Vulvovaginal candidiasis (VVC) is a common mucosal infection that is primarily caused by the fungus Candida albicans. When VVC is recurrent it can be a debilitating condition, characterized by itching, burning, pain, vaginal discharge, external dysuria, and dyspareunia that can result in distress and diminished health-related quality of life. In this article, we provide an update of the latest information on the impact, diagnosis, and treatment for women with acute and recurrent VVC.
Key words: antifungal, azoles, Candida albicans, non-albicans Candida, recurrent, vulvovaginal candidiasis
Vulvovaginal candidiasis (VVC) is a common mucosal infection that is primarily caused by the fungus Candida albicans. The normal vaginal microbiome is characterized by Lactobacillus species and other fermentative bacteria, mostly anaerobes, which exist in a well-balanced relationship to maintain a normal vaginal environment.1,2 In particular, Lactobacillus species maintains acidification of the vagina within a pH range of 3.0 to 4.5 that protects against colonization with opportunistic pathogens.1 Candida species are often part of the genital microbiome and can be found in the vaginal tracts of up to 70% of healthy asymptomatic women.2 Vulvovaginal candidiasis occurs when Candida species integrate into the vaginal epithelium causing an inflammatory response leading to a variety of symptoms including vaginal discharge, irritation, burning, swelling, itching, external dysuria, and dyspareunia. Factors associated with an increased risk for VVC include pregnancy, uncontrolled diabetes, immune system dysfunction, hygiene, genetics, sexual behavior, and use of antibiotics, SGLT [sodium-glucose cotransporter-2] inhibitors, immunosuppressant drugs, oral contraceptives, intrauterine devices, or vaginal estrogen.3
VVC infections can be categorized as complicated or uncomplicated. Uncomplicated infections are those occurring infrequently, are associated with mild-to-moderate symptoms in healthy women, and almost always are caused by C. albicans.4 Complicated infections are those associated with any of the following: severe symptoms, occurring four times per year or more, occurring in immunocompromised women, or non-albicans Candida species. Immunocompromised women include those with uncontrolled diabetes, autoimmune diseases, or AIDS/HIV, and those taking immunomodulating drugs such as steroids.2,4–7 Recurrent VVC is defined as four or more episodes annually with each recurrence considered an acute episode requiring treatment of the active infection.7,8 In more than half of women experiencing recurrent VVC, no clear risk factor can be identified.5,9 Recurrent VVC can be especially difficult to manage because the pathogen is more often a non-albicans Candida species.5, 9–11
This article provides an update of the latest information on the impact, diagnosis, and treatment for women with both acute and recurrent VVC.
Impact of VVC
VVC results in an estimated 1.4 million US outpatient visits each year.12 From 2005 to 2014, direct US healthcare costs related to VVC were an estimated $368 million per year.3,13,14 VVC is estimated to account for up to $14.4 billion US dollars annually of lost economic productivity.6
Worldwide, recurrent VVC is estimated to affect 138 million women annually and 372 million women over the course of their lifetime. By 2030, an estimated 158 million women will be affected by recurrent VVC annually.6 The impact of recurrent VVC goes beyond reduced economic productivity and the burden on healthcare resource utilization, to include a significant loss in health-related quality of life. Symptoms that persist despite treatment or frequently recur can create a debilitating condition.5,9–11
A survey of women in Europe and the United States found that depression and anxiety occurred in 68% and 54%, respectively, in more than 12,000 women with recurrent VVC during and outside of episodes, compared to less than 20% in the general population (P < .001).10 Compared with healthy controls, women with recurrent VVC scored significantly worse on measures of quality of life, including general health, mental health, physical function, bodily pain, social functioning, dyspareunia, and emotional wellbeing.11
Diagnosis
Unfortunately, high rates of self-diagnosis and self-treatment for VVC result in approximately half of women receiving inadequate treatment.15 Recently, in the midst of the Covid-19 pandemic, an increasing use of telephone triage, rather than in-person visits to a healthcare provider, has limited the use of proper physical examination and diagnostic procedures for diagnosing VVC. Even when patients are seen by a healthcare provider, they may not receive sufficient diagnostic testing, which often results in prescribing inappropriate treatment with poor outcomes. In a survey of provider awareness of guidelines for the diagnosis of vaginitis and a chart review of women with vaginitis, only 66% of patients with a documented diagnosis of VVC received a guideline-recommended workup.16 Providers reported that access to point-of-care tools (microscope, pH test strips, potassium hydroxide solution) to diagnose vaginitis was limited.16
