Why has a 30-year-old woman never been able to enjoy pain-free sex?
A 30-year-old woman named CL presents to the office as a new patient. Her health history is notable for dysmenorrhea; irritable bowel syndrome (IBS), diarrhea dominant; and dyspareunia since the onset of sexual activity at age 18. CL has never been pregnant. CL tells the nurse practitioner (NP) that her dysmenorrhea has been well managed since age 15 with combination oral contraceptives (COCs) and an occasional NSAID on day 1 of her period. She does not take any medication for her IBS, and she avoids foods that might cause bouts of diarrhea. CL tells the NP she is seeking relief of the pain she experiences during sex, which has worsened over the past 6 months and which she fears might ruin her current relationship. She tearfully tells the NP that three prior relationships ended badly because she was unable to have comfortable, enjoyable sex. Although she said she was able to tolerate sexual touching and intercourse in the past, her symptoms have been gradually getting worse.
She goes on to tell the NP that she has been dating her current partner for 6 months and that they have had sex 3 times, which caused great discomfort. She says she tried various over-the-counter (OTC) lubricants and different positions, to no avail. Although CL says she enjoys cuddling and kissing with her partner, she has been avoiding this type of contact for the past 2 months because of fear that it would lead to intercourse. At the same time, she wants to enjoy intercourse and please her partner.
What else would be helpful to know about CL’s symptoms and health history?
CL tells the NP she feels she is “a mess down there.” She reports experiencing itching and burning in the vulvar area. The symptoms are present on most days and worsen with her period. She ascribes the symptoms to yeast infections (3-4 in the past year), which she treats with OTC antifungal creams. She also reports that, in the past year, she has been treated by another healthcare provider (HCP) for bacterial vaginosis with a vaginal gel. In addition, she has gone to an urgent care center twice for treatment of urinary tract infections— even though she is unsure whether the urine culture findings were positive or negative.
A year ago, at the suggestion of a friend to whom she mentioned the painful sex, CL went to a clinic for sexually transmitted disease testing and a Pap smear with a human papillomavirus (HPV) co-test. She reports that all findings were negative but adds that she hesitates to “go through that again” because the speculum examination was very painful. She says, “I don’t even use tampons because they hurt too much.” She tells the NP that she wears a panty liner every day because she does not take her COC consistently; when she forgets to take a pill or takes it late, she has some spotting. She remarks that she never feels really clean because of the frequent diarrhea, and uses feminine wipes daily.
As she tears up again, CL tells the NP that, over the past few months, she has felt more stressed than usual because of worries about her relationship and because of work pressures with a new boss. She laughs a little as she says, “I don’t drink, use drugs, or smoke, but when I get home from work, all I want to do is take a long hot bath with some scented oils to help me relax and feel clean.”
The NP seeks more details about the dyspareunia, and asks CL to rate the pain she experiences during and after sexual intercourse using a Likert scale, with 0 = no pain and 10 = maximum pain. CL rates the pain she feels in the vestibule area when her partner’s penis enters the vagina as an 8, and describes it as burning or a “razor blade” sensation. During penile thrusting, she experiences vaginal pain rated as a 6. After sexual touching, she has heightened burning pain in the vestibule area rated as a 4; the pain lasts 12 to 24 hours.
Which differential diagnoses would you consider at this point?
CL describes chronic vulvar itching and burning, significant vestibular pain during and after sex, and vaginal pain with thrusting. These symptoms may have similar or overlapping causes such as:
• Vulvitis;
• Chronic candidiasis or chronic imbalance in vaginal microbiome;
• Vulvar dermatoses (eg, lichen sclerosus);
• Vulvar cancer (pruritus is the most common early symptom);
• Vulvovaginal atrophy (associated with the patient’s prolonged COC use);
• Vaginismus; and
• Vulvodynia.
What would you include in your problem-focused physical examination?
The NP begins with inspection of the vulva and vestibule to assess for anatomic changes or variations, pigmentation changes, lesions, and integrity of vulvar tissues. The inspection reveals normal vulvar structures and mildly erythematous, dry labia majora with no other pigmentation changes or lesions. The labia minora are moist and pink. Vulvar hair is absent; CL reports that she has been removing this hair with a razor and shaving cream for years.
