Clostridioides difficile (C. difficile), commonly known as C. diff, is a bacteria associated with severe diarrhea and inflammation of the colon. The Centers for Disease Control and Prevention estimates almost 500,000 C. difficile infections (CDIs) in the United States annually.1 Approximately 1 in 6 patients who are positive for C. difficile will be reinfected over the next 2 to 8 weeks.1 One of the most known risk factors for CDI is recent antibiotic use. The risk for CDI is heightened with prolonged antibiotic use or using two or more antibiotics.2,3 Pregnant women who have previously been treated with broad-spectrum antibiotics, such as penicillins, cephalosporins, and fluoroquinolones, for an infection (ie, a urinary tract infection) are at increased risk of developing CDI.2,4,5 However, there are other general risk factors that should be considered, which include previous infection with C. difficile, a compromised immune system, a recent stay within a hospital or nursing home, or age older than 65 years.1
In pregnancy, the prevalence of CDI is believed low, with a recent rate of infection during pregnancy noted of 0.07-0.7/1,000 births.4 However, the rates of CDI in pregnancy are trending upward.6,7 Risk factors such as advanced maternal age (> 35 years), treatment with antibiotics, and presence of an inflammatory bowel disease (eg, ulcerative colitis or Crohn’s disease) are associated with increased rates of infection.4 An original research article published by the American College of Obstetricians and Gynecologists in 2018 identified smoking, multiple gestations, pneumonia, pyelonephritis, Caesarean section, and perineal wound infections as additional risk factors for CDI during pregnancy.6 The morbidity and mortality associated with CDI is significant and, as women’s health clinicians, we must maintain clinical suspicion for this infection and be astute to subjective findings and the management and treatment plans for this condition.
Assessment findings for C. difficile in pregnancy are not unique from the general population. Symptoms may include fever, severe diarrhea, nausea, loss of appetite, or abdominal tenderness or pain.1 Differentials for CDI include Crohn’s disease, ulcerative colitis, irritable bowel syndrome (IBS), malabsorption, as may be seen in celiac disease, salmonella infection, shigellosis, viral gastroenteritis, diverticulitis, peritonitis, and abdominal sepsis.8 Patients with CDI may have elevated white blood cell counts and an increased serum creatinine level if there is associated acute kidney injury.
Case description
VL, a 21-year-old woman at 32 weeks’ gestation, presented for a routine prenatal appointment at an advanced practice nurse-led community clinic with a complaint of persistent diarrhea that initially presented 2 weeks ago, was worsening over the past 3 days, and interrupting daily activities. She was unable to work due to urgent and unpredictable episodes of diarrhea that occurred 6 to 10 times a day. She noted that every time she ate or drank, she would have acute postprandial diarrhea. She was drinking an electrolyte sports drink throughout the day hoping it would help. VL denied fever, nausea, vomiting, malaise, anorexia, and blood or mucous in the stool. She affirmed abdominal cramping before each bowel movement, but no consistent pain, bloating, or abdominal distention. Diarrhea was described as green-tinted and mostly liquid with a foul odor. Even though all family members were sharing the same bathroom, no one else was ill or having similar symptoms. VL denied known exposure to food poisoning or gastrointestinal viruses but had cleaned up vomit without available personal protective equipment (PPE) at a gas station where she worked. VL reported good fetal movement throughout her illness. There were no hospitalizations or antibiotic use during this pregnancy. Her two previous pregnancies were delivered via low transverse Caesarean sections. VL was not currently taking any prescription medications including her prenatal vitamins as they made her feel ill. She was taking over-the-counter bismuth subsalicylate every hour or two without relief. A physical examination revealed weight 243 lbs/110.2 kg, body mass index 43.05, blood pressure 128/72 mm Hg, pulse 117, temperature 98.9°F/37.2°C, pink and moist mucous membranes, lower bilateral abdominal tenderness, and negative rebound tenderness or guarding. Fetal heart tones were 145 beats per minute and fundal height was 34 cm.
