The American College of Obstetricians and Gynecologists (ACOG) published a practice bulletin on gestational diabetes mellitus (GDM) in February 2018,1 just 7 months after publishing a previous bulletin on the same topic.2 According to ACOG, the purpose of the update was to provide additional information on the pharmacologic treatment of gestational diabetes mellitus. This article elucidates the recent changes in ACOG’s bulletin, as well as those in the American Diabetes Association guidelines.3
Gestational diabetes mellitus (GDM) is one of the most common complications of pregnancy, affecting 7%-9% of pregnancies overall, with prevalence rates as high as 25% in at-risk racial/ethnic groups (African Americans, Latinas, Native Americans, Asian Americans, and Pacific Islanders).1,4 Additional risk factors for GDM include advanced maternal age, overweight/obesity, having a first-degree relative with diabetes mellitus (DM), minimal physical activity, hypertension, cardiovascular disease, and any clinical condition associated with insulin resistance (IR). Having previous GDM confers a greater risk of developing GDM in subsequent pregnancies.
For a pregnant woman, GDM confers an increased risk for preeclampsia and operative vaginal and cesarean births, along with an increased risk for complications associated with these births (e.g., infection, hemorrhage, anesthesia risks). Poorly controlled plasma glucose (PG) levels can lead to polyhydramnios, or excessive amniotic fluid, which can make a woman feel greater discomfort during the latter weeks of pregnancy. GDM can also heighten a woman’s emotional stress because of the added dietary restrictions, the physical activity prescriptions, and the increased concern about the fetus’s/infant’s well-being.1-3 Receiving a diagnosis of GDM can be particularly distressing for a pregnant woman who has always followed a healthful diet and engaged in regular physical activity (Box 1).5

A fetus affected by uncontrolled GDM is at increased risk for perinatal complications, including death, shoulder dystocia, bone fracture, and nerve palsies.6 Macrosomia, or excessive fetal growth, occurs primarily because of elevated circulating levels of insulin.7 If a woman has elevated levels of PG during labor, the neonate is at markedly increased risk for hypoglycemia (the increased glucose levels in utero transfer readily to the fetus, stimulating the fetal pancreas to produce more insulin; when the source of glucose—that is, the placenta—is eliminated at the time of birth, persistently elevated insulin levels can cause neonatal glucose levels to drop precipitously).8 In addition, infants of mothers with diabetes can experience delayed surfactant production, which can lead to respiratory distress syndrome.9
How can healthcare providers (HCPs) best care for women with GDM? In this article, the author incorporates the most recent professional association guidelines with evidence-based best practices—covering prevention, screening/diagnosis, management, and practice implications—to help these pregnant patients beat the sugar blues.
Prevention
Healthcare providers caring for women who wish to become pregnant but who have risk factors for GDM can provide preconception counseling to reduce this risk. HCPs should encourage women who are overweight (body mass index [BMI] ≥25) or obese (BMI ≥30) to lose weight before becoming pregnant and to engage in regular physical activity. HCPs should also manage existing health conditions associated with IR. Once at-risk women become pregnant, they can follow the Institute of Medicine’s (IOM’s) guidelines for recommended weight gain during pregnancyA, based on their pre-pregnant BMI, to reduce their GDM risk.10
Screening and diagnosis
Early screening
All pregnant women identified as being at high risk for diabetes, based on aforementioned risk factors, should be screened at the first prenatal visit, ideally before 13 weeks’ gestation. The goal of early screening is to detect pre-gestational diabetes—that is, diabetes that was present, but not diagnosed, before a woman’s pregnancy. Prior to publication of the 2017 American Diabetes Association (ADA) guidelines,11 any form of glucose intolerance first diagnosed during pregnancy was labeled gestational. The newer guidelines acknowledge that diagnosis before the late second trimester or the early third trimester of pregnancy is not truly gestational. During the first half of pregnancy, women are diagnosed with pre-gestational diabetes if they meet any of these standard criteria for DM diagnosis3:
• fasting PG (FPG) ≥126 mg/dL (7.0 mmol/L); or
• hemoglobin A1C (HbA1C) ≥6.5% using a standardized assay (according to the ADA, this test, when used alone, may have decreased sensitivity when compared with oral glucose tolerance test [OGTT] approaches); or
• random PG ≥200 mg/dL (≥11.1 mmol/L), subsequently confirmed by elevated FPG or HbA1C; or
• 2-hour PG ≥200 mg/dL following a 75-g oral glucose load (of note, this diagnostic level differs from that used at 24-28 weeks to diagnose GDM).
