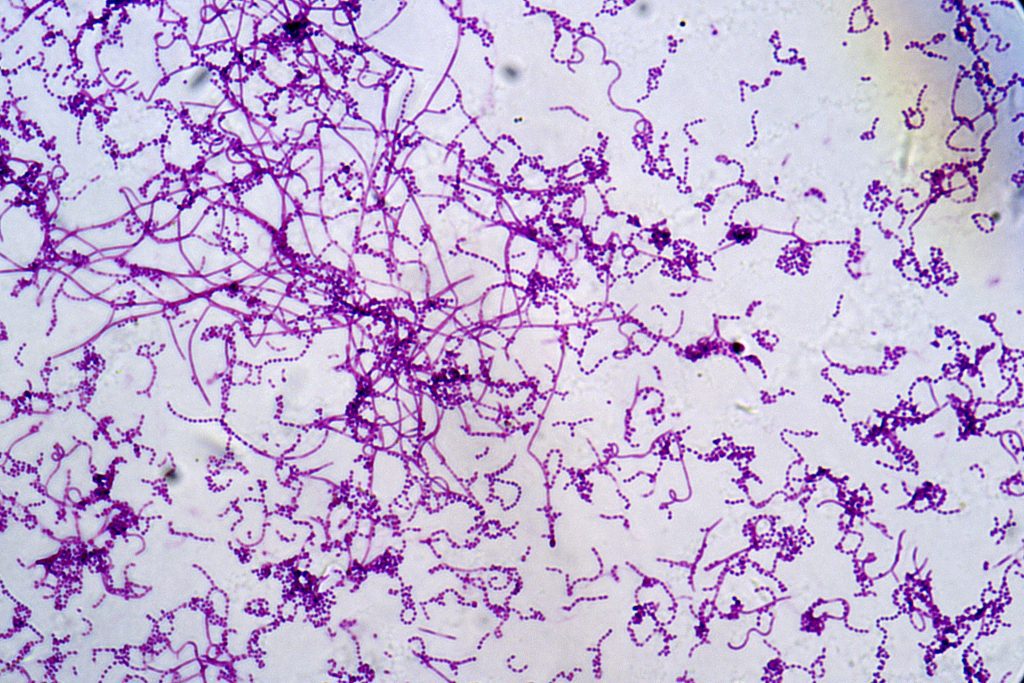
EC, a 54-year-old postmenopausal female presents as an established patient at the women’s health clinic. She informs the women’s health nurse practitioner (WHNP) that she has returned to the clinic because she continues to experience malodorous vaginal discharge and itching despite treatment for bacterial vaginosis (BV) 2 months ago. This diagnosis was made using a rapid DNA probe test. EC confirms completion of the prescribed oral metronidazole and states “the medication you gave me did not work.” EC adds “I believe the change in my vaginal pH is due to recent antibiotic medication therapy for ingrown toe-nails.” She is sexually active and denies vaginal bleeding, pelvic pain, urinary symptoms, and dyspareunia. She has a 25-year history of medroxyprogesterone acetate injection use for birth control, of which she reports stopping 3 years ago. EC denies any previous history of vaginal infections or sexually transmitted illnesses. She reports having had two uncomplicated pregnancies resulting in uneventful vaginal deliveries. Other health history is notable for hypertension, chronic low back pain, anxiety, and depression. She is on several medications for management of these conditions. Preventive health measures including breast, colorectal, and cervical cancer screenings are all up to date with normal results.
Physical examination and laboratory tests
EC is afebrile. An abdominal examination reveals active bowel sounds, and there are no notable masses or tenderness during abdominal palpation. EC has a normal appearing external genitalia without the presence of blood or lesions. The vaginal vault is without blood or lesions and has a presence of thick white discharge. The vaginal walls are pale pink with minimal rugated areas. The cervix contains a copious amount of thick white discharge. There is no cervical motion tenderness or malodor detected on clinical examination. The bimanual pelvic examination reveals an anterior, nontender, and mobile uterus with no masses or enlargement. The ovaries are nonpalpable.
Initial laboratory findings revealed mild anemia with a hemoglobin level of 11.6 g/dL and hematocrit 36.4%, normal white blood cell count of 7.6 K/cm, and an elevation of C-reactive protein to 7.21 mg/L (normal 0–3.0) and erythrocyte sedimentation rate (ESR). Urinalysis revealed hazy clarity, trace amount of ketones, and few bacteria. The urine culture was negative for any growth. A repeat DNA probe test revealed negative Candida species, C. glabrata, BV, and trichomonas. A culture for urea plasma and mycoplasma was negative. An elution swab of the vaginal discharge was performed to identify the presence of anerobic or aerobic pathogenic organisms. This culture is appropriate when determining the cause of malodorous vaginal discharge when common tests are negative. The results revealed a heavy growth of Actinomyces odontolyticus.