Diagnosis and treatment
Diagnosis
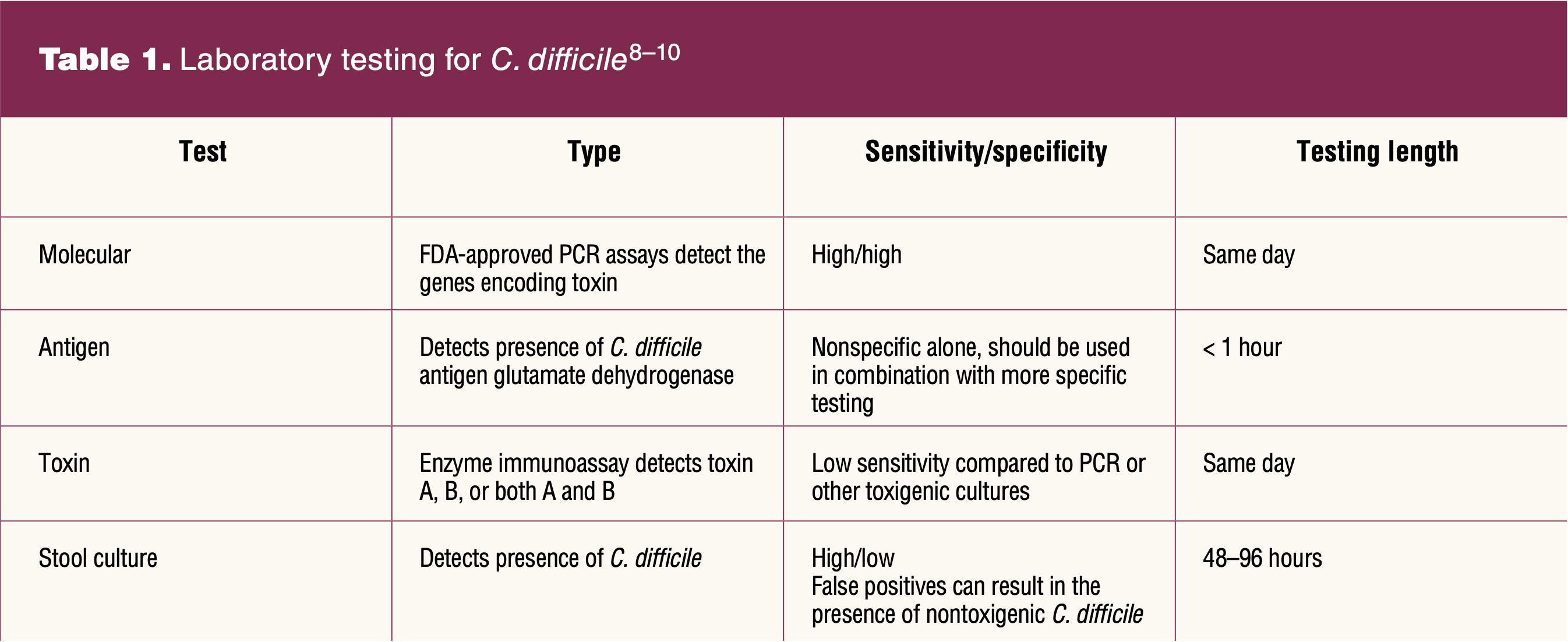
Patients with a new onset of unexplained diarrhea of 3 or greater loose stools in 24 hours should be tested for CDI.8 Testing should not be repeated within 7 days during the same episode of diarrhea and should not be performed on asymptomatic patients or those taking laxatives.8,9
There are four main laboratory test options to identify C. difficile: molecular tests, antigen detection, toxin tests, and stool culture (Table 1).9 Each test has varying sensitivity and specificity for C. difficile bacteria and varies in time lengths for results. Clinicians should check with their local lab facility to determine the institutional criteria for patient stool submission and testing. Of note, C. difficile is unstable and degrades quickly, which may lead to undetectable bacteria levels 2 hours or more after collection.9 Stool samples should be promptly tested or refrigerated until testing can be completed.9
Treatment
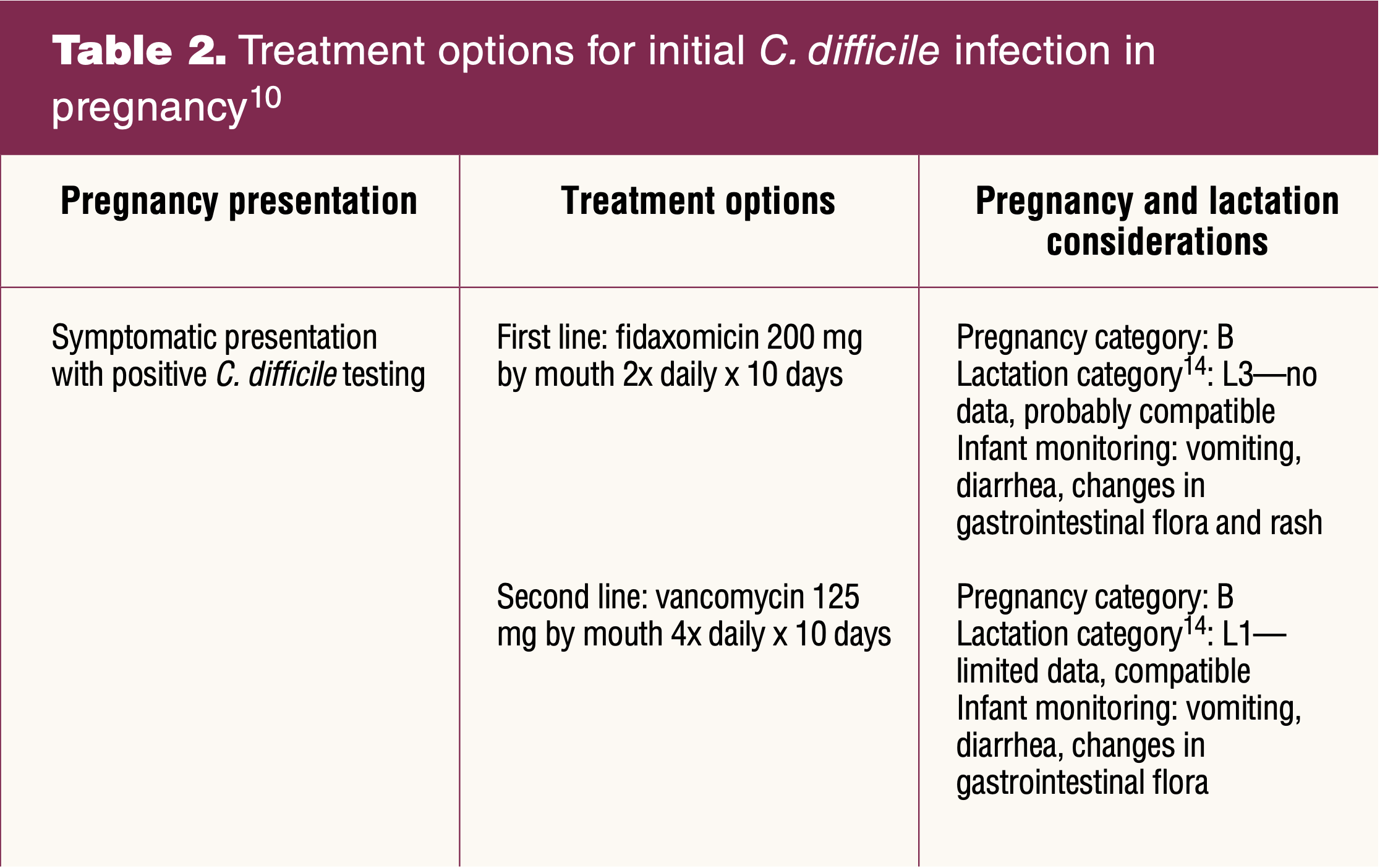
Treatment recommendations for initial and recurrent CDI are based on the most recent 2021 clinical guidelines by IDSA and SHEA, available online at academic.oup.com/cid/article/73/5/e1029/6298219A. Initial CDI is when a patient presents symptomatically, has a positive C. difficile test result, and has not been previously treated for a CDI (Table 2). Recurrence is any subsequent symptomatic presentation of diarrhea (≥ 3 liquid stools in 24 hours) after treatment completion (Table 3). If patients are taking an antibiotic on initial diagnosis, it is important to stop the current antibiotic to prevent recurrence.2,10 Previously, vancomycin was recommended as first-line treatment, but based on randomized controlled trial data, fidaxomicin reduces recurrent episodes of CDI and is the current recommended first-line treatment.10
