Intrahepatic cholestasis of pregnancy (ICP) is a hepatic disorder that complicates 0.2% to 2% of pregnancies and poses significant risk to the fetus including preterm delivery and stillbirth. Pruritis is often the only symptom of ICP typically occurring in the second and third trimesters. Diagnosis is made when total bile acids are greater than 10 μmol/L. Ursodeoxycholic acid is first-line treatment. Antepartum surveillance is recommended when delivery would be considered in response to abnormal results. Delivery timing is guided by bile acid levels, patient presentation, and history.
Intrahepatic cholestasis of pregnancy (ICP) is a hepatic disorder that complicates 0.2% to 2% of pregnancies, with higher rates demonstrated in certain populations.1 Although ICP poses minimal health risks for the pregnant person, a diagnosis of ICP carries significant risk to the fetus including spontaneous and indicated preterm delivery, meconium-stained amniotic fluid, and stillbirth. Symptom onset typically occurs in the second and third trimesters, most presenting after 30 weeks. Cholestasis spontaneously resolves postpartum.1 Newly published literature and updated evidence-based recommendations from the Society for Maternal-Fetal Medicine (SMFM) for the assessment and management of ICP are presented to provide the reader with the most current assessment, diagnostic, treatment, and patient education recommendations.2
Pathogenesis
Cholestasis occurs when bile acid secretion is impaired leading to the accumulation and circulation of toxic bile salts. Various conditions of the liver, the bile duct, or the pancreas can impair bile acid secretion and result in cholestasis. Hormonal, genetic, and exogenous factors like advanced maternal age, in vitro fertilization, multiple gestations, and hepatitis C infection contribute to the development of ICP. During pregnancy, progressive insulin resistance, increases in progesterone and estrogen and their metabolites, and genetically influenced alterations in the bile acid pathway impact maternal lipid metabolism and liver function impairing hepatic bile acid homeostasis. In a pregnancy complicated by ICP, there is an excessive increase in bile acids in both the pregnant person and the fetus causing damage to liver cells and tissues.1,3 Although these changes resolve rapidly in the pregnant person postpartum, evidence suggests that fetuses exposed to elevated bile acids may develop metabolic syndrome later in life.4
Assessment and diagnosis
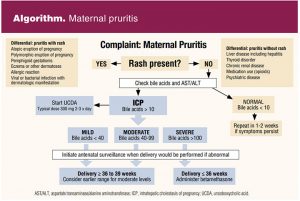
Assessment of the pregnant patient presenting with pruritis requires a detailed history and physical examination to establish the cause. Pruritis without a rash is a common complaint during pregnancy and usually has no underlying pathology. Conditions not related to pregnancy that may cause both pruritus and rash include allergies, dermatologic conditions like eczema, viral or bacterial infections with dermatologic manifestations, and use of medications like opioids. Rash is associated with the most common pregnancy-related pathologic causes of pruritis including atopic eruption of pregnancy (AEP) and polymorphic eruptions of pregnancy (PEP). Rash and pruritis associated with AEP and PEP are uncomfortable and often difficult to alleviate, but neither condition poses risk to the pregnant person or the fetus. In contrast, ICP presents as pruritis without rash and carries risk to the fetus including stillbirth.
The pruritis associated with ICP tends to be severe, worse at night, worse on the soles and palms, and typically develops in the second or third trimesters. Although there is no rash, there may be excoriation from scratching. Signs and symptoms seen with other hepatic diseases like jaundice, dark urine, abdominal pain, and colic are not typically seen with ICP.1–3 A family history or personal history of ICP should raise suspicion as this condition likely has a genetic component.
Diagnosis
Although studies have looked at various biomarkers to diagnose ICP, the measurement of serum bile acid levels and liver transaminase (alanine aminotransferase [ALT] and aspartate transaminase [AST]) levels are the most clinically useful despite limited data (SMFM grade 1B recommendation).1,5 An increase in maternal total serum bile acids combined with the symptom of pruritus and the absence of diseases associated with similar laboratory findings, supports the diagnosis of ICP. Increases in ALT and AST can also be seen but are not necessary for diagnosing ICP.