Most often, an accurate diagnosis of VVC can be made when the healthcare provider combines a problem-focused health history and physical examination along with point-of-care vaginal pH testing and wet mount microscopy. Symptoms of VVC include mild-to-severe itching, burning, pain, vaginal discharge, dyspareunia, external dysuria, and vulvar edema. Typical signs that may be observed during the clinical exam of the vulva and vagina include edema, erythema, thick white curdy discharge, and in more severe cases, fissures and excoriations.2 Vaginal pH testing will confirm the normal range of 3.5 to 4.5. Identification of budding yeast, spores, and/or branching pseudohyphae on microscopic examination of a vaginal specimen in 10% potassium hydroxide solution is diagnostic for C. albicans (Table 1).17
However, the utility of vaginal microscopy in the clinical setting may be limited by lack of equipment, lack of proper training, inconsistent interpretation of results, and low sensitivity for VVC (40%–70%).15,18,19 Contributing to the low sensitivity, some non-albicans species of Candida do not form pseudohyphae or hyphae and may not be recognized on microscopy. Given these limitations, healthcare providers might consider nucleic acid amplification tests (NAATs) recommended as an option in guidelines for the diagnosis and treatment of VVC and increasingly available.7,8,17
The sensitivity and specificity of NAAT for the Candida species group is 91.7% and 94.9%, respectively.18,19 The sensitivity and specificity for identifying C. glabrata separately is 84.7% and 99.1%, respectively.18,19 The availability of a NAAT panel that identifies the most common Candida species and the microorganisms causing bacterial vaginosis and trichomoniasis offers an advantage over culture in determining appropriate therapeutic options.20 Culture of a vaginal specimen remains an option in women with symptoms of recurrent or resistant VVC to confirm the diagnosis and determine if caused by a non-albicans Candida species, or for symptomatic women with negative findings on microscopy when VVC is suspected.7,15,17
Treatment approach
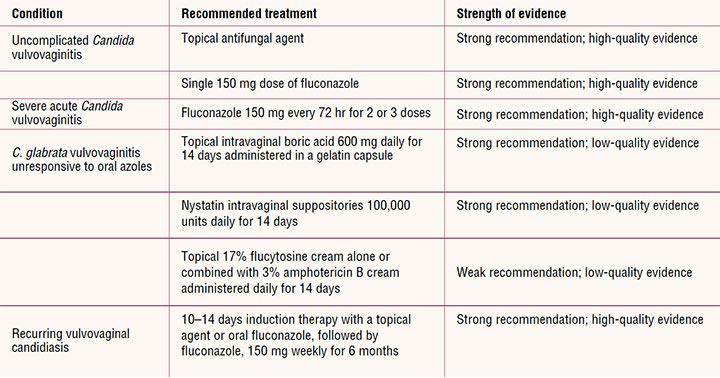
All US Food and Drug Administration (FDA)-approved currently recommended drug therapy for uncomplicated and complicated VVC belong to the azole class of antifungals. Treatment with different products within this class is equally effective, and products vary only by the route of administration and duration of treatment (Table 2).17 Azoles inhibit the synthesis of ergosterol, a main component of the fungal cell membrane, which inhibits growth of Candida.21 Azoles have a high success rate for relieving symptoms associated with uncomplicated infection. Relief of symptoms and negative cultures occur in 80% to 90% of patients who complete treatment.7
Table 2. Summary of IDSA treatment guidelines for VVC17
The most common side effects with topical azole medications are burning and irritation at the site, headache, and nausea. The most common side effects for oral fluconazole are headache, nausea, abdominal pain, diarrhea, dyspepsia, and dizziness. Serious adverse and systemic allergic reactions are rare.22 A thorough medication history is important prior to use of fluconazole as it is associated with several clinically significant drug–drug interactions.22 When using fluconazole, providers should avoid coadministration with other drugs known to prolong the QTc interval.22 Patients receiving long-term fluconazole may also experience photosensitivity.22
For uncomplicated VVC in nonpregnant women, treatment recommendations include a topical azole agent for 1 to 7 days depending on the product or a single 150-mg oral dose of fluconazole.17 For severe acute VVC, 7 to 10 days of a topical azole or oral fluconazole 150 mg given every 72 hours for a total of 2 to 3 doses is recommended.7,8,17
For recurrent VVC, a maintenance regimen that begins with 10 to 14 days of a topical azole or an initial dose of oral fluconazole, followed by fluconazole 150 mg weekly for 6 months may be considered.7,17 Oral fluconazole is effective as preventive therapy in most women with recurrent VVC. However, management is complicated by the fact that 50% of women will have another clinical episode after maintenance therapy is discontinued.7,17,21,23,24 Because of the fungistatic rather than fungicidal nature of azoles, overgrowth can recur, causing continued symptoms and the emergence of azole-resistant Candida species.2,21 Currently the rate of resistance to azoles among Candida species is low, but resistance increasingly is reported in women with recurrent VVC.25 In a recent study of women with recurrent VVC due to C. albicans who were treated with oral fluconazole, resistance was identified in 7.5% who completed at least 6 months of therapy.26 The incidence of resistance to topical azoles is unknown because of the difficulty in studying and quantifying rates.