Case diagnosis, treatment, and education
A stool culture was ordered in accordance with institutional policies and the result was positive for CDI. A recent white blood cell count was slightly elevated at 15.2 cells/μL. No creatinine levels were available for review. Because VL was seen prior to the publication of the most recent guidelines, she was treated with vancomycin 125 mg by mouth four times daily for 10 days.8 She was instructed to take the medication as directed and to finish all 10 days. VL was advised to return to the ob/gyn clinic if her diarrhea worsened, did not improve, or recurred after 10 days.
Patient education
VL was provided education on the spread of C. difficile and how to keep her family safe and reduce risk of reinfection. The most important preventive measure is handwashing with soap and water throughout the day, before eating, and after toileting.11 C. difficile is spread by skin-to-skin and skin-to-contaminated surface contact.11 Using separate bathrooms is advisable, but as VL only had one bathroom in her home, she was provided education on cleaning the bathroom after each use. Effective cleaning measures to kill C. difficile are using a mixture of 1 part bleach to 9 parts water.11 If a patient has a graduated measuring cylinder with ounces and milliliters, they can use 100 mL of bleach to 900 mL of water. High-touch surfaces, like doorknobs, refrigerator handles, toilet handles and seats, and countertops, should be cleaned often.11 Electronics and remote controls should not be shared if possible as bleach may damage these items.11 It is recommended to not share linens, towels, and clothing, and these items should be washed on the hottest water setting, with bleach if items are bleach safe, before others use them.11
