29-33.
- Duh K,Funaro MG, DeGouveia W, et al. Crosstalk between the immune system and neural pathways in interstitial cystitis/bladder pain syndrome. Discov Med. 2018;25(139):243-250.
- Gupta P, Gaines N, Sirls LT, Peters KM. A multidisciplinary approach to the evaluation and management of interstitial cystitis/bladder pain syndrome: an ideal model of care. Transl Androl Urol. 2015;4(6):611-619.
- Cox A, Golda N, Nadeau G, et al. CUA guideline: diagnosis and treatment of interstitial cystitis/bladder pain syndrome. Can Urol Assoc J. 2016;10(5-6):E136-E155.
- Meijlink JM. Interstitial cystitis/bladder pain syndrome. An overview of diagnosis & treatment. International Painful Bladder Foundation. October 2020.
- Interstitial Cystitis Association. Potassium sensitivity test. Updated June 30, 2015.
- Lee HE, Oh SJ. The effectiveness of bladder training in overactive bladder. Curr Bladder Dysfunct Rep. 2014;9(1):63-70.
- Larish AM, Dickson RR, Kudgus RA, et al. Vaginal diazepam for nonrelaxing pelvic floor dysfunction: the pharmacokinetic profile. J Sex Med. 2019;16(6):763-766.
- Doiron RC, Bona M, Nickel JC. Possible drug-induced, vision-threatening maculopathy secondary to chronic pentosan polysulfate sodium (Elmiron®) exposure. Can Urol Assoc J. 2020;14(2):10-11.
- US Food and Drug Administration. Elmiron-100 mg capsules prescribing information. Updated June 2020.
- Garzon S, Laganà AS, Casarin J, et al. An update on treatment options for interstitial cystitis. Prz Menopauzalny. 2020;19(1):35-43.
- US Department of Health and Human Services. FDA pregnancy categories. Updated March 8, 2021.
- Homma Y, Akiyama Y, Tomoe H, et al. Clinical guidelines for interstitial cystitis/bladder pain syndrome. Int J Urol. 2020;27(7):578-589.
- Chen JL, Kuo HC. Clinical application of intravesical botulinum toxin type A for overactive bladder and interstitial cystitis. Investig Clin Urol. 2020;61(suppl 1):S33-S42.
- Wang J, Chen Y, Chen J, et al. Sacral neuromodulation for refractory bladder pain syndrome/interstitial cystitis: a global systematic review and meta-analysis. Sci Rep. 2017;7(1):11031.
- Wang Z, Zhang L. Treatment effect of cyclosporine A in patients with painful bladder syndrome/interstitial cystitis: a systematic review. Exp Ther Med. 2016;12(1):445-450.
- Cleveland Clinic. Urinary reconstruction & diversion. Updated September 14, 2020.
- Cleveland Clinic. Augmentation cystoplasty (bladder augmentation). Updated June 1, 2015.
- https://my.clevelandclinic.org/health/treatments/15846-augmentation-cystoplasty-bladder-augmentation.
Web resource: npwh.org/courses/home/details/1635
KEY WORDS: interstitial cystitis, bladder pain, lower urinary tract symptoms, bladder pain symptoms, bladder pain treatment, pelvic floor dysfunction.