Recurrent pregnancy loss (RPL) can be a very distressing diagnosis with far-reaching implications, particularly psychological anguish. Women’s health nurse practitioners (WHNPs) can undertake an initial assessment for those who experience RPL and those who specialize in fertility can provide more extensive evaluation and treatment as collaborative team members. WHNPs are in a unique position to educate patients on the potential causes of RPL, initiate diagnostic testing, and refer to specialized settings, as well as offer emotional support and resources.
Key words: recurrent pregnancy loss, reproductive-age couple, products of conception, congenital uterine anomalies, thyroid peroxidase, thyroid-stimulating hormone
Pregnancy loss is a relatively common event, occurring in 15% to 25% of clinically recognized pregnancies.1 Recurrent pregnancy loss (RPL) is much less common. Although a grief response is expected with any pregnancy loss, RPL can be an extremely distressing diagnosis for patients that has far-reaching implications including psychological anguish.1,2 Additionally, it can be challenging for healthcare providers to establish the diagnosis, evaluate to determine the etiology, and recommend treatment.3 Using a well-established evidence-based approach can be difficult to accomplish due to the enigmatic nature of RPL as well as conflicting guidelines, leaving patients and their providers feeling hopeless or frustrated.4,5 Women’s health nurse practitioners (WHNPs) in a primary care setting can conduct an initial assessment for women/couples who experience RPL to guide next steps; provide general information about the potential causes, further evaluation, and available resources for referral; and give emotional support. WHNPs specializing in fertility collaborate as team members in more extensive evaluation and treatment. The purpose of this article is to describe the components of an initial assessment and provide an overview of potential etiologies and treatments for this complex and poorly understood diagnosis.
Initial assessment
The initial assessment of a reproductive-age couple for RPL can be challenging for reasons including conflicting guidelines for what constitutes the diagnosis and issues with the accuracy of self-reporting a history of pregnancy losses.1 RPL is defined by the American Society for Reproductive Medicine (ASRM) as the loss of two or more pregnancies, regardless of consecutiveness.4,6 How to best define RPL is continually debated among various interest groups.5,7 It is also important to note that currently no international consensus exists on treatment, risk factors, or definition.5 By ASRM’s criteria, RPL is not inclusive of biochemical, ectopic, or molar pregnancies and can be further classified into primary (no history of having a live birth), secondary (history of live birth with subsequent losses), or tertiary (multiple live births that have had pregnancy losses interspersed).1,4,8 RPL affects 2% to 5% of reproductive-age couples.4 Even this statistic, however, is debated as a result of differing diagnostic criteria.3,4
The most important element of an initial assessment for RPL is eliciting a comprehensive and careful history from the couple in an empathetic manner. The obstetric history is crucial and provides many needed details to guide the diagnostic process. Revisiting the details of prior pregnancy losses may be difficult for the couple. The WHNP should acknowledge this and provide emotional support and compassion during this part of the process.7
Critical components of the obstetric history are number of prior pregnancies, pregnancy outcomes, and results of any genetic testing that was performed on the products of conception (POC) with a pregnancy loss.8 The timing of the pregnancy loss may provide an important clue to the etiology or steer the workup (eg, single unexplained loss of morphologically normal fetus > 10 weeks’ gestation is an indication for antiphospholipid testing).1,3 A thorough review of how each of the lost pregnancies was documented (via ultrasound or histopathology) and what sonographic signs were present (eg, empty gestational sac, fetal heartbeat) is important.4
The history should also include querying various lifestyle factors, including environmental toxin exposure, substance use, and whether there is a personal or family history of thrombophilia and/or a family history of recurrent pregnancy loss. These particulars are all relevant in piecing together the clinical picture and guiding further evaluation.1,5,7
Etiology and further evaluation
