Despite advances in contraception, rates of unintended pregnancy remain high in the United States. Emergency contraception is a safe and effective way to reduce this risk. There have been various changes in available methods and corresponding legislation. This article provides an update on available methods, ongoing barriers to access, and strategies to increase use and reduce risk.
Womens Healthcare. 2022;10(3):21-27. doi: 10.51256/WHC062221
Unintended pregnancy and its consequences continue to present a public health challenge. Unintended pregnancy is commonly defined as a pregnancy that was not planned at that time; whether pregnancy was wanted at a later time or not wanted at all further clarifies a woman’s intentions.1 Outcomes of unintended pregnancy may include an unplanned birth, miscarriage, or termination. The Behavioral Risk Factor Surveillance System identified 76.2% of women age 18 to 49 years “at risk” for unintended pregnancy.2 The term “at risk” included being sexually active with a male partner, not currently pregnant or seeking pregnancy, not using permanent contraception (female or male sterilization), and not having had a hysterectomy.2 Of these women, one-third (30%) reported not using any method of contraception.2,3
Safe and effective contraceptive methods to prevent unintended pregnancy have been available for decades. Sterilization has remained the most common method over time, followed by oral contraceptives.4 Over the past 20 years, we have seen increased use of long-acting reversible contraceptives such as intrauterine devices (IUD), with a corresponding decrease in other methods.4
The majority of unintended pregnancy results from inconsistent or nonuse of an effective method of contraception.5 Postcoital methods of contraception, commonly referred to as emergency contraception (EC), are an option when pregnancy is not desired in instances of method failure, misuse, inadequate use, or absence of method.5–7 Sexual assault, coercion, and rape may also be indications for EC. As well, EC has the potential to impact rates of unintended pregnancy, birth from an unplanned pregnancy, and abortions.5–7
The purpose of this article is to provide an overview of currently available EC methods, review pertinent information to facilitate the most effective and safe use of these methods, and address issues of barriers to access and use.
Emergency contraception
Methods to prevent pregnancy after acts of unprotected intercourse (UPI) have been available since the mid-1970s.8 In 1998, the US Food and Drug Administration approved the first prescription product specifically for emergency, or postcoital, contraception.6 In 2006, the first oral, progestin-only emergency contraceptive pills (ECP) became available over the counter (OTC) to women ages 18 years and older.6,8 By 2013, age and gender restrictions for OTC EC purchase were lifted and generic forms became available.9 Over the past 15 years, there have been numerous changes in regulations related to EC access. These changes have been linked to confusion among providers, pharmacies and associated staff, and the general public, likely impacting utilization of this highly effective method to reduce unintended pregnancy risk.5,9
Use of OTC ECP increased significantly between 2008 and 2015, from 11% to 23%, across all races and ethnicities.7 This increase was attributed to fears of primary method failure (41%) or unprotected sex (50%).7 Repeat use of EC has remained fairly consistent at 45% among ever users, with 10% of all women reporting the use of EC more than once in 2015.3,7 In 2018, 0.2% of women between the ages of 15 and 49 years reported using EC in the last month.3 The National Survey of Family Growth reports increased use of EC with higher levels of education.3 Women with a bachelor’s degree or higher are more than twice as likely to have used EC than women without a high school diploma or GED, at 27.9% versus 12.6%, respectively.3
Despite an increase in EC use, myths, misconceptions, as well as barriers to access persist, particularly among the most vulnerable. Providers can effectively address these issues and provide guidance on overcoming barriers to sexually active, reproductive-age women; however, only 7% of ever users of EC reported counseling about EC from providers in 2015.7
Emergency contraceptive options
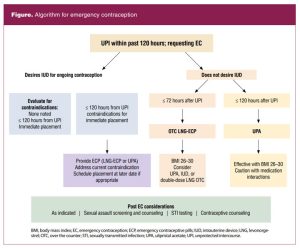
Currently, there are three widely accepted and available forms of emergency contraception: two oral hormonal preparations and one nonhormonal intrauterine device.5,6,10 Only one method is available OTC. All of these are effective, but there are pros and cons to each method. The US Medical Eligibility Criteria (MEC) specifically indicate EC may be made available to any and all women who do not desire pregnancy.6 Risks associated with EC use do not outweigh the benefit, even for women who may otherwise have contraindications.6
Levonorgestrel 1.5 mg
The most commonly known and widely used form of EC is a progestin-only (levonorgestrel [LNG] 1.5 mg) pill.6,10 Often referred to as “the morning after pill,” this option gained wider recognition when it became available OTC. The primary mechanism of action of LNG-ECP is delay of ovulation. It is important to note that LNG-ECP will not cause harm to an already established pregnancy and does not interfere with implantation of an already fertilized egg.5,6,10 These misconceptions may contribute to individual decisions against ECP use.
