Endometriosis is a gynecologic condition in which endometrial cells normally lining the uterus grow outside the uterine cavity, predominately attaching to fallopian tubes, ovaries, bowel, and bladder.1 The prevalence of endometriosis is reported to be as high as 10% in women of reproductive age and is known for its distinguishing symptoms of debilitating pelvic pain, dysmenorrhea, dyspareunia, and menstrual gastrointestinal distress.1 Endometriosis is most common among women age 30 to 45 years and typically resolves at the onset of menopause.2 Gold standard diagnosis of endometriosis is by visual inspection with laparoscopy and histologic verification.3
Unfortunately, despite its common occurrence, there is a disappointing amount of public and provider awareness, leading to an average of 10 years before diagnosis is made.4 Contributing factors delaying diagnosis are multisymptom presentation, inability to be visualized on ultrasound, insufficient use of screening guidelines, and the existing stigma of expected pain during menses.5 Extended diagnostic timeframes impede treatment causing significant impact on quality of life, fertility preservation, and employment status.5,6 Endometriosis also increases risk for other comorbidities including autoimmune disorders, cardiovascular conditions, and atopic diseases.2
The National Institute for Health and Care Excellence (NICE) released guidelines to improve endometriosis diagnosis and management in September 2017.3 Although this is a step in the right direction, the guidelines are underutilized and a specific self-reported screening questionnaire has not been developed.2 Historically, self-reported screening questionnaires have proved beneficial in expediting the diagnosis of other diseases such as binge eating disorders, psoriasis, and fibromyalgia. Further, patient-based screening tools create an opportunity for patients to discreetly report symptoms and allow for healthcare providers to discuss openly during an office visit.4
Purpose
The purpose of the DNP quality improvement (QI) project was to create and execute an evidence-based screening protocol in a gynecologic setting for endometriosis modeled after NICE guidelines with the development of a self-reported screening questionnaire. The project goals were to achieve a 20% increase in diagnosis of endometriosis by laparoscopy and to identify symptoms prior to 10 years of onset. A standardized screening protocol for endometriosis was not utilized at the chosen gynecology practice prior to the QI project.
Description of setting and population
The QI project took place at three offices within the same gynecology practice located throughout Central Ohio and led by a physician and two nurse practitioners. The site provided complete gynecologic services to a predominantly White population with approximately two-thirds of patients with private insurance and the remaining one-third having government-issued insurance. Women age 18 to 49 years were targeted for the QI project. During implementation, typical patient volume ranged from approximately 15 to 25 daily.
Description of the quality improvement project
The QI project was approved by the Institutional Review Board at Missouri State University. A self-reported screening questionnaire was developed after selecting five questions derived from content found in the algorithm provided by NICE guidelines for endometriosis screening (Figure 1).3 An educational workshop was held with the intent of increasing staff knowledge about endometriosis and the need for the QI project. Participants completed pre and post educational surveys. Identity was protected by blind numerical assignment to each pair of surveys. Over the course of a 22-week period, the questionnaires containing only patient identification number were administered. Specifically targeted were gynecologic agreeable patients being seen for annual examinations or acute visits with complaints of dysmenorrhea, dyspareunia, and abdominal and/or pelvic pain after reading a disclosure statement detailing the intent for use of information. Questionnaires were then reviewed by the provider and treatment recommendations discussed with patients at visits.
Methods to evaluate outcomes
A paired t-test was used to compare pre- and post-test survey scores. Completed questionnaires were categorized as negative, suspicious, or positive for endometriosis with use of a designated screening scale constructed from NICE screening guidelines. With the exception of the negative forms, an Excel spreadsheet was used to log the following information: patient identification number; date of visit; new or established; age; years with symptoms; and recommended treatment plan. Patients whose treatment plan indicated the need for surgical laparoscopy and excision procedures were classified separately and outcome data recorded. Data were analyzed using a paired t-test comparing laparoscopies performed from June 14, 2020, to January 31, 2021, and during project implementation from June 14, 2021, to January 31, 2022. Data collection ceased November 14, 2022, to allow for typical surgical wait times of 8 to 10 weeks. The number of years with symptoms was calculated using descriptive statistics and reported in the form of a percentage.