The ADA recommends that, for any of these measures, in the absence of unequivocal hyperglycemia, results should be confirmed by repeat testing.11
As a point of clarification, HCPs should note that laboratory testing relies on plasma glucose measurements derived from collected venous blood samples. Point-of-care meters, including home meters, rely on measurements of blood glucose (BG) from droplets of capillary whole blood. Continuous glucose monitoring systems rely on sensors that measure glucose in interstitial fluid, not blood. Glucose measurements from these different sources vary, with venous PG values typically being around 15% higher than capillary BG values.
Routine screening
All pregnant women should be screened for GDM at 24-28 weeks. According to the ADA, two screening methods are acceptable3:
Two-step method
Because of the endorsement of the American College of Obstetricians and Gynecologists (ACOG), the two-step method is used by 95% of obstetricians in the United States.1 This approach entails having women drink a 50-g oral glucose solution, followed by a 1-hour venous PG measurement. With this 1-hour OGTT, fasting beforehand is not required. Women whose PG levels meet or exceed an institution’s screening threshold return to the office or clinic for a 100-g, 3-hour diagnostic OGTT; for this step, fasting beforehand is required.
Therefore, screening and diagnosis occur in two separate steps. The 1-hour OGTT is considered positive at PG levels exceeding 130-140 mg/dL. The higher the threshold PG level is set, the lower the sensitivity but the higher the specificity and the lesser the likelihood of a false-positive result. ACOG recently affirmed that either value can be used as a cutoff for an abnormal initial screening result, but also that each practice should decide on a consistent value to be used that is based on DM prevalence within its community.1 Communities with a higher prevalence of DM may decide to use a lower cutoff value in order to avoid false-negative results.
For many years, a positive diagnosis of GDM has required that two or more threshold PG levels on the 3-hour OGTT be met or exceeded. However, for the first time, ACOG, noting that research evidence has demonstrated an increased risk for adverse perinatal outcomes with even one abnormal value,2 has stated that one elevated value may be used to establish the diagnosis of GDM. In the 2018 Practice Bulletin, an additional statement was added: Further research is needed to clarify the risk of adverse outcomes in patients with one abnormal value on the 100-g, 3-hour OGTT and whether they would benefit from treatment.1 Once again, as with the screening cutoff decision, each practice should choose to use either one abnormal value or two abnormal values consistently as the basis for a GDM diagnosis, taking into consideration the prevalence of GDM in its community. Some communities with a high prevalence of GDM have been using one abnormal value as a cause for concern all along, using a term such as impaired glucose tolerance instead of GDM.
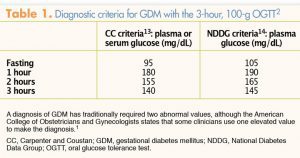
The Fifth International Workshop Conference on GDM12 has recommended using Carpenter and Coustan criteria for the 3-hour OGTT13 as opposed to National Diabetes Data Group criteria,14 although ACOG states that either set of criteria may be used (Table 1). In addition, ACOG has stated that individual practices may choose to use the one-step method, which combines screening and diagnosis into one test, instead of the two-step method.1,2
 One-step method
One-step method
ACOG recommends the two-step method primarily because it was endorsed by consensus of an expert panel at the National Institutes of Health.15 However, this consensus panel had some limitations, including the fact that the meeting was terminated a day early because of weather conditions, suggesting that the final decision reached by the panel may have differed with further discussion. The one-step method, endorsed by the World Health Organization, 16 is the method used throughout most of the rest of the world. A major advantage of this method is that women undergo PG testing in just one step, thereby eliminating the inconvenience and delay in diagnosis entailed with the two-step method. A disadvantage of one-step testing is that women must fast beforehand. After blood for an FPG measurement is drawn, a 75-g oral glucose load is administered and PG levels are evaluated after 1 and 2 hours. GDM diagnostic criteria for the 75-g 2-hour OGTT are as follows:
• FPG: ≥92 mg/dL (≥5.1 mmol/L)
• 1-hour PG: ≥180 mg/dL (≥10.0 mmol/L)
• 2-hour PG: ≥153 mg/dL (≥8.5 mmol/L)
Only one abnormal value is required for a diagnosis of GDM with the 75-g, 2-hour OGTT.3
Management of GDM with lifestyle intervention
In most cases, GDM management starts with lifestyle interventions; basic components include healthful eating, physical activity, and self-monitoring of BG levels.
