Approximately 40% of all pregnancies in the United States are unplanned, and about 50% of all unintended pregnancies occur in women who are not using contraception. In this article, the authors discuss state-based initiatives to increase access and decrease barriers to care by promoting pharmacy-provided hormonal contraceptives. The authors describe their experience in participating on a legislative commission to craft legislation to expand access.
Even though unintended pregnancy rates have fallen to a 30-year low,1 40% of all pregnancies have remained unplanned and 36% have followed an interpregnancy interval of less than 18 months.2 As with most social issues, the risk for unintended pregnancy does not affect all populations equally; distinct disparities exist along racial and economic lines. Women of color as well as those of other minority groups, women of lower education and income levels, and younger women, when compared with the rest of the female adult population, are disproportionately affected by unintended pregnancy.1,3
About 50% of unintended pregnancies occur in women who are not using contraception.4 Lack of access to healthcare, lack of information/knowledge about contraception, and cultural norms surrounding contraceptive use have been cited by women as reasons why they do not use a birth-control method.4 Helping women and their partners choose contraceptive methods that are most appropriate for them, as well as ensuring that they receive education and assistance regarding how to use these methods effectively and consistently, can help decrease the still-too-high rate of unintended pregnancy in this country.3
Overestimating hormonal contraceptive risks
Misconceptions regarding the safety of hormonal contraceptives (HCs) may result in lack of use, inconsistent use, or early discontinuation, any of which can lead to an unintended pregnancy. The risks associated with use of HCs, especially when prescribed appropriately, are lower than those associated with pregnancy itself, particularly unintended pregnancy.5 Although use of combined HCs (CHCs)—that is, contraceptives containing estrogen and a progestogen—can increase certain health risks, the overall risk to women without pre-existing risk factors or conditions is extremely low. Increased risk is more typically seen in women with co-morbid conditions such as obesity, hypertension, migraines, or clotting disorders. Studies have shown that women are able to self-screen reliably for these conditions using a safety checklist.6
Mørch et al.7 assessed for any links between HC use and the risk of invasive breast cancer in a nationwide prospective cohort study involving all women in Denmark aged 15-49 years who had not had cancer or venous thromboembolism (VTE) and who had not received treatment for infertility. Over a mean follow-up of 11 years, current and recent HC users, compared with females who had never used HCs, had a relative risk of 1.20 of developing breast cancer. This finding translated into approximately one extra case of breast cancer per 7,690 HC users, including women who used hormonal intrauterine devices.7
A systematic review showed that users of CHCs, compared with women who did not take hormones, had a relative risk of 1.22-1.93 for VTE, depending on the type of progestogen used in their CHC formulation.8 A Cochrane Review meta-analysis showed that, compared with nonusers of hormones, users of combined oral contraceptives (COCs), some of which contained estrogen 50 mcg, had a relative risk of 1.6 for myocardial infarction (MI) or ischemic stroke.9 The absolute risk of these events remained low in all three studies. In another study of a cohort of Danish women without prior cardiovascular disease who were followed for 15 years, the risks for both thrombotic stroke and MI were increased by a factor of 0.9-1.7 with use of low-dose COCs (i.e., those containing ethinyl estradiol [EE] 20 mcg) and by a factor of 1.3-2.3 with use of COCs containing EE 30-40 mcg.10 Progestogen type did not appear to have a significant impact on these risks. These findings also supported the use of estrogen-free HCs such as progestin-only pills (POPs) as safe alternatives for women in whom estrogen is contraindicated.5
Underestimating the risks of unintended pregnancy
Unintended pregnancy, as compared with pregnancy that is planned, is associated with an increased risk for maternal and infant health problems.3 Lack of prenatal care plays a large role in the increased rates of maternal and neonatal morbidity/ mortality associated with unintended pregnancy.11 Lack of a basic nutritional consultation and prenatal vitamin supplementation, awareness to avoid alcohol and other teratogenic substances during the first trimester, and the inability to proactively obtain appropriate care for the mother’s own health conditions, if necessary, may have a significantly adverse impact on the health of both mother and child.6 These increased risks have an economic cost as well: In 2010 alone, unintended pregnancies cost the U.S. healthcare system an estimated $21 billion.12
Addressing inequities in access to contraceptives: Pharmacy provision of HCs
Even if women understand the low risk of HC use and the relatively higher risks associated with unintended pregnancy, they need to have physical and financial access to contraceptives—hormonal and otherwise—in order to use them. Therefore, contraception is not only a medical issue but also a public health issue and an economic issue.