The types of assays used for bile acid level testing are typically performed in a specialty lab, and results may take several days. A total serum bile acid level of greater than 10 μmol/L is diagnostic. Interestingly, itching may manifest weeks before the liver biochemistry is altered. Repeated measurement of bile acid concentration is indicated in the presence of ongoing symptoms.1,6
New evidence suggests that levels 100 μmol/L or greater result in the highest risk for complications. Reassuringly, when bile acids are lower than 100 μmol/L, the risk of stillbirth is comparable to the general population.7
Treatment
Treatment goals include reduction in both maternal pruritis symptoms and adverse perinatal outcomes. Ursodeoxycholic acid (UCDA) is a naturally occurring bile acid that is used to treat hepatobiliary disorders. Ursodeoxycholic acid works in several ways. It increases bile acid secretion and stabilizes the plasma membrane, allowing the placenta to more effectively transfer bile acids from the fetus, and protects liver cells from the toxic effects of bile acids.1,8 It is first-line treatment for maternal pruritis and improvement of laboratory values (SMFM grade 1A recommendation).2 The evidence supporting the use of UCDA and improvement of fetal outcomes is less certain.8,9
Ursodeoxycholic acid has no known adverse effects on the fetus and is usually well tolerated by the pregnant individual. It is taken orally 300 mg 2 to 3 times per day (10–15 mg/kg/d and can be titrated to a maximum dose of 21/mg/kg/d). Initiation of empiric treatment based on symptoms at the time of testing is acceptable. Pruritis should improve in 1 to 2 weeks. Laboratory values usually improve within 3 to 4 weeks. Although not rigorously studied, S-adenosyl-methionine and cholestyramine are alternative treatment options. Antihistamines like diphenhydramine and hydroxyzine may also help alleviate pruritis.2
Antepartum surveillance
The rate of stillbirth is increased when ICP is diagnosed, most significantly when bile acids are 100 μmol/L or greater and usually occurring in the third trimester.1 Stillbirth related to ICP is believed to be caused by an acute, sudden event rather than a chronic placental vascular process. Spontaneous preterm labor and birth associated with ICP likely results from the elevation in bile acids activating myometrial oxytocin receptors.7
The efficacy of fetal monitoring to predict or reduce the risk of stillbirth related to ICP is unknown. However, recent trials support fetal surveillance noting lower rates of adverse perinatal outcomes potentially due to intensive monitoring and late preterm or early term delivery. Clinical guidelines are reliant on expert consensus to determine optimal management.1,2,7 The Society of Maternal-Fetal Medicine recommends initiating antepartum surveillance at a gestational age when delivery would be performed in response to abnormal results (SMFM grade 2C). Patients should be informed that this recommendation acknowledges the uncertainties surrounding the benefits, risks, and burdens of antenatal surveillance.2,10 Additionally, the optimal frequency and method of antenatal testing is not clear. Recommendations should be individualized by taking into consideration bile acids levels and the presence of comorbidities. The frequency of repeat bile acid measurement has not been agreed on. Weekly testing is not recommended but repeat testing may help guide delivery timing.2
Individuals with ICP may be at increased risk for preeclampsia, typically occurring 2 to 4 weeks after ICP diagnosis. Those who develop ICP early during pregnancy and those with multiple gestations are at higher risk.11 Currently there are no specific monitoring recommendations other than blood pressure and proteinuria surveillance.11 If not done at an earlier prenatal visit as part of routine screening, testing for hepatitis C infection may be considered as it is a risk factor for ICP.
Delivery
Timing of delivery is based on risk stratification guided by serum bile acid concentration and patient presentation.7 Pregnant individuals with total bile acid levels of 100 μmol/L or greater should give birth at 36 0/7 weeks (SMFM grade 1B recommendation). Those with bile acid levels lower than 100 μmol/L can give birth between 36 0/7 and 39 0/7 weeks. Those with bile acid levels of under 100 but 40 μmol/L or greater should be considered for earlier delivery of the given time range, whereas those who have bile acid levels lower than 40 μmol/L should give birth later in the time range. Antenatal corticosteroids should be administered for fetal lung maturity when delivery is planned before 37 weeks (SMFM grade 1A recommendation).2
Giving birth earlier than 36 weeks can be considered for individuals with ICP and bile acids greater than 100 μmol/L accompanied by excruciating and unremitting pruritus not responding to medication, or a prior history of stillbirth prior to 36 weeks due to ICP, or pre-existing or acute hepatic disease with evidence of worsening hepatic function (see Algorithm).
 Strategic consideration of the balance between neonatal mortality and morbidity associated with early birth and the risk of stillbirth should be weighed and discussed using shared decision making.2 The nurse practitioner (NP) providing care for a patient with ICP should collaborate with maternal fetal medicine and delivering providers to coordinate optimal antepartum management and delivery timing.
Strategic consideration of the balance between neonatal mortality and morbidity associated with early birth and the risk of stillbirth should be weighed and discussed using shared decision making.2 The nurse practitioner (NP) providing care for a patient with ICP should collaborate with maternal fetal medicine and delivering providers to coordinate optimal antepartum management and delivery timing.