Healthful eating
Even though diet therapy is the cornerstone of GDM treatment, clinical trials to date have not established the optimal diet for GDM.17,18 Because carbohydrates are the  macronutrients that appear to have the greatest effect on postprandial BG levels, women are typically advised to monitor their intake of carbohydrates more closely than that of other macronutrients and to follow a diet composed of 33%-40% complex carbohydrates (avoidance of simple sugars), 20% proteins, and 40% fats.
macronutrients that appear to have the greatest effect on postprandial BG levels, women are typically advised to monitor their intake of carbohydrates more closely than that of other macronutrients and to follow a diet composed of 33%-40% complex carbohydrates (avoidance of simple sugars), 20% proteins, and 40% fats.
To prevent large BG fluctuations, the ADA advises consuming three meals a day, with two to three snacks.3 Moderate use of artificial sweeteners such as aspartame is permitted. BG levels normalize in 70%-85% of women with GDM who follow these dietary guidelines.3 An important part of nutrition therapy is an evening snack consisting of 15-30 g of carbohydrates. The overnight fast should not exceed 10 hours. Otherwise, BG levels could drop low enough to stimulate secretion of additional counterregulatory hormones (e.g., epinephrine, cortisol), resulting in increased BG levels. The dietary reference intake (DRI) for carbohydrates in pregnancy is 175 g/day, which ensures adequate glucose intake to meet fetal and maternal needs. DRIs are 71 g/day for protein and 28 g/day for fiber.3
Physical activity
Women with GDM should perform 30 minutes of moderate-intensity aerobic exercise at least 5 days/week.1 Although evidence is inconclusive, daily activity such as walking or riding a stationary bike should help normalize and stabilize BG levels; in some cases, engaging in a daily routine of physical activity may even eliminate a need for medication.19-21
Monitoring blood glucose
In general, women with GDM should test their fasting BG (FBG) daily, as well as check BG 1 or 2 hours after each meal (either checkpoint is acceptable; no evidence indicates that one is clearly superior to the other).1 If women are achieving target BG levels consistently after 1 week of dietary treatment, the ADA states that they can perform BG monitoring on an every-other-day basis rather than daily.3 When reviewing patients’ BG logs, most HCPs want to see that at least 80% of the BG values are normal. If this goal is not achievable through lifestyle modification alone, then medication should be initiated to achieve these targets:
• FBG: <95 mg/dL
• 1-hour postprandial BG: <140 mg/dL
• 2-hour postprandial BG: <120 mg/dL
Medication initiation
In its 2018 Practice Bulletin, ACOG affirms the use of insulin as the preferred pharmacotherapy for GDM when medication is necessary.1 Likewise, in its 2018 guidelines, the ADA recommends insulin as first-line pharmacotherapy for this indication.3 Therefore, only insulin is discussed in this article. The strong recommendation for insulin is due, in large part, to lack of knowledge about long-term effects of intrauterine exposure of the fetus to oral antidiabetic medications. Both glyburide and metformin, two commonly used oral medications for DM, cross the placenta.22,23
ACOG suggests a starting insulin dosage of 0.7-1.0 units/kg/day for women with GDM,1 although many HCPs start with half this amount in order to gauge patient response before titrating upward. Multiple injections with a combination of long- or intermediate-acting insulins are used in conjunction with short-acting insulin for meal boluses (for women who have high postprandial BG excursions).1 Whenever insulin is used, the potential for overdosing and precipitating hypoglycemia exists. In pregnant women, this risk is of particular concern because many women have relatively low FBG levels (<70-80 mg/dL) during pregnancy. If a woman is not taking medication with a potential to decrease BG, she is unlikely to experience hypoglycemia because her secretion of counterregulatory hormones will surge and normalize her BG levels. However, if a woman is taking insulin to lower BG, too much medication can override her capacity to counterregulate.
For that reason, all pregnant insulin users should know the signs/symptoms of hypoglycemia (e.g., shakiness, sweating, irritability, clammy skin) and carry 15-30 g of rapidly absorbed glucose with them at all times to correct episodes of low BG. In general, 15 g is given initially with symptom onset; if BG does not normalize within 15 minutes, another dose of a rapidly absorbed carbohydrate (e.g., skim milk, glucose tablets) is given. If a woman is unconscious, she should not receive anything by mouth. Every pregnant woman who uses insulin should have an emergency glucagon kit for treatment of low BG in the event of unconsciousness. With the accuracy of current home BG monitoring systems, loss of consciousness is rarer now than in the past.