Outcomes
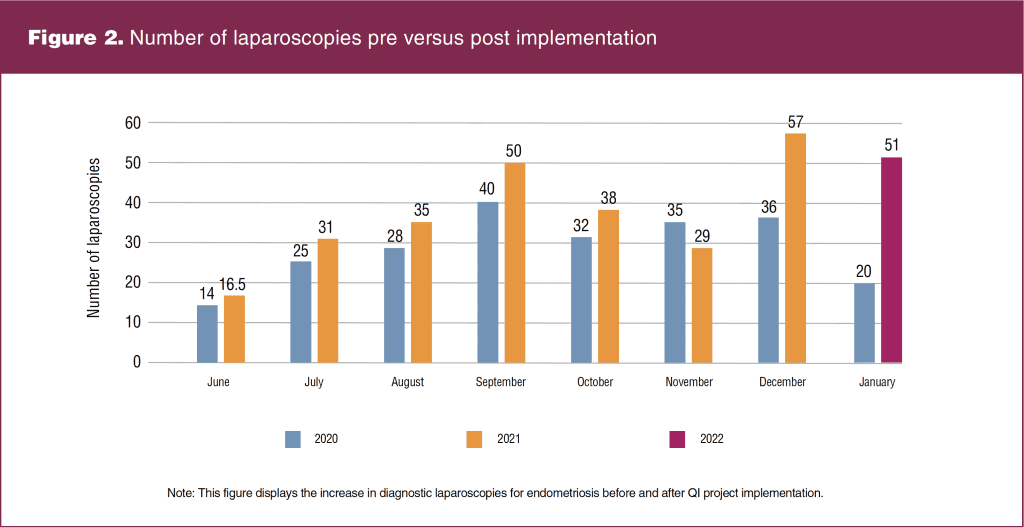
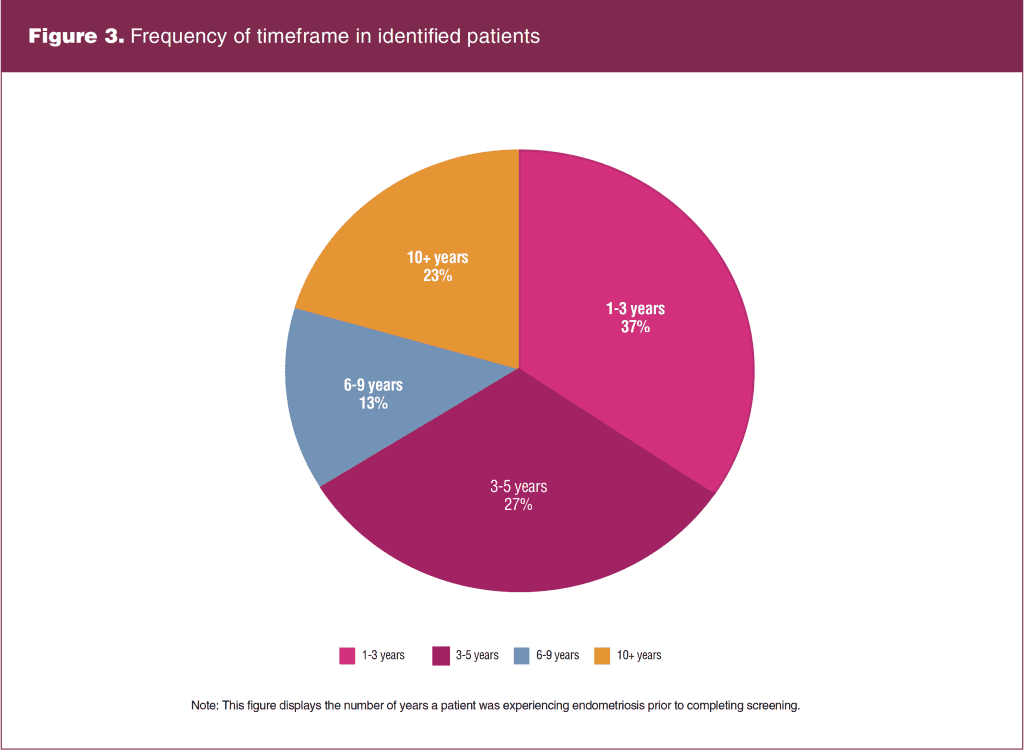
Reliability analysis subscales were deemed reliable and consistent for all 11 completed surveys. Paired t-tests revealed for categories knowledge (t = -5.05, P = .001, d = -1.46); screening (t = -2.72, P = .020, d = 0.78), and barriers (t = -2.81, P = .017, d = -0.81) were statistically significant indicating an increase in clerical and medical staff awareness. Of the 911 completed screenings, 141 patients were identified as suspicious for endometriosis. Recommended treatment varied based on severity and longevity of symptoms and previous failed treatments, with 49 patients advised to trial hormonal suppression of menses, five choosing to try for pregnancy, and 87 to undergo surgical laparoscopy. The total number of patients who had laparoscopies was 65; 22 failed to undergo laparoscopy secondary to infection with Covid-19, transfer of care, or noncompliance of ordered testing. A paired t-test comparing the number of laparoscopies performed pre and post screening had a statistically significant increase (t = -2.39, P = .042, d = -0.85). As seen in Figure 2, there was an overall 25% increase, exceeding the project goal of 20%. As pictured in Figure 3, 76.6% of the 141 patients were identified prior to an average diagnostic timeframe of 10 years. Also important to note, more than half of those who had laparoscopies were discovered to be in the early stages of endometriosis, with 27 patients in stage 1, 24 in stage 2, 10 in stage 3, and four in stage 4. Unexcepted outcomes with use of screening included modifications in treatment plans for 26 patients with existing endometriosis.