Because multiple etiologies for RPL have been proposed, some better supported than others, the evaluation for a cause can be expensive and exhausting.9 The potential etiologies are categorized broadly into genetic, anatomic, immunologic, endocrinologic, or thrombotic (Table). Perhaps the most frustrating category is unexplained RPL (URPL).2,3 Even the most thorough of investigations yield a cause less than 50% of the time.1,9 Genetic factors account for about 25% of RPL.2 Genetic analysis is not routinely recommended and when considered should include genetic counseling to assess individual risk factors. Evaluation for a genetic etiology falls into two main categories: parental analysis (karyotypes to detect chromosomal abnormalities of a maternal or paternal origin, such as balanced translocations) and POC analysis (to detect pregnancy tissue chromosomal abnormalities).5 If the POC karyotype shows that the embryo had an abnormal number of chromosomes (aneuploidy) the etiology for that loss is essentially known, given that the most common cause of early pregnancy loss is embryonic aneuploidy.4,8 If the embryo was euploid with the normal number of chromosomes, a full diagnostic workup to investigate for other causes should ensue.4 The risk of having a pregnancy loss related to embryonic aneuploidy increases as maternal age advances, with the loss rate approaching 50% in women over the age of 40.1
Table. Recurrent pregnancy loss: Etiology, evaluation, and potential findings
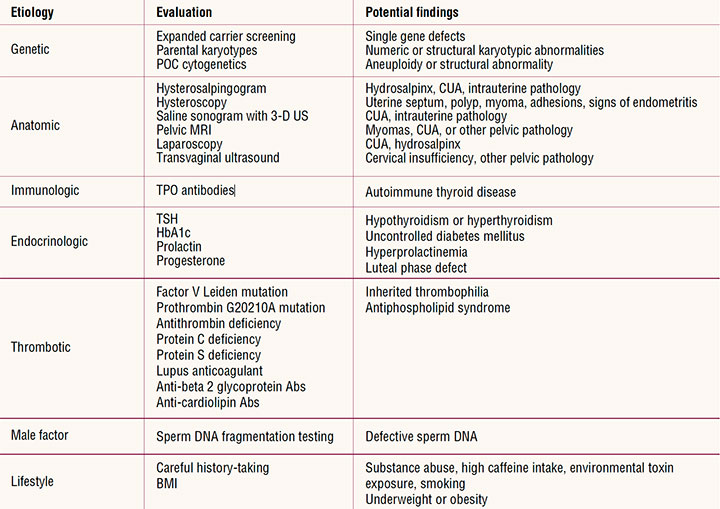
Abs, antibodies; BMI, body mass index; CUA, congenital uterine anomalies; MRI, magnetic resonance imaging; POC, products of conception; TPO, thyroid peroxidase; TSH, thyroid-stimulating hormone; US, ultrasound.
Anatomic uterine abnormalities are also potential causes of RPL and fall into the categories of congenital or acquired. They can be diagnosed using hysterosalpingogram, hysteroscopy, saline sonogram, pelvic magnetic resonance imaging, laparoscopy, or 3-D ultrasound.8 Congenital uterine anomalies are found in 12.6% of patients with RPL and include any structural defect resulting from abnormal Müllerian duct development such as septate, unicornuate, bicornuate, or didelphys uteri.1,4 They are usually associated with second trimester pregnancy loss.1 Of these anomalies, septate is the one most highly associated with pregnancy loss.1 Acquired uterine abnormalities that may distort the uterine cavity include endometrial polyps, intrauterine adhesions, and fibroids.4 Cervical insufficiency, which can be congenital or acquired, is typically associated with second trimester losses.4
Successful embryo implantation involves a complex interplay of immune activation at the maternal-fetal interface. If this is dysfunctional or absent, then there is a risk of rejection.10 Immunologic factors contributing to RPL can be categorized as autoimmune (response to one’s own antigens) or alloimmune (response to foreign antigens).8 Autoimmune factors encompass antiphospholipid syndrome (APS) (described here under autoimmune thrombophilia), thyroid autoimmunity, antinuclear antibodies (ANA), and celiac disease. Thyroid autoantibodies have been implicated as an autoimmune cause of RPL.2,11 There is no routine testing recommended for ANA.8 Postulated alloimmune causes include human leukocyte antigen (HLA), cytokines, and natural killer cells.2 Currently, there is no evidence to support routine testing for these immunologic causes, but this remains controversial.7,11,12 Additionally, recent studies have found an increased prevalence of intestinal permeability in idiopathic RPL, suggesting a potential immune response leading to inflammation of the endometrium and thus pregnancy loss. If no other causes of RPL can be identified, evaluating a woman for undiagnosed intestinal permeability disorders may be an option to consider.13
Various endocrine conditions can predispose to RPL, occurring in about 8% to 12% of cases.8 Untreated hypothyroidism can lead to pregnancy loss, so thyroid function screening is recommended.1,2 ASRM recommends evaluating thyroid stimulating hormone (TSH) with no further testing if it is in the normal range.1,5 The European Society of Human Reproduction and Embryology (ESHRE) advocates for including both TSH and thyroid peroxidase (TPO antibodies) as the initial investigation, followed by thyroxine (T4) testing if TSH/TPO are abnormal.5,7 Additionally, ASRM guidelines recommend HbA1c and prolactin levels as part of the investigative workup.1 Importantly, neither well-controlled hypothyroidism or diabetes is linked to RPL.1 Hyperprolactinemia may cause dysfunction of the corpus luteum, leading to inadequate progesterone levels and pregnancy loss.2 Mid-luteal progesterone levels are commonly obtained to evaluate for luteal phase deficiency, although the reliability and reproducibility of these levels are inherently difficult.12