Case study
Sue, a 41-year-old G3 P2002, presents for a routine prenatal visit at 33 weeks 4 days’ gestation. She has had two term vaginal births 17 years and 10 years previously, with the first infant, a boy, weighing 7 lb 10 oz and the second, a girl, weighing 8 lb. Her first two pregnancies and births were uneventful. Sue is 5 feet 5 inches tall; her prepregnancy weight was 234 lb (BMI, 38.3). Because of her age and BMI category (class II obesity), Sue had undergone early DM screening. Her HbA1C result, 5.2%, indicated that she did not have overt diabetes. However, because of her class II obesity, the nurse practitioner (NP) advised Sue to gain only 11-20 lb during this pregnancy. As of the current visit, she has gained 15 lb overall.
GDM diagnosis
At 28 weeks’ gestation, Sue was diagnosed with GDMA1 (i.e., GDM controlled with dietary modification). Sue states that she has been following a 2,500 calorie/day diet,
with carbohydrates composing 33%-40% of total calories, as prescribed by the dietician. Other than avoiding consumption of pork products, Sue has no dietary restrictions. She
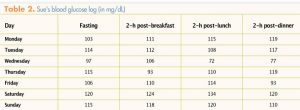
reports that she has been walking in her neighborhood for 20 minutes on most days. She provides her BG log for the past week for the NP to review (Table 2). The NP notes that although most of Sue’s postprandial BG values are normal, her FBG levels are all elevated. Prior to this week’s log, more than 90% of Sue’s BG values, both fasting and postprandial, were normal.
Management
The NP asks Sue whether she has been having a snack in the evening. Sue replies that, as per her dietician’s instruction, she has been eating cheese and crackers at bedtime.
Based on a consultation with the collaborating physician, as well as the fact that only Sue’s FBG levels have been elevated for the past week, the NP decides to start Sue on NPH insulin 12 units at bedtime. Sue is referred to a certified diabetes educator for instruction on how to administer subcutaneous insulin, recognize hypoglycemia, and treat hypoglycemia if it occurs. Sue asks the NP why she needs insulin now. The NP explains that Sue’s lifestyle modifications alone are not able to compensate for her increased IR and that elevated FBG does not usually respond to further dietary changes. Sue also asks whether she needs to undergo any additional testing beyond the twice-weekly fetal non-stress tests and amniotic fluid index determinations. The NP informs her that she is already getting the testing required for GDMA2 (i.e., GDM requiring medication therapy) as it is the same testing that she already undergoes because of her advanced maternal age.
Outcome
After starting NPH insulin, Sue’s FBG levels normalize. She goes on to have a normal spontaneous labor and give birth vaginally to an 8 lb 2 oz girl at 39 weeks’ gestation. Sue is placed on an intravenous insulin drip during labor to keep her BG under tight control to avert neonatal hypoglycemia. Following delivery, Sue has normal BG values without medication. She is able to breastfeed without difficulty. The NP making postpartum rounds advises Sue that she should choose a method of birth control, preferably before discharge, and undergo a 75-g OGTT at 4-12 weeks postpartum.
The 75-g OGTT is especially important to make sure that Sue does not have persistent glucose intolerance (diabetes or prediabetes). The OGTT is more sensitive than an HbA1C test because of increased red blood cell turnover in pregnancy and birth-related blood loss.3 If the test result is negative, she should then undergo testing for DM every 1-3 years afterwards; 15%-70% of women with GDM go on to develop overt DM—most often type 2—in their lifetime.1 It is especially important to screen women periodically while they are of childbearing age; achieving optimal glucose control before conception in women with overt DM is essential to reduce the increased risk for congenital anomalies.3 For this same reason, use of an effective birth control method is vital until a woman is ready to conceive another child. Nonhormonal intrauterine contraception is especially effective and does not appear to affect glucose metabolism, as may occur with hormonal methods in some women.
Case Implications for healthcare providers
This synthesis of current diagnosis and treatment guidelines can aid HCPs in caring for women with GDM. Besides the usual recommendations for anyone at risk for DM—healthful eating and increased physical activity—another important lifestyle recommendation pertains specifically to mother and infant during the postpartum period: breastfeeding. The beneficial metabolic effects of breastfeeding persist beyond weaning; they not only decrease the risk of progression to type 2 DM for the woman but also decrease the risk of development of DM in her offspring, thereby conferring health benefits that last well into the future.24 Box 2 lists important points for readers.

Kimberly K. Trout is Assistant Professor of Women’s Health in the Department of Family and Community Health at the University of Pennsylvania School of Nursing in Philadelphia. The author states that she does not have a financial interest in or other relationship with any commercial product named in this article.