Discussion and implication for practice
There was a steady increase in diagnostic laparoscopy and laser removal of endometriosis, with the exception of the month of November. Needed surgical days were unavailable for patient volume and therefore overflow can be seen in December and January figures.
Facilitators included healthcare providers, office management staff, and other personnel at the project site. It is poignant to note additional assistance was received from the following staff at Missouri State University: project chair; director of DNP program, and RSTAT department. Implementation of the QI project did not cause financial strain or major disruption in workflow. The cooperation and willingness of stakeholders to facilitate the intended project plan allotted for successful completion and achievement of project outcomes.
Barriers for the QI project were inability to effectively observe implementation simultaneously in all office locations. Only 70% of qualifying patients appearing on provider schedules received the questionnaire, lowering the expected sample size. No shows, patient refusal to participate, misplaced screening forms, or failure of front office staff to administer forms were likely causative factors. Lack of provider consistency in addressing the screening form at visits also lowered the expected number of identified patients.
Lessons learned include the need to edit the screening questionnaire based on patient feedback. Frequent confusion occurred with use of the term “pelvic pain,” with “cramps” being more widely understood and assessment of gastrointestinal symptoms. Project sustainability is likely due to its simplistic nature and ease of replication. Key findings are of the 141 patients identified as suspicious of endometriosis; 64 were established, highlighting former unrecognition of symptoms.
Conclusion
The screening protocol was successful in increasing timely identification of endometriosis. Although this is reassuring, results are limited due to a short implementation period and use trialed in only one gynecology practice. Active screening for endometriosis allowed the opportunity for patient verbalization of previous dismissal of symptoms, fueling the need for adoption of a standardized screening protocol in gynecology settings. Accelerating the diagnosis of endometriosis can allow for improved medical management.
Acknowledgment: The authors extend sincere thanks to Melissa Penkalski, DNP, APRN, CPNP-PC, AE-C; Brandie McGuire, WHNP-BC; Cindy Schaffer, Practice Manager; and Hannah Johnson, RSTAT, for QI project support.
Sarah M. King practices obstetrics and gynecology at Progressive Women’s Healthcare in Dayton, Ohio. Kathryn A. Adams is Clinical Associate Professor for the School of Nursing at Missouri State University in Springfield, Missouri. Caroline E. Peterson practices obstetrics and gynecology at Progressive Women’s Healthcare in Dayton, Ohio. The authors have no actual or potential conflicts of interest in relation to the contents of this article.
Womens Healthcare. 2023;11(1):41-44. doi: 10.51256/WHC022341
References
1 Farshi N, Hasanpour S, Mirghafourvand M, Esmaeilpour K. Effect of self-care counselling on depression and anxiety in women with endometriosis: a randomized controlled trial. BMC Psychiatry. 2020;20(1):391.
2 Vannuccini S, Lazzeri L, Orlandini C, et al. Mental health, pain symptoms and systemic comorbidities in a woman with endometriosis: a cross-sectional study. J Psychosom Obstet Gynaecol. 2018;39(4):315-320.
3 National Institute for Health and Care Excellence. Endometriosis: diagnosis and management. National Guideline Alliance. NICE Guideline NG73. September 6, 2017. https://www.nice.org.uk/guidance/ng73.
4 Surrey E, Carter CM, Soliman AM, et al. Patient-completed or symptom-based screening tools for endometriosis: a scoping review. Arch Gynecol Obstet. 2017;296(2):153-165.
5 Surrey E, Soliman AM, Trenz H, et al. Impact of endometriosis diagnostic delays on healthcare resource utilization and costs. Adv Ther. 2020;37(3):1087-1099.
6 van der Zanden M, Teunissen DAM, van der Woord IW, et al. Barriers and facilitators to the timely diagnosis of endometriosis in primary care in the Netherlands. Fam Pract. 2020;37(1):131-136.