Thrombophilia can be either acquired or inherited.5 APS is an acquired thrombophilia (autoimmune) causing antibodies to be directed toward phospholipids, which leads to hypercoagulability.5 The antiphospholipid antibodies are lupus anticoagulant, anti-beta 2 glycoprotein antibodies, and anticardiolipin antibodies.2,8 APS is purportedly responsible for 5% to 20% of RPL.1,2,10 Women with two or more pregnancy losses may be screened for APS when parental genetic causes and maternal anatomic or hormonal abnormalities have been excluded.10 Inherited thrombophilias include factor V Leiden mutation, prothrombin G20210A gene mutation, antithrombin, protein S, and protein C deficiencies.8 ASRM and ESHRE recommend to only screen patients with a personal history of thromboembolism or family history of thrombophilia.1,5,8 Finally, MTHFR gene polymorphisms, especially C677T, have been linked to RPL.14 Of the thrombophilias, RPL secondary to acquired conditions such as APS is the primary contributor.10
Lifestyle and environmental factors are an important consideration in the workup of RPL. Smoking is a moderate risk factor for RPL and, as this negative effect appears to be dose dependent, cessation should be encouraged.7,8 Maternal obesity, or being significantly underweight, are also risk factors, and weight loss or weight gain toward a normal body mass index is important to discuss.7 Excessive alcohol consumption is a possible risk factor for pregnancy loss and should be limited or discontinued.1,7 Caffeine consumption, specifically more than 3 cups of coffee per day, has been associated with RPL.1 Additionally, there may be an association between environmental toxins (pesticides, heavy metals) and RPL, although further research is needed.7 No convincing evidence exists to associate infectious pathogens with RPL.1 It has been speculated, however, that chronic endometritis may play a role in RPL.8
Finally, a more recent topic of research has been male factors and their possible contribution to RPL. High levels of sperm DNA damage have been implicated.2 Advanced paternal age, reactive oxygen species in semen, or environmental factors may contribute to sperm DNA damage.1 Sperm DNA fragmentation testing can be considered, but it is not routinely recommended.1,7
Treatment
Treatment for RPL is not as straightforward as providers or patients might hope. Disagreement among guidelines, heterogenic studies, and differences in reporting all coalesce to effectively cloud the choice of optimal treatment.2,5 An added challenge is pressure from distressed patients to institute empiric treatment measures as they seek to avoid further losses. However, treatment should be directed to only an established cause using measures supported with evidence and/or strong consensus among experts.
Certain etiologies of RPL do have well-established and mostly uncontested treatments. A septate uterus is often amenable to hysteroscopic surgical correction. The evidence to show definitive benefit of surgical treatment for acquired uterine abnormalities is lacking, but general consensus is to consider repair if there is a significant uterine cavity defect.1 Cervical cerclages, usually placed around 12 to 14 weeks’ gestation, can improve the obstetric outcomes of those patients with a history of cervical insufficiency.3 Heparin and low-dose aspirin are recommended for the individual with APS. Heparin can be initiated with a positive pregnancy test and platelet counts should be regularly checked with this therapy to assess for thrombocytopenia. Low-dose aspirin can begin prior to conception.2,4 A dopamine agonist may be used to normalize prolactin levels for the individual with hyperprolactinemia.1,7,8 Treatment for control of diabetes and hypothyroidism can be initiated. However, given that RPL is often idiopathic and causality cannot be demonstrated, many times there is no proven treatment.2,9
In vitro fertilization (IVF) and preimplantation genetic testing (PGT) may be an option for couples with RPL related to recurrent aneuploidy.3 Although IVF/PGT may shorten time to pregnancy and reduce pregnancy loss, its cost efficacy has been the subject of much debate.3
Many popular empiric treatment modalities for URPL such as low-dose aspirin, progesterone supplementation, glucocorticoids, or intralipid therapy have no evidentiary basis for use and are not recommended.7 It could be argued that they do not cause harm and can be instituted empirically, but caution must be exercised because this does not always hold true. Antithrombotics do not increase live birth rate for URPL and should not be given to women without thrombophilias.3,7,10 Currently, no benefit of various types of immunotherapy has been confirmed.1,8
Counseling/education