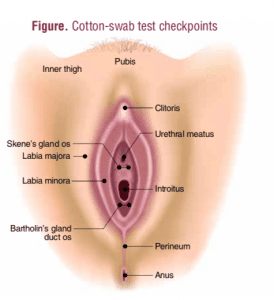 In the absence of abnormal visible findings other than mild erythema of the labia majora, the NP performs a cotton-swab test to specifically localize any areas of altered sensation in the vulvar area. The test is done prior to digital palpation and an attempt at insertion of a speculum. A standard cotton tip applicator is used to apply light touch starting on the upper inner thigh and following in a clockwise fashion in a manner that includes the labia majora, the vestibular duct openings for Skene’s and Bartolin glands, the clitoris/hood, and the perineum. The patient is asked to rate her pain on a scale of 0 to 10 at each location (Figure). CL’s cotton- swab test produces scores of 0 on the inner thighs and labia majora, 2 at the clitoris, and 2 at the perineal body. Pain at the vestibular gland duct openings is rated at 6-7 for the Skene’s glands and 7-8 for the Bartholin glands.
In the absence of abnormal visible findings other than mild erythema of the labia majora, the NP performs a cotton-swab test to specifically localize any areas of altered sensation in the vulvar area. The test is done prior to digital palpation and an attempt at insertion of a speculum. A standard cotton tip applicator is used to apply light touch starting on the upper inner thigh and following in a clockwise fashion in a manner that includes the labia majora, the vestibular duct openings for Skene’s and Bartolin glands, the clitoris/hood, and the perineum. The patient is asked to rate her pain on a scale of 0 to 10 at each location (Figure). CL’s cotton- swab test produces scores of 0 on the inner thighs and labia majora, 2 at the clitoris, and 2 at the perineal body. Pain at the vestibular gland duct openings is rated at 6-7 for the Skene’s glands and 7-8 for the Bartholin glands.
Because CL has high vestibular pain scores, the NP does not perform a speculum exam. The NP uses a cotton swab to obtain a vaginal specimen for pH and microscopic wet mount evaluation. The pH is 4.0. A whiff test is negative. Mature squamous epithelial cells are present, with no pseudohyphae, clue cells, or motile trichomonads seen on the wet mount. A vaginal specimen is sent for fungal culture to assess for infection caused by atypical Candida species. The culture results are negative.
Pelvic floor muscle (PFM) assessment is done using one finger inserted into the vagina without touching the vestibule. The Oxford Grading System uses a 6-point scale to measure PFM strength: 0 = no contraction; 1 = flicker; 2 = weak; 3 = moderate; 4 = good (with lift); and 5 = strong. CL’s PFM strength is rated a 4. Palpation of the levator ani group and obturator internus muscles reveals hypertonicity and tenderness.
Based on the history and exam findings, what is the diagnosis?
CL has three interconnected diagnoses contributing to the dyspareunia: contact vulvitis, vulvodynia, and vaginismus.
Vulvitis may be caused by chronic or recurrent infection by pathogens such as Candida, herpes simplex virus, or HPV; contact with allergens or irritants; or injury/ trauma. CL has reported regular use of vulvar contact irritants such as panty liners, feminine wipes, shaving cream, and scented bath oils. Pubic hair shaving may heighten one’s sensitivity to contact irritants. CL’s physical exam and wet mount findings tend to rule out infection as the cause of the vulvitis. The labia majora dryness may be related to prolonged use of COCs.
Vulvodynia is defined as vulvar discomfort (usually described as burning pain) occurring in the absence of relevant visible findings or a specific, clinically identifiable, neurologic disorder.1 It is a diagnosis of exclusion when all other potential causes have been ruled out and when symptoms persist longer than 3 months.1 The etiology of vulvodynia is unknown but may be related to genetic susceptibility, chronic inflammation, a combination of factors (eg, PFM abnormalities, neuropathic pain, anxiety, primary/secondary sexual dysfunction), regionally elevated cytokines produced by vulvar vestibule-specific fibroblasts, hormonal changes, or dietary oxalates.2-8 Chronic inflammation related to the contact irritants, recurrent infections, hormonal changes, or chronic skin conditions acts as a trigger. Normal sensations are perceived as abnormal, resulting in heightened sensitivity.
Estimates of vulvodynia prevalence range from 3% to 18%.9 Onset is most likely to occur between the ages of 18 and 25 years. Among symptomatic women, 60% see an average of three HCPs before receiving the correct diagnosis and 40% remain undiagnosed.9 Vulvodynia is classified as localized or generalized. Localized vulvodynia is subdivided into primary, in which vestibular pain begins during the first attempt at vaginal penetration, or secondary, in which pain occurs after a period of pain-free sex. Generalized vulvodynia may include all of the vulva or be limited to one side, with pain occurring in the absence of a triggering event. Of note, about 10% of women with generalized vulvodynia have a coexisting pain syndrome such as interstitial cystitis/painful bladder syndrome, IBS, fibromyalgia, or chronic fatigue syndrome.10
Prolonged use of COCs as a risk factor for vulvodynia is controversial. Several studies have supported the theory that use of COCs or progesterone-only contraceptives in females younger than age 18 causes down-regulation of estrogen receptors, leading the vestibular epithelium to become thin and fragile.11,12 Although some women with vulvodynia have increased perception of pain when taking COCs, stopping COCs does not necessarily lead to resolution of the symptoms.12
Vaginismus is defined as recurrent or persistent involuntary spasm of the musculature of the outer third of the vagina that interferes with vaginal penetration and that causes personal distress or interpersonal difficulties.13
What can the NP offer CL as first-line treatment?