Postpartum care
Patients can be reassured that ICP typically resolves spontaneously shortly after delivery and that breastfeeding is not contraindicated.10 Repeating bile acids measurement in the postpartum period is not usual practice. Data suggest that individuals with a history of ICP are at risk for developing hepatobiliary diseases later in life. Evaluation of liver function tests at 6 weeks postpartum should be considered.1,2,10
Individuals diagnosed with ICP should be counseled on the likelihood of recurrence in subsequent pregnancies, which has been estimated to be up to 90%.1,2,10 Pregnancy-related cholestasis does not pose any restrictions on hormonal contraception. Estrogen-containing options are acceptable given that these fall into the category 2 classification on the US Medical Eligibility Criteria. Progesterone-only methods are category 1 and carry no restrictions.12
Conclusion
The sometimes unrelenting and intense pruritis associated with ICP can cause significant distress. Although these symptoms can often be managed, the anxiety surrounding the diagnosis can be more difficult to control.
 Understanding and utilizing the most up-to-date evidence, NPs can effectively diagnose and manage ICP (Box). Thorough patient counseling and shared decision making regarding patient referral, antenatal surveillance, and delivery planning is imperative for optimal outcomes and can lessen patient anxiety when ICP is diagnosed.
Understanding and utilizing the most up-to-date evidence, NPs can effectively diagnose and manage ICP (Box). Thorough patient counseling and shared decision making regarding patient referral, antenatal surveillance, and delivery planning is imperative for optimal outcomes and can lessen patient anxiety when ICP is diagnosed.
Resources in English and Spanish can be found here.A Also useful is their quick healthcare provider microlearning video.B The CREOGS Over Coffee podcastC is another great resource.
Dalisa Barquero is WHC Nurse Practitioner, Clinical Lead, at Mountain Park Health Center in Phoenix, Arizona. Genevieve M. Hofmann is Assistant Professor at the University of Colorado School of Medicine in Aurora, Colorado. The authors have no actual or potential conflicts of interest in relation to the contents of this article.
References
- Wood AM, Livingston EG, Hughes BL, Kuller JA. Intrahepatic cholestasis of pregnancy: a review of diagnosis and management. Obstet Gynecol Surv. 2018;73(2):103-109.
- Society for Maternal-Fetal Medicine (SMFM); Lee RH, Greenberg M, Metz TD, Pettker CM. Society for Maternal-Fetal Medicine consult series #53: Intrahepatic cholestasis of pregnancy: replaces consult #13, April 2011. Am J Obstet Gynecol. 2021;224(2):B2-B9.
- Piechota J, Jelski W. Intrahepatic cholestasis in pregnancy: review of the literature. J ClinMed. 2020;9(5):1361.
- Papacleovoulou G, Abu-Hayyeh S, Nikolopoulou E, et al. Maternal cholestasis during pregnancy programs metabolic disease in offspring. J Clin Invest. 2013;123(7):3172-3181.
- Manzotti C, Casazza G, Stimac T, et al. Total serum bile acids or serum bile acid profile, or both, for the diagnosis of intrahepatic cholestasis of pregnancy. Cochrane Database Syst Rev. 2019;7(7):CD012546.
- Gardiner FW, McCuaig R, Arthur C, et al. The prevalence and pregnancy outcomes of intrahepatic cholestasis of pregnancy: a retrospective clinical audit review. Obstet Med. 2019;12(3):123-128.
- Ovadia C, Seed PT, Sklavounos A, et al. Association of adverse perinatal outcomes of intrahepatic cholestasis of pregnancy with biochemical markers: results of aggregate and individual patient data meta-analyses. Lancet. 2019;393(10174):899-909.
- Chappell LC, Bell JL, Smith A, et al. Ursodeoxycholic acid versus placebo in women with intrahepatic cholestasis of pregnancy(PITCHES): a randomised controlled trial. Lancet. 2019;394(10201):849-860.
- Walker KF, Chappell LC, Hague WM, et al. Pharmacological interventions for treating intrahepatic cholestasis of pregnancy. Cochrane Database Syst Rev. 2020;7(7): CD000493.
- Bicocca MJ, Sperling JD, Chauhan SP. Intrahepatic cholestasis of pregnancy: review of six national and regional guidelines. Eur J Obstet Gynecol Reprod Biol. 2018;231:180-187.
- Mor M, Shmueli A, Krispin E, et al. Intrahepatic cholestasis of pregnancy as a risk factor for preeclampsia. Arch Gynecol Obstet. 2020;301(3):655-664.
- Centers for Disease Control and Prevention. CDC contraceptive guidance for health care providers. U.S. Medical Eligibility Criteria for Contraceptive Use, 2016.
KEY WORDS: intrahepatic cholestasis of pregnancy, bile acids, stillbirth, preterm birth, pruritus, ursodeoxycholic acid