References
1. American College of Obstetricians and Gynecologists. Practice Bulletin No. 190: Gestational Diabetes Mellitus. Washington, DC: Author; 2018
2. American College of Obstetricians and Gynecologists. Practice Bulletin No. 180: Gestational Diabetes Mellitus. Washington, DC: Author; 2017.
3. American Diabetes Association. Standards of medical care in diabetes—2018. Diabetes Care. 2018;41(suppl 1):S1-S143.
4. Correa A, Bardenheier B, Elixhauser A, et al. Trends in prevalence of diabetes among delivery hospitalizations, United States, 1993-2009. Matern Child Health J. 2015;19(3):635-642.
5. Muse Q. Meeting the dietician. My GDM experience. PENN Medicine in the News Blog. October 2, 2017.
6. Crowther CA, Hiller JE, Moss JR, et al; Australian Carbohydrate Intolerance Study in Pregnant Women (ACHOIS) Trial Group. Effect of treatment of gestational diabetes mellitus on pregnancy outcomes. N Engl J Med. 2005;352(24):2477-2486.
7. American College of Obstetricians and Gynecologists. Practice Bulletin No. 22: Fetal Macrosomia. Washington, DC: Author; 2000.
8. Yehuda I, Nagtalon-Ramos J, Trout K. Fetal growth scans and amniotic fluid assessments in pregestational and gestational diabetes. J Obstet Gynecol Neonatal Nurs. 2011;40(5):603-614.
9. Rubarth LB. Infants of diabetic mothers. Neonatal Netw. 2013;32(6):416-418.
10. Trout KK, Ellis KK, Bratschie A. Prevention of obesity and diabetes in childbearing women. J Midwifery Womens Health. 2013;58(3):297-302.
11. American Diabetes Association. Standards of medical care in diabetes—2017. Diabetes Care. 2017;40(suppl 1):S1-S132.
12. Metzger BE, Buchanan TA, Coustan DR, et al. Summary and recommendations of the Fifth International Workshop-Conference on Gestational Diabetes Mellitus. Diabetes Care. 2007;30(suppl 2):S251-S260.
13. Carpenter MW, Coustan DR. Criteria for screening tests for gestational diabetes. Am J Obstet Gynecol. 1982;144(7):768-773.
14. National Diabetes Data Group Classification. and diagnosis of diabetes mellitus and other categories of glucose intolerance. Diabetes. 1979;28:1039-1057.
15. National Institutes of Health consensus development conference statement: diagnosing gestational diabetes mellitus, March 4-6, 2013. Obstet Gynecol. 2013;122(2 pt 1):358-369.
16. World Health Organization (WHO). Diagnostic Criteria and Classification of Hyperglycaemia First Detected in Pregnancy. Geneva, Switzerland: Author; 2013.
17. Hernandez TL, Van Pelt RE, Anderson MA, et al. A higher-complex carbohydrate diet in gestational diabetes mellitus achieves glucose targets and lowers postprandial lipids: a randomized crossover study. Diabetes Care. 2014;37(5):1254-1262.
18. Trout KK, Homko CJ, Wetzel-Effinger L, et al. Macronutrient composition or social determinants? Impact on infant outcomes with gestational diabetes mellitus. Diabetes Spectr. 2016;29(2):71-78.
19. Brankston GN, Mitchell BF, Ryan EA, Okun NB. Resistance exercise decreases the need for insulin in overweight women with gestational diabetes mellitus. Am J Obstet Gynecol. 2004;190(1):188-193.
20. Halse RE, Wallman KE, Newnham JP, Guelfi KJ. Home-based exercise training improves capillary glucose profile in women with gestational diabetes. Med Sci Sports Exerc. 2014;46(9):1702-1709.
21. Anjana RM, Sudha V, Lakshmipriya N, et al. Physical activity patterns and gestational diabetes outcomes – The wings project. Diabetes Res Clin Pract. 2016;116:253-262.
22. Hebert MF, Ma X, Naraharasetti SB, et al; Obstetric-Fetal Pharmacology Research Unit Network. Are we optimizing gestational diabetes treatment with glyburide? The pharmacologic basis for better clinical practice. Clin Pharmacol Ther. 2009;85(6);607-614.
23. Vanky E, Zahlsen K, Spigset O, Carlsen SM. Placental passage of metformin in women with polycystic ovary syndrome. Fertil Steril. 2005,83(5):1575-1578.
24. Trout KK, Averbuch T, Barowski M. Promoting breastfeeding among obese women and women with gestational diabetes mellitus. Curr Diab Rep. 2011;11(1):7-12.