Because studies on the cause and treatment of vulvodynia are limited, the American College of Obstetricians and Gynecologists recommends that therapy be based on evidence from descriptive studies, expert reports, and clinical experience— with the understanding that the condition is difficult to treat and that no single approach is successful for all women.14
Individualized treatment usually involves multiple therapies over time.9 Treatment is considered in a step-wise progression, starting with self-management and moving upward on the scale of complexity. Total symptom resolution can be unrealistic. Primary goals are symptom reduction, return of satisfactory sexual function, and improved quality of life. Patient education concerning the manageability yet chronicity of this neuropathic condition is essential in setting realistic goals and in instituting a treatment plan that leads to improvement and satisfaction.
Mental health counseling is an important component. Approximately 50% of women with vulvodynia have a concordant diagnosis of anxiety.15 An increased occurrence of childhood physical/sexual abuse in women with vulvodynia has been reported.16 Referral to a mental health specialist with expertise in women’s sexual health and chronic pain conditions can be helpful.
Self-management is essential in CL’s case. She is educated to avoid use of feminine hygiene wipes/washes, panty liners (at times other than menses), and repetitive use of OTC antifungals.9 She is encouraged to stop removing her vulvar hair, which provides a protective barrier for sensitized vestibular tissues. She is advised to avoid exposure to very hot water in bathtubs and hot tubs, which can exacerbate vestibular discomfort. Application of unscented, hypoallergenic emollients to the vulvar skin and sitz baths in lukewarm water can be soothing, however.
CL and her partner should explore alternatives to penetrative sex (eg, light physical contact with her genitalia). As pain diminishes with treatment over time, CL can consider restarting penetrative sex. The NP advises her to implement strategies for PFM relaxation, to adopt positions during intercourse that minimize pressure on
sensitive areas, and to use liberal amounts of water-soluble lubrication.
A low-oxalate diet has been suggested to reduce high levels of oxalate in urine. However, little evidence supports the efficacy of this dietary modification in reducing vulvar pain.7,8
What types of pharmacologic agents are appropriate?
Topical medications can be used on a short-term basis.17 Options include lidocaine 5% ointment, doxepin 5% cream in water-soluble base, gabapentin 2%-6% in watersoluble base, and amitriptyline 2% with baclofen 2% in water-soluble base. When topicals are used, those with an ointment base, rather than a cream base, are preferred.17 Cream bases contain more preservatives and stabilizers, which can act as contact irritants and cause burning on application.
The next line of treatment is the use of oral neuropathic pain modulators, either antidepressants (eg, amitriptyline, desipramine, venlafaxine) or anticonvulsants (eg, gabapentin, pregabalin, lamotrigine, topiramate).17 Side-effect profiles and patient tolerance drive dosing regimens. Pruritus can be managed with an antihistamine such as hydroxyzine or cetirizine. In addition, nerve blocks, topical nitroglycerin, topical capsaicin, interferon injection, and trigger point injections have shown some efficacy in treating vulvodynia.17 Vaginal valium, injectable botulinum toxin, and topical baclofen have been used in the treatment of vaginismus.
What role does physical therapy play in CL’s treatment?
Pelvic floor physical therapy (PT) is an essential component of treatment for CL once she can withstand cotton-swab touch with lowered vestibular pain scores. A person with expertise in pelvic floor PT may use a combination of interventions to decrease CL’s dyspareunia and to improve her sexual function.17,18 Interventions include biofeedback, which can help CL learn to relax her PFMs; external soft tissue mobilization and myofascial release techniques; trigger point pressure; and transcutaneous electrical stimulation applied to the sacral nerve. The physical therapist or NP may teach CL how to use vaginal dilators to gradually overcome the tension in the PFMs. When CL is ready to resume sexual activity, she can use the dilator to prepare herself for and facilitate penetration.
When is surgery an option?
For women who continue to experience intractable symptoms after all other treatments have been tried, vestibulectomy—excision of the vestibule with vaginal advancement—is an option. Patient selection is critical for success; women can consider this option only if symptoms are confined to the vestibule. Success rates range from 60% to 85%.19,20 Referral to a pelvic and reconstructive surgeon is advised in these cases.