Action at federal and state levels
Both state-based and national initiatives are evolving to address inequities in access to contraceptives. At the federal level, a French drug company, HRA Pharma, has initiated an actual-use study to provide important data to the FDA for over-the-counter (OTC) status of POPs. Ibis Reproductive Health, a research and advocacy group on women’s health priorities, has partnered with HRA Pharma for support through the FDA application process. The application process may be streamlined for POPs because the FDA has already approved OTC status for emergency-contraceptive POPs and because POPs have fewer contraindications than do CHCs.
Although only 4% of women use POPs,13 results of a recent study suggested that about one-third of teenagers and women would be interested in using POPs if they were available OTC.13,14 The FDA-approved product information sheet lists the first-year failure rate for POPs as 0.5%, with a typical failure rate closer to 5% because of late or omitted pills.15 Action at the FDA level to approve OTC POPs could greatly expand contraceptive access to teenagers and women, although passing legislation at the state level might be accomplished in a timelier, less cumbersome manner than the lengthy and complex FDA process. In fact, 12 states have already passed or proposed legislation to expand access to HCs in pharmacies—without needing to have a prescription specifically written by a healthcare provider (HCP) for a given patient (Box 1).16-28 Pharmacies have the advantage of convenient locations, extended hours of operation, and availability of walk-in access without appointments.
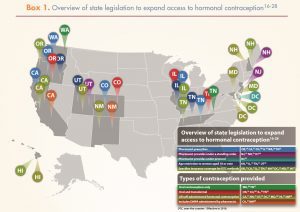
In order to achieve this end—that is, provision of HCs by pharmacists— states can take one of three general routes: standing order, collaborative practice agreement (CPA), or pharmacist prescribed. A standing order is one written by a prescriber (e.g., physician, advanced practice registered nurse, physician assistant) that gives a non-prescribing clinician such as a pharmacist the authority to dispense the medication as long as predetermined conditions have been met. This route requires detailed legislation to address all legal issues, a prescriber willing to write the standing order, and annual updates for the order.29 CPAs are contracts between a pharmacist and other members of a healthcare team, including a prescribing provider, that outline and allow for certain expanded services that the pharmacist can provide within that practice under specific conditions.30-32 This type of arrangement is seen frequently in hospital settings wherein a pharmacist takes the lead in managing complex medication regimens such as chemotherapy, chronic pain treatment, or therapy for other pharmacologically complex diseases.30 In the case of pharmacist-prescribed medication, prescriptive authority comes from the state and does not depend on the medical license of a separate HCP. As with standing orders and CPAs, the pharmacist must undergo additional training and the patient must meet certain criteria in order to obtain contraceptives.
In all three situations, patients must complete a self-screening risk assessment tool, check their blood pressure in the pharmacy, and submit the completed form to the pharmacist. The pharmacist reviews the health history and, if no contraindications are present, provides the HC. If a contraindication is identified, the pharmacist refers the woman to an HCP. Numerous studies have shown that patient self-screening is accurate and safe.33-35
Types of pharmacist-provided contraceptives, use of standing orders, protocols, pharmacist-prescribing roles, age restrictions, and insurance coverages vary from state to state (Box 1).16-28 In addition, data from states that have expanded pharmacy access indicate that barriers may remain despite legislation. Barriers include the time required for pharmacist training, already demanding pharmacist workloads, and, most notably, a lack of insurance reimbursement for pharmacists’ time spent in evaluation. Legislation in six states has addressed the latter-most barrier by expanding pharmacists’ scope of practice to enable direct billing for time spent providing the service.