Azole treatment failures for VVC may be associated with azole-resistant non-albicans Candida species, Candida biofilm formation, and inadequate azole concentration in the vaginal tissue.27–29 New non-azole antifungals to treat acute and recurrent VVC are currently under review by the FDA.
Treatment of VVC due to non-albicans Candida
Approximately 5% to 10% of VVC is caused by non-albicans Candida, most often C. glabrata. C. glabrata and other non-albicans Candida may account for as much as 20% of recurrent VVC.7,30 Other vaginal non-albicans Candida species identified include C. krusei, C. parapsilosis, C. lusitaniae, C. famata, C. tropicalis, and C. dublinesis.30 Women with VVC due to non-albicans Candida may have less severe symptoms compared to those women with VVC caused by C. albicans, although the persistence of symptoms has an overall impact on their quality of life. VVC due to non-albicans Candida is more common in women with underlying medical conditions, such as diabetes and obesity, and in those with chronic VVC.31 Limited treatment options are available for VVC caused by non-albicans Candida as they are less responsive to azole treatment.7,23,32,33 Current treatment recommendations consist of extended 7 to 14 days therapy with a topical azole. Off-label approaches to address symptoms and attempt mycologic suppression may be considered if azoles are not effective.7 Compounded 600 mg intravaginal boric acid gelatin capsules once daily for 2 weeks has demonstrated clinical and mycologic eradication rates of approximately 70%.7 Other suggested therapies for non-albicans Candida include nystatin intravaginal suppositories, or 17% flucytosine cream alone or in combination with 3% amphotericin B.8,17,31,34 Whereas the risk of VVC increases during pregnancy, treatment options are limited.7,35 The Centers for Disease Control and Prevention recommends only
topical azoles applied for 7 days for use in pregnant women.7 Case reports describe a potential association of congenital anomalies in infants exposed in utero to high-dose fluconazole (400–800 mg/day) during the first trimester. Epidemiologic studies suggest a potential risk of spontaneous abortion and congenital abnormalities in infants exposed in utero to 150 mg of fluconazole as a single or repeated dose in the first trimester. There are no adequate and well-controlled studies in humans of the use of fluconazole in pregnancy.22
Nystatin intravaginal suppositories should be used with caution, as there are no adequate and well-controlled studies in humans.34 Flucytosine use is contraindicated during pregnancy.17
Conclusion
VVC, whether acute or recurrent, can have a significant impact on women’s lives. Many women self-diagnose and self-treat leading to delays in symptom resolution. Providing an accurate diagnosis and efficacious treatment in addition to educating women about this common condition is essential. A problem-focused health history and physical examination along with vaginal pH, wet mount microscopy, and/or NAAT provide the tools needed to make the diagnosis of VVC. Accurate diagnosis is critical to choosing treatment regimens to achieve symptom relief. Current oral and topical medications for the treatment of VVC are predominantly within the azole class. These drugs are highly effective with good safety profiles in the treatment of most uncomplicated VVC in nonpregnant women. Optimal treatment for severe, recurrent, or non-albicans Candida VVC remains a challenge. New non-azole antifungals are now in late-stage development for acute and recurrent VVC, and their availability may provide more options for treatment. W
Barbara Dehn is with the El Camino Women’s Medical Group in Mountain View, California. Susan Kellogg-Spadt is the Director of Female Sexual Medicine at the Center for Pelvic Medicine in Rosemont, Pennsylvania. Kahlil Demonbreun is Women’s Health Medical Director at the Columbia Veteran Affairs Health Care System and a women’s health nurse practitioner in Orangeburg, South Carolina. Support for the development of this manuscript was received from Scynexis, Inc., in Jersey City, New Jersey. Barbara Dehn reports that she is a consultant and Kahlil Demonbreun reports that he serves on an NP advisory panel for Scynexis. Susan Kellogg-Spadt reports no actual or potential conflicts of interest in relation to the contents of this article.