Reflection for practice
Vulvodynia is a complex chronic pain condition. The interrelated physical, psychological, and psychosexual components make management challenging for both clinician and patient. CL has sought an NP’s help in seeking relief of her dyspareunia. The treatment plan focuses on symptom reduction to allow for sexual functioning that provides intimacy and satisfaction. Total relief of pain may not be achievable. A multidisciplinary approach to treatment may produce the best results. In fact, when available, referral to vulvovaginal disease specialists can facilitate an individualized, coordinated, comprehensive approach to treatment.
Susan Hoffstetter is a Fellow in the International Society of the Study of Vulvovaginal Diseases and a Professor at Saint Louis University School of Medicine in St. Louis, Missouri. The author states that she does not have a financial interest in or other relationship with any commercial product named in this article.
References
Haefner HK. Report of the International Society for the Study of Vulvovaginal Disease terminology and classification of vulvodynia. J Low Genit Tract Dis. 2007;11(1):48-49.
Goldstein AT, Kim N, Burrows LJ, Goldstein I. Genetic differences may reflect differences in susceptibility to vulvodynia in general or in spontaneous remission propensity: a response. J Sex Med. 2015;12(2):578-579.
Akopians AL, Rapkin AJ. Vulvo- dynia: the role of inflammation in the etiology of localized provoked pain of the vulvar vestibule (vestibulodynia). Semin Reprod Med. 2015;33(4):239-245.
Edwards L. Vulvodynia. Clin Obstet Gynecol. 2015;58(1):143-152.
Foster DC, Piekarz KH, Murant TI, et al. Enhanced synthesis of proinflammatory cytokines by vulvar vestibular fibroblasts: implications for vulvar vestibulitis. Am J Obstet Gynecol. 2007;196(4):346e1-8.
Eva LJ, MacLean AB, Reid WM, et al. Estrogen receptor expression. Am J Obstet Gynecol. 2003;189(2):458-461.
Greenstein A, Militscher I, Chen J, et al. Hyperoxaluria in women with vulvar vestibulitis syndrome. J Reprod Med. 2006;51(6):500-502.
Harlow BL, Abenhaim HA, Vitonis AF, Harnack L. Influence of dietary oxalates on the risk of adult onset vulvodynia. J Reprod Med. 2008; 53(3):171-178.
Harlow BL, Stewart EG. A population based assessment of chronic unexplained vulvar pain: have we underestimated the prevalence of vulvodynia? J Am Med Womens
Assoc. 2003;58(2):82-88.
Reed BD, Harlow SD, Sen A, et al. Relationship between vulvodynia and chronic comorbid pain conditions. Obstet Gynecol. 2012;120(1):145-151.
Bohm-Starke N, Johannesson U, Hilliges M, et al. Decreased mechanical pain threshold in the vestibular mucosa of women using oral contraceptives: a contributing factor in vulvar vestibulitis? J Reprod Med. 2004;49(11):888-892.
Goldstein AT, Krapf J, Belken Z. Do oral contraceptive pills cause vulvodynia? Time to finally end the controversy. International Pelvic Pain Society Blog. October 2015. pelvic pain.org/professional/blog/ipps-blog/june-2014/do-oral-contraceptive-pillscause-vulvodynia-time.aspx.
Curtis M, Linares S, Antoniewicz L. Glass’ Office Gynecology. 7th ed. Philadelphia, PA: Wolters Kluwer Health; 2014.
American College of Obstetricians and Gynecologists. 2006. Committee Opinion Number 345. Vulvodynia. Washington, DC: American College of Obstetricians and Gynecologists; 2006.
Tribo MJ, Andio O, Ros S et al. Clinical characteristics and psycho-
pathological profile of patients with vulvodynia: an observational and descriptive study. Dermatology. 2008;216(1):24-30.
Harlow BJ, Stewart EG. Adult onset vulvodynia in relation to childhood violence victimization. Am J Epidemiol. 2005;161(9):871-880.
Haefner HK, Collins ME, Davis GD, et al. The vulvodynia guideline. J Low Genit Tract Dis. 2005;9(1):40-51.
DeBevoise TM, Dobinsky AF, Mc-Curdy Robinson CB, et al.
Pelvic floor physical therapy: more than Kegels. Womens Healthcare. 2015;3(2):34-41.
McCormack WM, Spence MR. Evaluation of surgical treatment of vulvar vestibulitis. Eur J Obstet Gynecol Reprod Biol. 1999;86(2):135-138.
Haefner HK. Critique of new gynecologic surgical procedures: surgery for vulvar vestibulitis. Clin Obstet Gynecol. 2000;43(3):689-700.
Web resource
npwomenshealthcare.com/authorinstructions/
We invite readers to submit interesting and elucidating case reports. Please see our Guidelines for Authors for more information about this short-form article option.