Education on urgent and emergent concerns in pregnant women include increasing or nonresolving fever, increasing abdominal pain and tenderness, increased malaise, dizziness, rapid heartbeat, shortness of breath, and decreased fetal movement. Pregnant patients should be advised to go to labor and delivery triage or the emergency room based on gestational age for worsening symptoms.
Fetal implications
Specific risk to the fetus via maternal CDI has not been well documented in the literature. It is well documented that systemic manifestations of CDI pose a risk to pregnant women and thereby also the fetus.5,12 Maternal complications of untreated or resistant CDI include toxic megacolon, renal failure, shock, sepsis, and maternal death.5,6 Although current literature does not clearly outline direct fetal effects from maternal CDI, fetal deaths have resulted due to maternal complications from CDI.5,13
Collaboration
Collaboration is an important aspect of healthcare and team management and for pregnant patients with medical complications affecting their pregnancy. An advanced practice nurse (nurse practitioner, nurse-midwife) should consult with an obstetrician, physician who specializes in obstetric care, or maternal-fetal medicine specialist for any pregnant patient suspected of having CDI. Maternal-fetal medicine physicians were consulted for the management of this patient.
Case resolution
VL had full resolution of symptoms after completing vancomycin and remained asymptomatic for the remainder of her pregnancy. She delivered a vigorous term infant by scheduled repeat low transverse Caesarean section at 39 weeks, and her postpartum course and newborn care was unremarkable.
