Postpartum depression is one of the most common disorders of the peripartum period and has significant adverse effects on the mother and infant. Prominent organizations recommend screening for depression during pregnancy and postpartum. Several validated screening tools are available. Treatment includes both nonpharmacologic and pharmacologic interventions. In this article, the author provides an overview of postpartum depression and describes current screening and treatment recommendations.
Background
Postpartum depression, or major depression that occurs within the first year after childbirth, affects approximately 10% to 20% of women who have had children.1 The actual prevalence is likely higher with underreporting due to lack of access to postpartum care for some women combined with a lack of consistent screening by healthcare providers.1 The incidence of postpartum depression increases in women with more than one child, women of lower socioeconomic status, single marital status, a history of depression, low level of religiosity, and young maternal age.2 Patients who have a preterm delivery, a traumatic birth experience, breastfeeding problems, history of drug abuse, or have been a victim of physical abuse are also at an increased risk for postpartum depression.3,4
Postpartum depression has a spectrum of adverse effects on both mother and infant. Mothers who experience postpartum depression may have poor adherence to healthcare recommendations, are less likely to breastfeed, and can have difficulties with maternal-infant
attachment.5,6 They may experience financial and interpersonal losses, and may be at risk for substance use disorders.7 Children born to mothers with postpartum depression are at high risk for delayed development of speech and language and impaired social development.5,8 Depressive psychosis, an extreme form of postpartum depression, can lead to maternal suicide and/or infanticide. In fact, maternal suicide within a year of giving birth is emerging as a major cause of maternal mortality and is probably underreported.7
The diagnostic criteria for major depressive disorder are outlined in the Diagnostic and Statistical Manual of Mental Disorders, 5th edition.9 The patient must exhibit five or more of the following symptoms for at least 2 weeks for a diagnosis: depressed mood most of the time and on most days, anhedonia, significant change in weight, sleep disturbances, psychomotor fluctuations, change in energy levels, excessive guilt or feelings of worthlessness, difficulty concentrating, recurrent thoughts of death or suicidal thoughts.9 The same criteria are applicable to the postpartum patient, but because symptoms such as sleep disturbances and changes in energy level are common in the postpartum period, the possibility of depression may be overlooked. All postpartum patients thus should be screened for depression.10
Screening recommendations
The US Preventive Services Task Force (USPSTF) recommends screening for depression in the general adult population, including pregnant and postpartum women.11 The American College of Obstetricians and Gynecologists recommends that all pregnant mothers be screened for depression at least once during the perinatal period and again at the postpartum visit.5 Although the postpartum visit and screening for postpartum depression are covered by Medicaid and most private insurance, up to 40% of women still do not attend this visit.12
In an effort to reach more women, the USPSTF and the American Academy of Pediatrics (AAP) recommend screening the new mother at infant well visits.13 The AAP has expanded the screening recommendations to the 1-month, 2-month, 4-month, and 6-month well visits. Medicaid and most private insurances allow this screening to be claimed as a service for the child as part of the Early and Periodic Screening, Diagnostic, and Treatment benefit.13
Screening tools
Several validated depression screening tools are available for use with postpartum patients. One systematic review was conducted to determine the most widely used screening tools for postpartum depression and the most appropriate time to conduct the screening.14 The review found that the Edinburgh Postnatal Depression Scale (EPDS) is the most commonly used depression screening tool. The EPDS is a 10-question scale that is appropriate for patients with low health literacy and has been translated into over 50 languages. Items relevant to anxiety are included and are important as the mother may experience this mood disorder along with depression. The EPDS typically takes less than 5 minutes to complete.3 The questionnaire instructs the patient to answer based on their feelings for the last 7 days. A score of 13 or greater indicates postpartum depression, and higher scores suggest more severe depression.3
The Beck Depression Inventory (BDI) is another commonly used screening tool.14 When compared to the EPDS, the BDI has more than 20 questions and takes longer to complete and score.3 The patient is instructed to answer the questions based on their feelings within the previous 2 weeks. When the clinician scores the BDI, a higher score indicates more severe depression, with a score range of 0 to 63.15 It is important to note that the BDI includes symptoms that may be present in generalized depression but may be normal in the postpartum period. For example, the BDI includes the symptom of sleeping difficulty, which is typical for the postpartum patient and would not necessarily be a cause for concern.3
All screening tools should be used within the context of the clinical presentation of the patient. If the patient demonstrates behaviors such as tearfulness, flat affect, or nonengagement with their infant, the provider should conduct further assessment regardless of screening results.3 Screening should be implemented with adequate resources available for accurate diagnosis, effective treatment, and appropriate follow-up.16 The Council on Patient Safety in Women’s Healthcare provides the Maternal Mental Health: Perinatal Depression and Anxiety Patient Safety Bundle to facilitate the development of a standard process within the clinical setting that ensures maternal mental health is always addressed. This safety bundle has four key components: readiness, recognition and prevention, response, and reporting and systems learning.7 See the Box for website addresses and other resources for patients and providers.