Actinomycosis
Actinomycosis is an opportunistic subacute to chronic bacterial infection, suppurative and granulomatous inflammation in nature, caused by the bacteria of the genus Actinomyces.1 There are over 30 species, with most having low pathogenicity and considered normal flora in the mouth, gastrointestinal tract, and female genital tract. Up to 60% to 70% of infections are attributed to A. israelii or A. gerencseriae.2 The incidence of actinomycosis is underestimated, with the diagnosis often difficult to distinguish. The most recent data report nearly 1 in 300,000 individuals are affected annually. Worldwide disease distribution includes an age range of 18 to 58 years, with an increased prevalence in the middle-aged in the United States.3 Additionally, men are more commonly affected than women. Actinomycosis, an endogenous infection, has no risk of person-to-person transmission. Finally, actinomycosis infection has no racial preference.
Risk factors for actinomycosis include diabetes, cancer, temporary immunosuppressive therapy, end-stage renal disease, chronic alcohol abuse, previous surgical history, implanted foreign bodies, aspiration, intrauterine device (IUD) use, intentional or accidental tissue trauma, having an organ transplant, and being immunocompromised with poor oral hygiene.4 The disease manifestation depends on the original site of invasion. Several anatomic sites that have been identified include cervicofacial (lumpy jaw), abdominal, pulmonary, genitourinary, central nervous system, cutaneous, and extra facial and joint.4 Clinical features of genitourinary or pelvic actinomycosis may include unintentional weight loss, lower abdominal pain or discomfort, pelvic pain, abnormal vaginal bleeding, or vaginal discharge.3,5 Although a benign condition, pelvic actinomycosis erodes the damaged or weakened epithelium allowing the invasion of local structures thus mimicking malignancy and other pelvic inflammatory diseases.6 When the mucosal barrier is compromised without regard to anatomic structures, actinomycosis can spread and cause an infection in surrounding tissues.7 In nearly all published clinical cases, the presence of an IUD has been reported. It is hypothesized that IUDs can precipitate actinomycosis infections by injuring the uterine mucosal lining and altering the carbohydrate metabolism of the endometrial cells.8
Further evaluation
Timely recognition is imperative to avoid unnecessary surgical procedures and minimize associated mortality and morbidity. Diagnosis of pelvic actinomycosis is inevitably challenging and requires the WHNP to be highly suspicious and willing to further investigate potential causes of unresolved symptoms such as vaginal discharge.
Investigators report blood tests are nonspecific, with normochromic anemia and mild leukocytosis with predominance of polymorphonuclear cells as notable clinical laboratory findings.4 This patient’s laboratory tests mirrored these listed results and included the other nonspecific laboratory findings of elevation of C-reactive protein and ESR, which signify an inflammatory illness state. Researchers state that organism identification from a sterile specimen is an option but represents a known source of increased failure rates due to inhibition of actinomycosis growth by contaminant/coexistent bacterium. The earliest culture growth identification may take up to a minimum of 5 and a maximum of 20 days.4
EC had a pelvic ultrasound 4 months earlier as a recommendation to follow up on a complex ovarian cyst revealing echo-texture and heterogeneous uterus likely due to intramural fibroids or less likely adenomyosis. The right ovary was not visualized, and the left ovary appeared within normal limits. Magnetic resonance imaging (MRI) of the pelvis was ordered because it aids in differentiating malignancy from acute inflammatory pathologies such as actinomycosis. Furthermore, MRIs provide the characteristics and extent of actinomycosis. The actinomycosis lesion is generally ill-defined, whereas neoplastic lesions are usually well-defined on radiologic imaging.9 The MRI report included the notation that sensitivity and specificity were decreased secondary to the lack of intravenous contrast that was not utilized due to history of mild contrast allergy. The endometrium appeared somewhat thickened, and subendometrial cysts were present favoring adenomyosis. No definite fibroids or endometriomas were identified. The right ovary was not discretely visualized, and the left ovary appeared normal. A non-contrast enhanced computed tomography (CT) scan was recommended by the radiologist but delayed for 3 months due to the patient taking an extended vacation.