Conclusion
C. difficile infection is on the rise in pregnant women and can negatively impact both pregnant women and fetuses.6,7 Clinicians should have a high suspicion for CDI in pregnant women with unexplained diarrhea lasting 3 days or longer even in the absence of recent antibiotic use. Pregnant patients who present with unexplained diarrhea need prompt testing and rapid treatment of CDI to prevent deleterious maternal and fetal effects due to worsening or prolonged symptoms. Women’s health nurse practitioners and certified nurse-midwives are frontline providers who can recognize, diagnose, and treat patients with CDI. Collaboration with obstetricians, physicians who specialize in obstetric care, or maternal-fetal medicine specialists is ideal because a team approach facilitates comprehensive treatment and continuity of care if the patient needs further testing, imaging, or treatment for severe or complicated CDI.
Angela D. Alston is Assistant Professor of Clinical Nursing, lead faculty-women’s health nurse practitioner specialty track, and Chief Diversity Officer at The Ohio State University College of Nursing in Columbus, Ohio. Laura J. Valle is Assistant Professor of Clinical Practice in the midwifery and women’s health nurse practitioner specialty tracks at The Ohio State University College of Nursing in Columbus, Ohio, and is the owner practitioner of Willow Women’s Health in Dayton, Ohio. The authors have no actual or potential conflicts of interest in relation to the contents of this article.
References
1 Centers for Disease Control and Prevention. What is C. diff? Reviewed July 20, 2021. https://www.cdc.gov/cdiff/what-is.html.
2 Crowther G, Wilcox MH. Antibiotic therapy and clostridium difficile infection – primum non nocere – first do no harm. Infect Drug Resist. 2015;8:333-337.
3 Stevens V, Dumyati G, Fine LS, et al. Cumulative antibiotic exposures over time and the risk of clostridium difficile infection. Clin Infect Dis. 2011;53(1):42-48.
4 Meda M, Virgincar N, Gentry V, et al. Clostridium difficile infection in pregnant and postpartum women in 2 hospitals and a review of literature. Am J Infect Control. 2019;47(1):e7-e14.
5 Rouphael NG, O’Donnell JA, Bhatnagar J, et al. Clostridium difficile-associated diarrhea: an emerging threat to pregnant women. Am J Obstet Gynecol. 2008;198(6):635.e1-635.e6.
6 Ruiter-Ligeti J, Vincent S, Czuzoj-Shulman N, Abenhaim HA. Risk factors, incidence, and morbidity associated with obstetric clostridium difficile infection. Obstet Gynecol. 2018;131(2):387-391.
7 Saha S, Pardi R, Theiler RN, et al. Incidence of Clostridioides difficile infection in peripartum women: a retrospective cohort study. Therap Adv Gastroenterol. 2020;13:1-5.
8 McDonald LC, Gerding DN, Johnson S, et al. Clinical practice guidelines for Clostridium difficile infection in adults and children: 2017 update by the Infectious Diseases Society of America (IDSA) and Society for Healthcare Epidemiology of America (SHEA). Clin Infect Dis. 2018;66(7):e1-e48.
9 Centers for Disease Control and Prevention. FAQs for clinicians about C. diff. Reviewed July 20, 2021. https://www.cdc.gov/cdiff/clinicians/faq.html.
10 Johnson S, Lavergne V, Skinner AM, et al. Clinical practice guideline by the Infectious Diseases Society of America (IDSA) and Society for Healthcare Epidemiology of America (SHEA): 2021 focused update guidelines on management of Clostridioides difficile infection in adults. Clin Infect Dis. 2021;73(5):e1029-e1044.
11 Centers for Disease Control and Prevention. Prevent the spread of C. Diff. Information for patients. Reviewed July 20, 2021. https://www.cdc.gov/cdiff/prevent.html.
12 Cózar-Llistó A, Ramos-Martinez A, Cobo J. Clostridium difficile infection in special high-risk populations. Infect Dis Ther. 2016;5(3):253-269.
13 de Curraize C, Rousseau C, Corvec S, et al. Variable spectrum of disease and risk factors of peripartum Clostridium difficile infection: report of 14 cases from French hospitals and literature review. Eur J Clin Microbiol Infect Dis. 2018;37(12):2293-2299.
14 Hale TW. Medications & Mothers’ Milk, 19th ed. New York, NY: Springer Publishing Company; 2021.