Action by the OCs OTC Working Group
An exhaustive search identified multiple physician groups that have issued policy statements on expanding access to contraceptives. Although no nursing organizations have issued statements of their own at this time, many of them have signed a Statement of Purpose published by the OCs OTC Working Group to expand access to OCs through OTC status (Box 2). This working group is a coalition of reproductive health, rights, and justice organizations; nonprofit research and advocacy groups; university-based researchers; and prominent clinicians who are committed to providing easier access to safe, effective, acceptable, and affordable contraceptives to all reproductive-age women.

Signing this Statement of Purpose is consistent with nursing organizations’ strong history of advocating for healthcare reform to improve access to high-quality care for all populations. Nursing’s history of advocacy has included support for the Affordable Care Act (ACA), which was intended to provide parity in health services and to improve access for women by requiring insurance companies to cover a standard package of essential health services. One essential benefit addressed coverage of 18 FDA-approved contraceptive methods, along with education and counseling; any and all appointments needed to initiate or discontinue a method; and follow-up visits to manage any concern related to the method. Provisions were added so that insurers cannot levy out-of-pocket charges such as deductibles or copays for any of these services.
Persistent barriers
The ACA addressed financial barriers to acquiring contraceptives by mandating insurance coverage. However, other obstacles persist, including a need to travel long distances for prescribers’ services, some prescribers’ lack of evening and/or weekend hours, an inability to leave work to seek care without penalty, or a need to schedule an appointment in advance. In 2015, federal legislation, the Allowing Greater Access to Safe and Effective Contraception Act, was proposed to encourage the FDA to approve OTC status for OCs. However, no provisions in the bill ensured teenagers’ access to OCs or ensured that insurance companies would cover the cost of the pills.36 This legislation failed; many policy advocates saw it as an attempt by insurers to avoid providing coverage for contraceptives. State legislatures’ provision for pharmacist-prescribed contraceptives would bridge this gap, increasing access to HCs, including OCs, while continuing to ensure insurance coverage for consumers.
Status of pharmacy provision of HCs in New Hampshire: A case study
In New Hampshire (NH), the NH Nurses Association (NHNA) and the NH Nurse Practitioner Association (NHNPA) share the services of a lobbyist who is tasked with monitoring all health-related bills. If commissions are developed, the lobbyist ensures that nursing is represented with membership on them. A bipartisan bill to expand pharmacy provision of HCs was introduced in 2016 and referred to a commission for further study. Three nursing organizations—the NHNA, the NHNPA, and the NH Board of Nursing (NHBN)—put out a call for volunteers to represent nursing on the commission. Members of the commission included representatives from the NH Pharmacists Association, the NH Board of Pharmacy, the NHBN, the NHNA, the NHNPA, the NH Medical Society, and the NH Board of Medicine, as well as health policy experts.
At the initial meeting, all members voiced commitment to the bill’s ultimate goal: to expand contraceptive access. The commission met 6 times and modified the original language of the bill so that it would now ensure teenagers’ access to contraceptives and guarantee that insurers would reimburse for contraceptives provided by a pharmacist. The commission compromised on two notable areas: (1) allowing pharmacists to prescribe under their own licenses because there were no precedents for this practice in the state, and (2) removal of depot medroxyprogesterone acetate from the list of contraceptives because not all members of the commission were comfortable with its inclusion. The final decision of the commission was unanimous—a compelling statement to legislators about the healthcare community’s support for the bill. Legislators needed substantial education about the goals of the bill. Ultimately, it passed with only minor changes.
Participating in the creation of new healthcare legislation can be a rewarding professional experience. In addition, serving on a collaborative healthcare commission with members from all sectors of the healthcare community and working toward one goal can be profoundly inspiring. Participation provides an opportunity to deepen understanding of healthcare policy, politics, and the law in general and to act as agents for change within the community. As the process in NH may diff er from the pathway elsewhere, readers are encouraged to reach out to their own state nursing organizations to find out more about opportunities to participate within their local governments.