This medication should be taken as soon as possible, and can be taken up to 72 hours after UPI, to prevent pregnancy.5,6,10 Failure rates vary, but when LNG-ECP is taken as recommended, these rates are reported as 0.3% to 2.6%. Efficacy does decrease incrementally with time, particularly between 72 and 120 hours after.5,6 There are some concerns about reduced efficacy with LNG-ECP in women with a body mass index (BMI) greater than 26 kg/m2 (appx 155 lbs), as blood serum levels of LNG have been reported as 50% lower in women with a BMI greater than 30 kg/m2(~ 175 lbs).5,6 Therefore, it is recommended that if other options for EC are not available, women with a BMI greater than 30 kg/m2 take a double dose of LNG-ECP. Double-dose regimens (3.0 mg LNG) could also be considered for women on enzyme-inducing drugs such as antiseizure medications and certain antiretroviral therapies.5
Use of LNG-ECP does not require physical examination or laboratory testing prior to use and has a documented safety profile. There are no risks to LNG-ECP use that outweigh the benefits of preventing unintended pregnancy. Relative and actual contraindications according to the MEC for routine hormonal contraceptive use do not apply to the short-term, limited use of LNG-ECP.5,6,11
Repeated acts of intercourse within the same menstrual cycle will require another method of pregnancy prevention or additional dosing of EC.5,6,10 Patients can resume or initiate combination or progestin-only hormonal contraceptives the same day they take LNG-ECP (quick start) as medically appropriate, and abstain from sexual intercourse or use a barrier method for at least 7 days. Alternatively, hormonal contraception can be initiated with the onset of menses after EC use.6
The most common side effects of LNG-ECP are nausea (13%–23%) and abdominal pain (13%–18%).5,6,10 If vomiting occurs (5.6%) within 3 hours of the dose, a second dose is recommended.5 Side effects typically resolve within 24 hours of administration. The timing of menses is dependent on the point in the cycle when EC is used. If menses is delayed by 1 week, or if lower abdominal pain and spotting are experienced, the individual should perform a pregnancy test and seek care as needed.6
Ulipristal acetate
Ulipristal acetate (UPA; 30 mg) is an anti-progestin that became available for use as EC in the US by prescription only in 2010.6 Similar to LNG-ECP, the mechanism of action is delay of ovulation. UPA is more effective than LNG-ECP within the first 72 hours and has FDA approval for use between 72 and 120 hours (3–5 days) after UPI.5,6,10 Failure rates are reported as 0.0% to 1.8% through 120 hours (5 days), and UPA has also proven effective in women with elevated BMI (> 30 kg/m2).5,6 Drug interactions resulting in reduced efficacy of UPA may occur with hepatic enzyme-inducing medications (administered up to 1 month prior to use) and drugs reducing gastric pH (antacids, H2blockers).5 Modifications in dosages have not been studied. Combination hormonal contraceptives, progestin-only contraceptives, or LNG-ECP taken within 5 days before or after UPA may decrease efficacy due to likely competition for receptor sites. More specifically, if EC is sought due to missed or late use of any of the hormonal contraceptive methods but these were taken within the previous 5 days, LNG-ECP is preferred over UPA.5,10 Hormonal contraceptives containing a progestin should not be initiated within 5 days of UPA use as the efficacy of either may be decreased.5,6,10 A barrier method or abstinence should be used for those 5 days and continued for the first 7 days after initiating or restarting hormonal contraception.5
There are no risks to use of UPA that outweigh the benefits of prevention of unintended pregnancy. No harms to a previously established pregnancy have been documented and no teratogenic effects have been documented with UPA use during pregnancy.5,6 Headache (19%) is the most common side effect, followed by dysmenorrhea (13%) with the next cycle.5,6 UPA has been found in small quantities in breast milk. Therefore, it is recommended that women who are breastfeeding discard breast milk for 24 hours, and up to 7 days per European guidelines, following use. There are no current US guidelines.5 Timing of menses after UPA use are cycle point dependent, but typically occurs approximately 2 days later than expected.5 If expected menses is delayed by 1 week or more, a pregnancy test should be performed and if needed follow up with a provider.5,6
Despite the increased efficacy, particularly in women with a higher BMI, the primary barrier to UPA is the need for a prescription, followed by limited availability in pharmacies.5,10 Providers could offer prescriptions to patients to have “on hand” while advocating for a more reliable, consistent method of birth control.
