Nearly 80 million Americans are infected with human papillomavirus (HPV), making it the most common sexually transmitted infection in the United States.1 The HPV 9-valent vaccine, recombinant, protects against nine HPV types and prevents cervical, vaginal, vulvar, anal, penile, and oropharyngeal cancer, as well as genital warts.2,3 The Centers for Disease Control and Prevention recommends the HPV vaccine for everyone starting at age 11 or 12 years and up to age 26 years.1 Individuals between the ages of 27 and 45 years may discuss their option to vaccinate with a healthcare provider.1 Despite the availability of a well-studied, safe, and effective vaccine, the national HPV vaccination coverage is below 50% among adolescents and the rates continue to lag among US military members.4,5
 In 2017, 66% of 13-to 17-year-old adolescents initiated the HPV vaccine series, but only 49% of adolescents completed the series in the US.4 HPV vaccination completion rates among US service members are even lower than the general population.4,5 The US Department of Defense’s overall HPV initiation rates for women and men, ages 17 to 26 years, serving in the military from 2007 to 2017 were 26.6% and 5.8%, respectively.5
In 2017, 66% of 13-to 17-year-old adolescents initiated the HPV vaccine series, but only 49% of adolescents completed the series in the US.4 HPV vaccination completion rates among US service members are even lower than the general population.4,5 The US Department of Defense’s overall HPV initiation rates for women and men, ages 17 to 26 years, serving in the military from 2007 to 2017 were 26.6% and 5.8%, respectively.5
Currently, many providers only assess HPV vaccination status at annual well-woman visits. A quality improvement (QI) approach to the efficient delivery of healthcare services is the use of a bundle intervention (BI). A bundle, which is a set of evidence-based strategies that, when implemented reliably and collectively, is shown to improve patient outcomes, can be used to increase uptake of prevention strategies such as HPV vaccination.6 A bundle approach for immunizations includes multiple modalities such as staff education, electronic health record (EHR) alerts, educational information, reminders, and immunization champions that are used to increase vaccine uptake.7 This QI project used an evidence-based bundle approach to address the Healthy People 2030 goal of reducing infection from the HPV types that can be prevented by the HPV 9-valent vaccine, recombinant, in young adults.8
Project purpose
The purpose of this QI project was to increase HPV vaccine uptake in a military outpatient setting. HPV vaccine uptake is defined as the next due dose, including dose 1, 2, or 3. Missed opportunities (MO) are when a patient who is eligible for the HPV vaccine presents for a visit and HPV vaccination status was not assessed, counseling about HPV was not provided, or a recommendation for the HPV vaccine was not given by the provider. The objectives were to reduce MO by 10% and increase HPV vaccine uptake by 10% from baseline among females ages 11 to 26 years.
Description of the setting and population
The targeted population was female military-connected personnel, including active-duty service members, spouses and daughters of service members, and veterans. The sample included nonpregnant females, ages 11 to 26 years, seeking care in a suburban, hospital-based outpatient obstetrics/gynecology (ob/gyn) clinic at a military treatment facility (MTF) in the Midwest over 6 months between August 2019 and January 2020. The project was determined not human subject research by the organization’s Institutional Review Board.
Description of the quality improvement project
Methods to evaluate outcomes
Patient charts were reviewed before and after implementation of the BI using an EHR and an immunization tracking system to measure outcomes. The primary outcomes were measured by comparing 3-month preintervention data to postintervention data for the numbers of MO and vaccine-eligible female patients who received the next due HPV vaccine. Secondary outcomes were measured by comparing provider assessment of patients’ HPV vaccination status and/or utilization of the computer-generated prompt in the clinical template pre-and post-intervention. An anonymous questionnaire was used to gather patients’ demographic information to describe the sample, and three questions were asked to determine factors impacting HPV vaccination decisions: “Do you intend to receive the HPV vaccine today?”; “If yes, which factor influenced you the most?”; “If no, which factor influenced you the most?”