Recommendations for action
Various public policy approaches are needed to address the high rate of unintended pregnancy in the U.S. An ideal strategy is to make a progestogen-only contraceptive available OTC. However, this approach requires a drug company to submit an application to the FDA, a process that can take at least 4 years. An alternate strategy is to expand pharmacy access through state legislation. Either approach needs full support from professional healthcare societies. The annual Gallup poll consistently ranks nursing as the most honest and ethical profession.37 As such, the public trusts nurses as leaders in healthcare. Nursing organizations are urged to develop policy statements regarding expanded contraception access. To this end, the National Association of Nurse Practitioners in Women’s Health (NPWH) has written a position statement on expanding access to contraceptives via pharmacies at the state level and via granting OTC status to OCs at the federal level. The position statement appears on pages 16-19 in this issue of the journal. Nursing colleagues need to seek opportunities to get involved in their local government process to help reach the goal of increasing access to contraceptives.
Joyce D. Cappiello is Assistant Professor of Nursing at the University of New Hampshire in Durham and a nurse practitioner at the Joan G. Lovering Health Center in Greenland, New Hampshire. Lindsay M. Schommer is Instructor in Neurology at Dartmouth Medical School in Hanover and a nurse practitioner at Dartmouth Epilepsy Center DHMC in Lebanon, both in New Hampshire. The authors state that they do not have a fi nancial interest in or other relationship with any commercial product named in this article.
References
1. Finer L, Zolna M. Declines in unintended pregnancy in the United States, 2008-2011. New Engl J Med. 2016;374(9):843-852.
2. Ahrens K, Thoma M, Copen C, et al. Unintended pregnancy and interpregnancy interval by maternal age, National Survey of Family Growth. Contraception. 2018;98(1):52-55.
3. CDC. Unintended Pregnancy Prevention. Updated January 22, 2015. cdc.gov/reproductivehealth/unintendedpregnancy/index.htm
4. Mosher WJ, Jones J, Abma J. Nonuse of contraception among women at risk of unintended pregnancy in the United States. Contraception. 2015;92(2):170-176.
5. Shulman L. The state of hormonal contraception today: benefits and risks of hormonal contraceptives: combined estrogen and progestin contraceptives. Am J Obstet Gynecol. 2011;205(4):S9-S13.
6. Barot S. Moving oral contraceptives to over-the-counter status: policy versus politics. Guttmacher Pol Rev. 2015;18(4):85-91.
7. Mørch LS, Skovlund CW, Hannaford PC, et al. Contemporary hormonal contraception and the risk of breast cancer. N Engl J Med. 2017;377(23):2228-2239.
8. Martínez F, Ramírez I, Pérez-Campos E, et al. Venous and pulmonary thromboembolism and combined hormonal contraceptives. Systematic review and meta-analysis. Eur J Contracept Reprod Health Care. 2012;17(1):7-29.
9. Roach RE, Helmerhorst FM, Lijfering WM, et al. Combined oral contraceptives: the risk of myocardial infarction and ischemic stroke. Cochrane Database Syst Rev. 2015;(8):CD011054.
10. Lidegaard Ø, Løkkegaard E, Jensen J, et al. Thrombotic stroke and myocardial infarction with hormonal contraception. N Engl J Med. 2012;366(24):2257-2266.
11. Curtis KM, Jatlaoui TC, Tepper NK, et al. U.S. Selected Practice Recommendations for Contraceptive Use, 2016. MMWR Recomm Rep. 2016;65(4):1-66.
12. Sonfield A, Kost K. Public Costs from Unintended Pregnancies and the Role of public Insurance Programs in Paying for Pregnancy-Related Care: National and State Estimates for 2010. February 2015. guttmacher.org/report/public-costs-unintended- pregnancies-and-role-public-insurance- programs-paying-pregnancy
13. Liang SY, Grossman D, Phillips KA. User characteristics and out-of-pocket expenditures for progestin-only versus combined oral contraceptives. Contraception. 2012;86(6):666-672.
14. Grindlay K, Grossman D. Interest in over-the-counter access to a progestin-only pill among women in the United States. Womens Health Issues. 2018;28(2):144-151.