Acknowledgment
The authors would like to acknowledge the editorial assistance of Richard Perry, PharmD, in the preparation of this manuscript, which was supported by Scynexis, Inc.
References
Amabebe E, Dilly Anumba DOC. The vaginal microenvironment: the physiologic role of Lactobacilli. Front Med. 2018;5:181.
Yano J, Sobel JD, Nyirjesy P, et al. Current patient perspectives of vulvovaginal candidiasis: incidence, symptoms, management and post-treatment outcomes. BMC Womens Health. 2019;19(1):48.
Goncalves B, Ferreira C, Alves CT, et al. Vulvovaginal candidiasis: epidemiology, microbiology and risk factors. Crit Rev Microbiol. 2016;42(6):905-927.
Goje OJ. Obstetrics and gynecology. Vulvovaginal yeast infections. Cancer Therapy Advisor. June 16, 2020. https://www.cancertherapyadvisor.com/home/decision-support-in-medicine/obstetrics-and-gynecology/vulvovaginal-yeast-infections/.
Rosati D, Bruno M, Jaeger M, et al. Recurrent vulvovaginal candidiasis: an immunological perspective. Microorganisms. 2020;8(2):144.
Denning DW, Kneale M, Sobel JD, Rautemaa-Richardson R. Global burden of recurrent vulvovaginal candidiasis: a systematic review. Lancet Infect Dis. 2018;18(11):e339-e347.
Centers for Disease Control and Infection. 2015 sexually transmitted diseases treatment guidelines: vulvovaginal candidiasis. https://www.cdc.gov/std/tg2015/candidiasis.htm.
Committee on Practice Bulletins—Gynecology. Vaginitis in nonpregnant patients. Practice bulletin no. 215. Obstet Gynecol. 2020;135(1):e1-e17.
Yano J, Peters BM, Noverr MC, Fidel PL Jr. Novel mechanism behind the immunopathogenesis of vulvovaginal candidiasis: “neutrophil anergy.” Infect Immun. 2018;86(3):e00684-17.
Aballea S, Guelfucci F, Wagner J, et al. Subjective health status and health-related quality of life among women with recurrent vulvovaginal candidiasis (RVVC) in Europe and the USA. Health Qual Life Outcomes. 2013;11:169.
Fukazawa EI, Witkin SS, Robial R, et al. Influence of recurrent vulvovaginal candidiasis on quality of life issues. Arch Gynecol Obstet. 2019;300(3):647-650.
Centers for Disease Control and Prevention. Fungal diseases: vaginal candidiasis. November 10, 2020. https://www.cdc.gov/fungal/diseases/candidiasis/genital/index.html.
Benedict K, Jackson BR, Chiller T, Beer KD. Estimation of direct healthcare costs of fungal diseases in the United States. Clin Infect Dis. 2019;68(11):1791-1797.
Meng L, Zhao H, Zhao S, et al. Inhibition of yeast-to-hypha transition and virulence of Candida albicans by 2-alkylaminoquinoline derivatives. Antimicrob Agents Chemother. 2019;63(4):e01891-18.