Copper intrauterine device
The copper (T380A) intrauterine device (IUD) is the most effective method of EC, although this indication is not FDA approved.6,9 The reported effectiveness rate with insertion up to 5 days (120 hr) after UPI is nearly 100%, with only 10 pregnancies in over 7,000 insertions.5,6,10 Some studies have documented efficacy with IUD insertion between 5 and 10 days after UPI with confirmation of a negative pregnancy test prior to insertion, but this is not the standard recommendation.5,6 The mechanism of action is not fully understood, although it is believed to be similar to the action of the IUD in general (ie, change in cervical mucus and change in intrauterine environment preventing implantation).5
Current pregnancy should be reasonably ruled out prior to IUD insertion.5,11 Screening for chlamydia and gonorrhea should be done if indicated by age or risk factors but insertion need not be delayed while waiting for results.11 Contraindications to IUD use for EC are the same as those for routine placement. These include current purulent cervicitis, active infection with chlamydia or gonorrhea, pelvic infection, untreated reproductive tract cancer, unexplained bleeding, uterine malformation, and active HIV without adequate suppression. There is heightened risk for uterine perforation and expulsion if an IUD is inserted during the first 4 weeks postpartum.11 Some patients may experience an increase in menstrual discomfort and flow after IUD insertion.10 As with routine IUD insertion, the patient should be advised to return if there are any concerns with the IUD including a suspected pregnancy, if a change in contraceptive method is desired, and when it is time to remove or replace the IUD.11 The primary benefit of an IUD is the immediate and ongoing contraceptive benefit. The T380A IUD is approved to remain in place for 10 years, offering long-term, reversible contraception.5,6,11 The primary drawback of the IUD for emergency contraception is the need for an appointment with a trained provider for insertion. Patients with health insurance coverage through a private provider or the Affordable Care Act typically can acquire an IUD for little or no cost, with most out-of-pocket expenses offset by the long-term benefit and reduced long-term costs.5
Additional clinical considerations
Addressing sexual and reproductive health, including pregnancy intentions, should be included in well and problem visits for reproductive-age women. Providing information about the safety, availability, and ways to access emergency contraception prior to situations for which the need is urgent may have an impact on unintended pregnancy rates.
Patients in need of EC may be facing difficult situations, have concerns about confidentiality, or may be embarrassed about not having used contraception. Nonjudgmental care is essential. It is important for providers to conduct physical and psychological assessments for reproductive coercion and sexual assault when discussing current or prior use of EC with patients.6 Starting a method of regular contraception should be discussed but should not be a prerequisite to receiving EC.
Always confirm that the patient does not desire a pregnancy related to the UPI that occurred. Explain what is known about the effectiveness and any individual factors that may reduce effectiveness of the different EC options. Reinforce that there is no ongoing protection from pregnancy after taking ECPs. EC use provides an opportunity to address ongoing needs for pregnancy prevention.
Data indicate younger women are at greater risk for unintended consequences of sexual activity such as unintended pregnancy and STIs.12 EC provides no protection against STIs or HIV, and its use indicates a potential increase in risk for STI. If and when women present at the office or clinic for or post EC, provide appropriate STI screening and risk reduction counseling along with contraceptive method counseling.12 Survivors of sexual assault often have concerns about pregnancy, STIs, and HIV. Screening for infections and presumptive treatment if desired should be part of the care provided.
Barriers to access and use of emergency contraception
Various forms of EC are available with and without a prescription. ECP became available without a prescription in 2006.8,9 By 2013, all age and gender restrictions were removed and generic forms became available. Despite increases in availability and reported use, EC remains an underutilized method of pregnancy prevention, particularly among those most vulnerable.
Knowledge
One of the primary barriers to use of EC is the persistent gap in knowledge. Various studies have reported knowledge gaps and misconceptions about EC, including available forms, timing of use, side effects, contraindications, safety, and mechanism of action.5,8 Misunderstandings of the application of MEC criteria for hormonal contraception to EC use have been cited as concerns impacting provider provision of EC.5,13 Many women, and providers, are still under the misconception that adolescents and nulliparous women are not candidates for an IUD and providers may be less familiar with UPA.5 Often, people continue to confuse emergency contraception with the medication used to medically induce an abortion.5,14 Some studies note provider resistance to providing education on EC in general and to adolescents in particular.6,14 The most commonly cited reasons are concerns that EC is associated with an increase in risky sexual behavior despite numerous studies refuting this belief, time constraints, and the belief that individuals cannot use the method appropriately independently.6,14
It is essential that providers remain cognizant of current guidelines and recommendations for emergency contraception and offer clarity, dispel myths and misconceptions, and offer guidance to empower individual decision making.