Plan and implementation of care
In the case of EC, an infectious disease (ID) specialist and pharmacist were consulted after the laboratory results revealed a heavy growth of Actinomyces odontolyticus. The ID specialist and pharmacist both recommended oral antibiotic therapy as the diagnostic images did not disclose a widespread actinomycosis infection. Antibiotic therapy is the cornerstone of actinomycosis treatment, but surgical intervention designed to remove the infected tissue may be warranted in some cases. The duration and type of administration of antibiotic therapy will depend on the initial response to treatment as well as the burden of the infection. EC agreed with this plan of care and was glad to not have an extended intravenous antibiotic course that would interfere with her travel plans. She confirmed completion of the prescribed medication. The noncontrast enhanced CT was done on return from her trip and revealed limited evaluation of the uterus and adnexa. The radiologist recommended a follow-up abdominal ultrasound for further evaluation.
Desired outcome
In the case of pelvic actinomycosis when an IUD is in place, the recommendation is to remove the device after initiating antibiotics. There are limited studies to provide evidence of the best strategies to adequately treat pelvic actinomycosis not associated with IUD use. Thus, it is best managed by a multidisciplinary care team that in this case include the WHNP as the primary care provider, an ID specialist, pharmacist, microbiologist, and radiologist. The prognosis for the patient without extensive or complicated disease is excellent with appropriate antibiotic therapy.2 Long-term intravenous antibiotic therapy in conjunction with surgical intervention may be warranted depending on imaging findings, severity of symptoms, and lack of improvement with oral antibiotic treatment. In evaluating EC’s care, repeat diagnostic imaging and laboratory testing will be required to determine the resolution of the actinomycosis infection.
Implications for women’s healthcare
Pelvic actinomycosis is considered a diagnostic challenge due to its inconspicuous and prolonged clinical course involving non-specific symptoms. Although pelvic actinomycosis infections are most often attributed to IUD use, EC demonstrated a unique clinical presentation. Her case presents the notion for the invasive disease to arise from other avenues as the etiology of pelvic actinomycosis is multifactorial. The WHNP is well-positioned to provide a comprehensive approach in investigating challenging cases such as that with EC. Their expertise in women’s health, history taking, physical examination, and diagnostic reasoning provide the foundation to move the process forward and to initiate appropriate consultations and referrals. The WHNP also brings an individualized, person-centered approach that promotes shared decision making and ensuring patients have an active role in their healthcare.
Ludrena C. Rodriguez is a women’s health nurse practitioner at the Veteran’s Health Administration in Fayetteville, North Carolina. The author has no actual or potential conflicts of interest in relation to the contents of this article.
Womens Healthcare. 2023;11(4):42-44,48. doi: 10.51256/WHC082342
References
1 Shin D, Hwang J, Hong SS, et al. Pelvic actinomycosis mimicking malignancy of the uterus: a case report. Investig Magn Reson Imaging. 2019;23(2):136-141. https://doi.org.10.13104/imri.2019.23.2.136.
2 Sharma S, Hashmi MF, Valentino III DJ. Actinomycosis. In: StatPearls [Internet]. Treasure Island, FL: StatPearls Publishing; Jan 2022.
3 Garcia-Garcia A, Ramirez-Duran N, Sandoval-Trujillo H, Romero- Figueroa MDS. Pelvic actinomycosis. Can J Infect Dis Med Microbiol. 2017;2017:9428650.
4 Cantres-Fonseca OJ, Vando-Rivera V, Fonseca-Ferrer V, et al. Actinomycosis: diagnosis, clinical features and treatment. In: Hozzein WN, ed. Actinobacteria. 2022. https://www.intechopen.com/chapters/82972.
5 Okulicz JF. Actinomycosis. Medscape. Updated November 3, 2022. https://emedicine.medscape.com/article/211587-overview.
6 Floyd R, Hunter S, Abu Saadeh F, et al. Pelvic actinomycosis. QJM. 2021;114(8):587-588.
7 Hyser E, Antala D, Friedman H, Stake J. Abdominopelvic actinomycosis: an unexpected diagnosis in an elderly female with a destructive-appearing soft tissue mass. IDCases. 2022;28:e01479.
8 Mirza E, Jaleel S. Pelvic actinomycosis presenting as a single episode of postmenopausal bleeding in a 60-year-old woman whose intrauterine contraceptive device had been removed 10 years previously: a case report. Case Rep Womens Health. 2020;28:e00267.
9 Ahmed S, Ali M, Adegbite N, et al. Actinomycosis of tongue: rare presentation mimicking malignancy with literature review and imaging features. Radiol Case Rep. 2018;14(2):190-194.