Outcomes
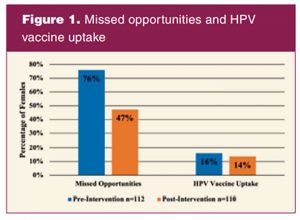 From August 2019 to October 2019 (pre-intervention), 112 females met the project criteria out of 399 patients seeking care in the ob/gyn clinic. From November 2019 to January 2020 (intervention), 110 females met the criteria out of 371 patients seeking care in the ob/gyn clinic. In the pre-intervention, there were 85 (76%) MO and 18 patients (16%) who received the HPV vaccine compared to 52 MO (47%) and 15 patients (14%) who received the HPV vaccine in the intervention period (Figure 1).
From August 2019 to October 2019 (pre-intervention), 112 females met the project criteria out of 399 patients seeking care in the ob/gyn clinic. From November 2019 to January 2020 (intervention), 110 females met the criteria out of 371 patients seeking care in the ob/gyn clinic. In the pre-intervention, there were 85 (76%) MO and 18 patients (16%) who received the HPV vaccine compared to 52 MO (47%) and 15 patients (14%) who received the HPV vaccine in the intervention period (Figure 1).
 Providers only used the prompt in the clinical template twice (2%) during the pre-intervention period compared to 19 times (17%) in the intervention period (Table).
Providers only used the prompt in the clinical template twice (2%) during the pre-intervention period compared to 19 times (17%) in the intervention period (Table).
During the pre-intervention period, providers assessed the HPV vaccination status of 23 patients (21%) compared to 59 patients (54%) in the intervention period (Table).
A total of 78 patients completed the questionnaire during the intervention period for a response rate of 71%. The majority of patients were Caucasian (75%), non-Hispanic (85%), and single (68%). Approximately half of the sample (47%) were active-duty service members. A small percentage of patients were ages 11 to 17 years (8%), and most patients were ages 18 to 26 years (92%).
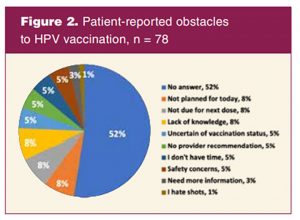 Patients completed the questionnaire before seeing a provider. EHRs were updated during visits based on the patient’s interview and survey to reflect the most accurate data because Tricare-insured patients could also receive healthcare including the HPV vaccination at a nonmilitary clinical site. Most patients (85%) did not report factors leading to HPV vaccination, but of those patients (15%) who did report a factor it was the provider’s recommendation that was the primary predictor for HPV vaccination. Approximately half (48%) of the sample reported obstacles to HPV vaccine uptake (Figure 2).
Patients completed the questionnaire before seeing a provider. EHRs were updated during visits based on the patient’s interview and survey to reflect the most accurate data because Tricare-insured patients could also receive healthcare including the HPV vaccination at a nonmilitary clinical site. Most patients (85%) did not report factors leading to HPV vaccination, but of those patients (15%) who did report a factor it was the provider’s recommendation that was the primary predictor for HPV vaccination. Approximately half (48%) of the sample reported obstacles to HPV vaccine uptake (Figure 2).
Some of the common reported obstacles to HPV vaccination were not being due for the next dose (8%), not having planned to receive the HPV vaccine (8%), and lack of knowledge (8%) (Figure 2).
Discussion
The BI was effective at reducing MO and leading to favorable modifications of providers’ behaviors. Although the BIs decreased MO by 29%, the intervention did not make a difference in actual HPV vaccine uptake. This finding is not consistent with the evidence that shows BIs improve HPV vaccination rates. However, the questionnaire results are congruent with the evidence that provider’s recommendations are a primary predictor of HPV vaccination. Furthermore, the BI led to an increase of 33% in the frequency of providers assessing the patient’s HPV vaccination status and an increase of 16% in the frequency of providers utilizing the clinical prompt.