Cost and availability
Additional barriers to access are related to cost, product availability, inaccurate information and restrictions imposed by pharmacy or vendor staff (eg, age restrictions), and concerns related to confidentiality and parental consent. For example, OTC availability does not necessarily equate with access. Often OTC ECP are kept in a locked cabinet or behind the counter and available stock may be limited. Some pharmacies have cited risk of theft of the product as justification for this practice, but it forces those seeking ECP to request direct assistance from store personnel, contributing to discomfort and embarrassment.5,8,9,13
Cost of OTC ECP has frequently been reported as a barrier to use, particularly for adolescents and women living in or close to poverty.5,7 Unintended pregnancy also disproportionately affects these groups.1,5 Unfortunately, most insurers do not provide options for reimbursement of products purchased OTC. Others do not provide coverage for any contraceptive services (eg, religious employers).5,6 Typical cost for OTC ECP ranges from $40 to $50, generic or brand, respectively.10 There are options to purchase LNG-ECP online at a lower cost ($25 including shipping) to have on hand should the need arise.5 Ulipristal as a prescription, depending on coverage, costs between $3 and $1,200, with a median cost of $50.5
Pharmacies in lower-income neighborhoods are 50% less likely to have the product available compared with pharmacies in higher-income neighborhoods.5,13 Additionally, pharmacies in lower-income neighborhoods were more likely to impose age restrictions and prescription requirements.13 Similarly, women in rural areas face significant challenges related to provider and product availability. Veterans and active members of the military have coverage for EC services, although product and provider availability may be limited, particularly depending on location.14 The most cost-effective method of EC is the IUD, especially when adding the cost-saving benefit of extended contraception.
Providers can assist patients to overcome barriers to EC access with information about online ordering, cost-saving measures, and by directing them to pharmacies that are known to support EC availability. Increased availability of IUD insertion training for providers, especially in rural and low-income settings, can improve access.5,13
System wide/policy barriers
There are various policy and system wide barriers to effectively reducing the unintended pregnancy rate and increasing access to EC. For example, 33 states do not require emergency departments to provide information on EC options to women seeking care for sexual assault.5,15 Providers and facilities may refuse to provide information on or actual EC to patients on moral or religious grounds.5,6,15 Pharmacies and pharmacists are also permitted to refuse to stock, dispense, or provide information on EC on moral or religious grounds. The majority of these positions are rooted in the misconception that EC, in any form, causes an abortion. FDA regulations require generic preparations of ECP to include a statement recommending the product “for women 17 years of age and older,” contributing to confusion among adolescents related to ability to access and inappropriately imposed age restrictions by pharmacy personnel.6,9,13
Numerous organizations have issued position statements and opinions in support of expanded access to effective methods of contraception, including emergency contraception. Among these organizations are the National Association of Nurse Practitioners in Women’s Health, Association of Women’s Health, Obstetrics, and Neonatal Nurses, and the American College of Obstetricians and Gynecologists.14,16,17
Improvements and advancements
Other methods for EC are being explored. Mifepristone (previously known as RU-486), in a low dose, is currently available in several countries for use as EC. It prevents ovulation by blocking the effect of progesterone through binding to its receptors and has an excellent safety and low side-effect profile.5 Research is under way for the use of LNG IUDs for EC, and initial results are promising.5 Although any IUD can be removed at any time, the “12 year” feature of the copper IUD may cause hesitation among younger women. The shorter intended duration of the LNG IUDs may be a beneficial “marketing tool” for use in general, as well as for EC.
The American College Health Association includes provision of contraceptive services, including EC, in their Best Practices guidelines.18 In 2017, Stanford University was the first college to offer ECP via a vending machine.19 Other colleges and universities have implemented this approach, but it is not yet widespread. Future research and/or initiatives regarding vending machine methods for distribution, particularly in lower-resource settings, are warranted.
The emergence of online options to purchase ECP, other forms of contraception, and access provider services is promising. This approach has the potential to increase availability of services to those who may otherwise face challenges, but the benefits of this method will not be actualized if young men and women are not aware of these options. Increasing marketing efforts of these types of services are warranted.