15. Ortho Micronor Prescribing Information. Reviewed on March 1, 2018. drugs.com/pro/ortho-micronor.html
16. Oregon House Bill 2527. June 15, 2017. olis.leg.state.or.us/liz/2017R1/Downloads/MeasureDocument/HB2527/Enrolled
17. Feder Ostrov B. California, Oregon to allow hormonal contraceptives without a doctor’s prescription. Kaiser Health News. July 15, 2015. khn.org/news/california-oregon- to-allow-hormonal-contraceptives-without-a-doctors-prescription
18. Colorado Senate Bill 16-135. An Act. June 6, 2016. leg.colorado.gov/sites/default/files/2016A_135_signed.pdf
19. Illinois General Assembly. Bill Status of HB 5809. January 10, 2017. ilga.gov/legislation/BillStatus.asp?DocTypeID=HB&DocNum=5809&GAID=13&SessionID=88&LegID=95273
20. Washington State House Bill 2879. June 2, 2015. gov.oregonlive.com/bill/2015/HB2879/
21. Justia US Law. TN Code § 63-10-219. 2016. law.justia.com/codes/tennessee/ 2016/title-63/chapter-10/part-2/ section-63-10-219/
22. New Jersey Senate Senate No. 1073. 2016. njleg.state.nj.us/2016/Bills/S1500/1073_I1.PDF
23. New Hampshire HB House Bill 1822-FN. Making hormonal contraceptives available directly from pharmacists by means of a standing order. 2018. legiscan.com/NH/text/HB1822/2018
24. Utah Senate Bill 184. Pharmacist Dispensing Authority Amendments. 2018. le.utah.gov/~2018/bills/static/SB0184.html
25. Council of the District of Columbia. Access to Contraception Amendment Act 2016. lims. dccouncil.us/Download/35647/B21-0707-Introduction.pdf
26. General Assembly of Maryland SB 0363. Pharmacists-Contraceptives-Prescribing and Dispensing. July 1, 2017. mgaleg.maryland.gov/ webmga/frmmain.aspx?pid=billpage &stab=01&id=sb0363&tab=subject 3&ys=2017rs
27. Hawaii, ACT 067, SB513 SD1 HD2 CD1. July 3, 2017. governor.hawaii. gov/2017-acts/act-067-sb513- sd1-hd2-cd1-7032017/
28. Protocol for Pharmacist Prescription of Hormonal Contraception in New Mexico. June 2016. rld.state.nm.us/uploads/files/OCConfirmedFinalJune2016.pdf
29. Litz R. How are pharmacists providing naloxone to patients? Pharmacy Times. Dec 2015. pharmacytimes.com/news/ how-are-pharmacists-providing- naloxone-to-patients
30. CDC. Advancing Team-Based Care Through Collaborative Practice Agreements: A Resource and Implementation Guide for Adding Pharmacists to the Care Team. 2017. cdc.gov/dhdsp/pubs/docs/ CPA-Team-Based-Care.pdf
31. Collaborative Practice Agreements (CPA) and Pharmacists’ Patient Care Services. 2013. aphafoundation.org/collaborative-practice-agreements
32. Adams AJ, Weaver KK. The continuum of pharmacist prescriptive authority. Ann Pharmacother. 2016;50(9):778-784.
33. Grossman D, Fernandez L, Hopkins K, et al. Accuracy of self-screening for contraindications to combined oral contraceptive use. Obstet Gynecol. 2008;112(3):572-578.
34. Shotorbani S, Miller L, Blough DK, et al. Agreement between women’s and providers’ assessment of hormonal contraceptive risk factors. Contraception. 2006;73(5):501-506.
35. White K, Potter J, Hopkins K, et al. Contraindications to progestin-only oral contraceptive pills among reproductive-aged women. Contraception. 2012;86(3):199-203.
36. Congress.gov. 115th Congress (2017-2018). S.93 – Allowing Greater Access to Safe and Effective Contraception Act. 2018. congress.gov/bill/115th-congress/ senate-bill/93
37. Brennan M. Nurses Keep Healthy Lead as Most Honest, Ethical Profession. December 26, 2017. news. gallup.com/poll/224639/nurses- keep-healthy-lead-honest-ethical- profession.asp