Counseling and education should start at the beginning of the assessment process and continue throughout further investigation and any treatment. The realistic expectations for the yield of any RPL investigative workup should be reviewed carefully. This includes discussing with the couple that 50% of cases may remain entirely unexplained even after putting in exhaustive effort, financial resources, and physical strain.1
Evidence-based treatment is not available in many cases of RPL and this factor should be shared with the couple.9 It should be noted, however, that couples still place importance on having a plan in place to prevent future pregnancy loss.9 Overall, the long-term prognosis is very good, with 50% to 60% of couples having a successful pregnancy in the future.1,4 The discussion of prognosis should take into consideration the number of preceding pregnancy losses as well as maternal age.7,9
Ongoing reassurance and emotional support have been shown to have a positive influence on patients with RPL.1 This is particularly important when considering the high likelihood of a successful outcome with empathy and supportive care alone.12
Psychological counseling and emotional support referral and resources should be offered to all couples with RPL due to its high emotional toll.1,15 Couples desire acknowledgment of their losses, along with sensitivity, and empathy. When available, a referral to a counselor who specializes in reproductive grief or loss may be especially helpful. Participation in RPL support groups may also be helpful. The organization Share: Pregnancy and Infant Loss Support can be contacted at nationalshare.org.A
Conclusion
RPL is devastating to reproductive-age couples and a significant source of distress and anxiety. Additionally, RPL can be an intimidating entity for healthcare providers to evaluate and treat due to varying guidelines and pressure from patients to prevent future losses. Because of their specialized training and education, WHNPs are in a unique position to educate patients on the potential causes of RPL, initiate diagnostic testing, and refer to specialized settings. Additionally, WHNPs are able to offer emotional support and resources to couples that are experiencing RPL.
Hayley E. Rapp is a women’s health nurse practitioner at Reproductive Medicine Associates of New Jersey in Marlton, New Jersey. The author has no actual or potential conflicts of interest in relation to the contents of this article.
References
Practice Committee of the American Society for Reproductive Medicine. Evaluation and treatment of recurrent pregnancy loss: a committee opinion. Fertil Steril. 2012;98(5):1103-1111.
Sultana S, Nallari P, Ananthapur V. Recurrent pregnancy loss (RPL): an overview. J Womens Health Dev. 2020;3(3):302-315.
Mehta S, Gupta B, eds. Recurrent Pregnancy Loss. Gateway East, Singapore: Springer Nature; 2018.
El Hachem H, Crepaux V, May-Panloup P, et al. Recurrent pregnancy loss: current perspectives. Int J Womens Health. 2017;9:331-345.
Youssef A, Vermeulen N, Lashley EELO, et al. Comparison and appraisal of (inter)national recurrent pregnancy loss guidelines. Reprod Biomed Online. 2019;39(3):497-503.
Practice Committee of the American Society for Reproductive Medicine. Definitions of infertility and recurrent pregnancy loss: a committee opinion. Fertil Steril. 2020;113(3):533-535.
ESHRE Guideline Group on RPL; Atik RB, Christiansen OB, Elson J, et al. ESHRE guideline: recurrent pregnancy loss. Hum Reprod Open. 2018;2:1-12.
Brezina PR, Kutteh WH. Recurrent early pregnancy loss. In: Falcone T, Hurd WW, eds. Clinical Reproductive Medicine and Surgery: A Practical Guide, 3rd ed. Cham, Switzerland: Springer International; 2017:269-288.
van Dijk MM, Kolte AM, Limpens J, et al. Recurrent pregnancy loss: diagnostic workup after two or three pregnancy losses? A systematic review of the literature and meta-analysis. Hum Reprod Update. 2020;26(3):356-367.
Alecsandru D, Klimczak A, Garcia Velasco JA, et al. Immunologic causes and thrombophilia in recurrent pregnancy loss. Fertil Steril. 2021;115:561-566.
D’Ippolito S, Ticconi C, Tersigni C, et al. The pathogenic role of autoantibodies in recurrent pregnancy loss. Am J Reprod Immunol. 2020;83(1):e13200.
Malik R. Evidence based management in recurrent pregnancy loss. In: Mehta S, Gupta B, eds. Recurrent Pregnancy Loss. Gateway East, Singapore: Springer Nature; 2018:181-194.
Tersigni C, D’Ippolito S, Di Nicuolo F, et al. Recurrent pregnancy loss is associated to leaky gut: a novel pathogenic model of endometrium inflammation? J Transl Med. 2018;16(1):102.
Du B, Shi X, Yin C, Feng X. Polymorphisms of methalenetetrahydrofolate reductase in recurrent pregnancy loss: an overview of systematic reviews and meta-analyses. J Assist Reprod Genet. 2019;36(7):1315-1328.
Koert E, Malling GMH, Sylvest R, et al. Recurrent pregnancy loss: couples’ perspectives on their need for treatment, support and follow up. Hum Reprod. 2019;34(2):291-296.