Conclusion
Emergency contraception is an available, safe, effective method to reduce the risk of unintended pregnancy after unprotected sexual intercourse. Women, particularly the most vulnerable, still face barriers to access and use, resulting in a continued risk of unintended pregnancy. At the clinical level, providers can discuss sexual activity and pregnancy intentions as essential components of well-woman care. As appropriate, effective methods to prevent pregnancy that include EC options to address method failure or misuse should be part of sexual and reproductive health counseling. Provider knowledge about the safe and effective use of EC and resources to provide access are crucial. At the policy level, efforts must continue to improve access to EC including improved insurance coverage and removal of unnecessary restrictions.
Mary T. Hickey is Associate Professor and Associate Dean at Adelphi University, College of Nursing and Public Health, in Garden City, New York. The author has no actual or potential conflicts of interest in relation to the contents of this article.
References:
- Finer LB, Zolna MR. Declines in unintended pregnancy in the United States, 2008-2011. N Engl J Med. 2016;374(9):843-852.
- Zapata L, Pazol K, Curtis K, et al. Need for contraceptive services among women of reproductive age–45 jurisdictions, United States, 2017-2019. Morbidity and Mortality Weekly Report (MMWR). 2021;70(25).
- Daniels K, Abma JC. Current contraceptive status among women aged 15–49: United States, 2017–2019. NCHS Data Brief. 2020;(388):1-8.
- Kavanaugh ML, Pliskin E. Use of contraception among reproductive-aged women in the United States, 2014 and 2016. F S Rep. 2020;1(2):83-93.
- Haeger KO, Lamme J, Cleland K. State of emergency contraception in the U.S., 2018. Contracept Reprod Med. 2018;3:20.
- American College of Obstetricians and Gynecologists. Practice bulletin no. 152: Emergency contraception. Obstet Gynecol. 2015;126(3):e1-e11. Reaffirmed 2018.
- Hussain R, Kavanaugh ML. Changes in use of emergency contraceptive pills in the United States from 2008 to 2015. Contracept X. 2021;3:100065.
- Hickey MT, White J. Female college students’experiences with and perceptions of over-the-counter emergency contraception in the United States. Sex Reprod Healthc. 2015;6(1):28-32.
- Cleland K, Bass J, Doci F, Foster AM.Access to emergency contraception in the over-the-counter era. Womens Health Issues. 2016;26(6):622-627.
- Stone R, Rafie S, El-Ibiary SY, Vernon V, Lodise N. Emergency contraception algorithm and guide for clinicians. Nurs Womens Health. 2017;21(4):297-305.
- Centers for Disease Control and Prevention. Reproductive health. US Selected Practice Recommendations for Contraceptive Use, 2016.
- Hickey MT, Shedlin MG. Emergency contraceptive pill users’ risk perceptions for sexually transmitted infections and future unintended pregnancy. J Am Assoc Nurse Pract. 2017;29(9):527-534.
- Wilkinson TA, Clark P, Rafie S, et al. Access to emergency contraception after removal of age restrictions. Pediatrics. 2017;140(1):e20164262.
- American College of Obstetricians and Gynecologists. Committee opinion no. 707: Access to emergency contraception. Obstet Gynecol. 2017;130(1):e48-e52.
- Guttmacher Institute. Emergency contraception. March 1, 2022.
- Nurse Practitioners in Women’s Health. Position statement: Expanding access to hormonal contraception. Womens Healthcare. 2019;7(3):16-20.
- Association of Women’s Health, Obstetric and Neonatal Nurses. Position statement: Emergency contraception. J Obstet Gynecol Neonatal Nurs. 2017;46(6):886-888.
- American College Health Association. ACHA guidelines. Best practices for sexual health promotion and clinical care in college health settings. January 2020.
- Caron C. Students look to vending machines for better access to morning-after pill. New York Times. September 28, 2017.
Web resources:
A. CDC – Emergency Contraception – US SPR – Reproductive Health
B. Emergency contraception | Office on Women’s Health (womenshealth.gov)
C. Planned Parenthood | Official Site
D. Unintended Pregnancy in the United States | Guttmacher Institute
E. Contraceptive Use in the United States by Method | Guttmacher Institute
Key words: emergency contraception, contraception, unintended pregnancy
Womens Healthcare. 2022;10(3):21-27. doi:10.51256/WHC062221