A major barrier encountered during the implementation of this project was vaccines were not available in the ob/gyn clinic. The HPV vaccine was only administered in the immunization clinic. This could be done on the same day that the vaccine was recommended or at a later date.
Areas to improve include stocking/administering the vaccine in all clinics and using a prompt specific for overdue vaccines instead of a preventive service prompt, which includes all immunizations, labs, and screening tests. Bundle components to further evaluate include automated reminders (calls/texts/letters), monthly feedback to providers, using a checklist to ensure providers use the Health Belief Model, and continuing support from ICs for providers to recommend the HPV vaccine.
Additional QI projects using evidence-based bundled approaches are needed to determine if these multi-component interventions lead to an increase in HPV vaccine uptake over time in an MTF. Further research is needed to identify a superior multicomponent intervention to improve HPV vaccination rates in ob/gyn clinics and military settings.
Conclusion
The small number of patients, short intervention period, and brief data collection period are limitations. The Covid-19 pandemic was a significant limitation as it led to an early cessation of tracking receipt of the HPV vaccine in the immunization clinic tracking system. Data were collected up to 64 days past the last day of the pre-intervention period, but only up to 13 days past the last day of the implementation period. It is likely that more patients who received the HPV vaccine would have been identified if tracking had continued for the intended amount of time to match pre-and post-intervention data. Also, the short intervention period led to the components within the bundle to be implemented simultaneously, rather than sequentially introduced. Further evaluation of this bundle is warranted in a larger population at additional sites and over a longer period.
Utilization of the BI reduced MO by 29%. However, there was no difference in HPV vaccine uptake. The findings suggest the unique military setting makes it challenging to translate evidence found in the civilian population to the military community. To improve HPV vaccination rates in the military population, we recommend stocking/administering the vaccine in the clinic, the Department of Defense’s (DOD) mandate the HPV vaccine to all active service members, and the DOD use an EHR capable of tracking vaccinations.
Dynasty I. Stokes is a women’s health nurse practitioner, sexual assault medical manager, and Chief Medical Informatics Officer in the United States Air Force. Randee Masciola is Associate Professor of Clinical Nursing, Doctor of Nursing Practice Program Director, clinical expert, and women’s health nurse practitioner at Ohio State University, College of Nursing, in Columbus, Ohio. Jennifer Kue is Associate Professor at University of South Florida, College of Nursing. Kathleen Drum is Medical Services Flight Commander and a women’s health nurse practitioner in the United States Air Force. Dianne Morrison-Beedy is Centennial Professor of Nursing and Chief Talent and Global Strategy Officer at Ohio State University, College of Nursing. The authors have no actual or potential conflicts of interest in relation to the contents of this article. The views expressed are those of the author and do not necessarily reflect the official policy or position of the Air Force, the Department of Defense, or the United States government. Clearance number 88ABW-2021-0772.
References
1. Centers for Disease Control and Prevention. Genital HPV infection–Fact sheet. January 19, 2021.
2. National Cancer Institute. Human papillomavirus (HPV) vaccines. May 25, 2021.
3. US Food and Drug Administration. Gardasil 9. August 21, 2020.
4. Centers for Disease Control and Prevention. HPV vaccination coverage data. August 24, 2017.
5. Clark LL, Stahlman S, Taubman SB. Human papillomavirus vaccine initiation, coverage, and completion rates among U.S. active component service members, 2007–2017. MSMR. 2018;25(9):9-14.
6. Institute for Healthcare Improvement. What is a bundle? 2021.
7. Holloway GL. Effective HPV vaccination strategies: what does the evidence say? An integrated literature reviews. J Pediatr Nurs. 2019;44:31-41.
8. US Department of Health and Human Services, Office of Disease Prevention and Health Promotion. Reduce infections of HPV typesprevented by the vaccine in young adults—IID‑07. Healthy People 2030.