Brown H, Drexler M. Improving the diagnosis of vulvovaginitis: perspectives to align practice, guidelines, and awareness. Popul Health Manag. 2020;23(S1):S3-S12.
Nyirjesy P, Banker WM, Bonus TM. Physician awareness and adherence to clinical practice guidelines in the diagnosis of vaginitis patients: a retrospective chart review. Popul Health Manag. 2020;23(S1):S13-S21.
Pappas PG, Kauffman CA, Andes DR, et al. Clinical practice guideline for the management of candidiasis: 2016 update by the Infectious Diseases Society of America. Clin Infect Dis. 2016;62(4):e1-50.
Schwebke JR, Gaydos CA, Nyirjesy P, et al. Diagnostic performance of a molecular test versus clinician assessment of vaginitis. J Clin Microbiol. 2018;56(6):e00252-18.
Schwebke JR, Taylor SN, Ackerman R, et al. Clinical validation of the Aptima bacterial vaginosis and Aptima Candida/Trichomonas vaginitis assays: results from a prospective multicenter clinical study. J Clin Microbiol. 2020;58(2):e01643–19.
Kawa D, Paradis S, Yu J, LeJeune M. Elevating the standard of care for women’s health: the BD Max Vaginal Panel and management of vaginal infections. 2017. Moleculardiagnostics.bd.com/wp-content/uploads/2017/08/MAX-Vaginal-Panel-Whitepaper.pdf.
Bhattacharya S, Sae-Tia S, Fries BC. Candidiasis and mechanisms of antifungal resistance. Antibiotics (Basel). 2020;9(6):312.
Diflucan [fluconazole] package insert. New York, NY: Pfizer; 2020.
Sobel JD, Sobel R. Current treatment options for vulvovaginal candidiasis caused by azole-resistant Candida species. Expert Opin Pharmacother. 2018;19(9):971-977.
Rosa MI, Silva BR, Pires PS, et al. Weekly fluconazole therapy for recurrent vulvovaginal candidiasis: a systematic review and meta-analysis. Eur J Obstet Gynecol Reprod Biol. 2013;167(2):132-136.
Marchaim D, Lemanek L, Bheemreddy S, et al. Fluconazole-resistant Candida albicans vulvovaginitis. Obstet Gynecol. 2012;120(6):1407-1414
Collins LM, Moore R, Sobel JD. Prognosis and long-term outcome of women with idiopathic recurrent vulvovaginal candidiasis caused by Candida albicans.
J Low Genit Tract Dis. 2020; 24(1):48-52.
Felton T, Troke PF, Hope WW. Tissue penetration of antifungal agents. Clin Microbiol Rev. 2014;27(1):68-88
Rodríguez-Cerdeira C, Martínez-Herrera E, Carnero-Gregorio M, et al. Pathogenesis and clinical relevance of Candida biofilms in vulvovaginal candidiasis. Front Microbiol. 2020;11:544480.
Zhou X, Li T, Fan S, et al. The efficacy and safety of clotrimazole vaginal tablet vs. oral fluconazole in treating severe vulvovaginal candidiasis. Mycoses. 2016;59(7):419-428
Mintz JD, Martens MG. Prevalence of non-albicans Candida infections in women with recurrent vulvovaginal symptomatology. Adv Infect Dis. 2013;3(4):238-242
Makanjuola O, Bongomin F, Fayemiwo SA. An update on the roles of non-albicans Candida species in vulvovaginitis. J Fungi (Basel). 2018;4(4):121.
Sobel JD, Kapernick PS, Zervos M, et al. Treatment of complicated Candida vaginitis: comparison of single and sequential doses of fluconazole. Am J Obstet Gynecol. 2001;185(2):363-369.
Ambizas EM, Maniara B. Nonprescription management of vulvovaginal candidiasis. US Pharm. 2015;40(9):13-19.
Nystatin vaginal inserts package insert. Nystatin Vaginal Insert – FDA prescribing information, side effects and uses (drugs.com).
Aguin TJ, Sobel JD. Vulvovaginal candidiasis in pregnancy. Curr Infect Dis Rep. 2015;17(6):462.