|
Patient safety bundles, including postpartum depression and obstetric emergencies |
|
|
Helpline for patients facing mental health or substance abuse emergencies |
|
|
Provides information to antepartum and postpartum patients regarding engaging in pleasant activities, improving social support, and promoting healthier ways of thinking |
|
|
The Reach Out, Stand Strong, Essentials for mothers of newborns (ROSE) includes 4 sessions that provide pregnant patients with tools to prevent postpartum depression |
|
|
Helpline for patients suffering from postpartum depression |
|
|
Patient friendly, easy-to-read key points related to postpartum depression |
|
Treatment
Nonpharmacologic
The current recommendation for initial treatment of postpartum depression is psychotherapy with a mental health provider. The efficacy of psychotherapy has been validated in the general population and in postpartum patients.10,17 Additionally, a positive effect on temperament and development in children of mothers treated with psychotherapy has been noted.10 The use of psychotherapy is particularly beneficial to patients who are breastfeeding and do not desire to expose the newborn to medications through breastmilk.10
Encouraging patients to engage in physical activity during pregnancy is evolving as a possible method to decrease incidence of postpartum depression. Physical activity has been noted to decrease depression in the general population. A meta-analysis of over 20 studies established that physical activity has a positive influence on depression symptoms in pregnant and postpartum patients.18 There were multiple modalities of exercise included in the studies: aerobic, strength exercises, yoga, stretching and breathing, and balance exercises.18
In one qualitative study of postpartum mothers, women expressed that using exercise as a treatment for postpartum depression felt less stigmatizing compared to pharmacologic interventions. Despite mixed opinions regarding the efficacy of exercise for postpartum depression, it should be noted that most women also expressed that exercise during the postpartum period allowed them to have time for self-care without guilt.19
Patients should be counseled regarding appropriate frequency and intensity of exercise in pregnancy prior to initiation of any new fitness routine. The American College of Obstetricians and Gynecologists (ACOG) recommends low impact exercise; walking, swimming, stationary bikes, and yoga during pregnancy. Healthcare providers should discuss warning signs that the patient is overexerting herself, including vaginal bleeding, dizziness, shortness of breath, or fluid leaking from the vagina.20
Pharmacologic
Pharmacologic management is an option for the patient who declines to attend therapy with a mental health provider or if the patient has previously demonstrated a benefit from pharmacologic intervention, and can be used in conjunction with cognitive-based therapy. The use of selective serotonin reuptake inhibitors (SSRIs) is acceptable in the postpartum period, and this class of medication has been demonstrated to provide a statistically significant improvement in postpartum depression symptoms.21
The level of SSRIs in breast milk has been noted to be minimal, and there are few adverse effects for the infant.10 When adverse effects from postpartum use of SSRIs have been noted in infants, the effects have been nonspecific, including irritability and poor feeding.22 Parents should be alerted to the warning signs of SSRI toxicity in the breastfed infant, including lethargy, increased irritability with inability to be comforted, and impaired feeding.21 It is also important to ask the breastfeeding patient about milk quantity due to the possible risk of decreased milk production.23 When making the decision regarding choice of SSRI for treatment in the breastfeeding mother, it is important to note that the relative infant dose (RID), or ratio of infant dose to maternal dose, is lowest in sertraline and paroxetine.23 It should be noted that a patient who has been on an SSRI during pregnancy should continue on the same medication if it has been effective in an effort to decrease the number of drug exposures for the infant.10
In studies comparing treatment with SSRIs to placebo, patients in the treatment groups had higher remission rates and lower scores on the EPDS, indicating low level or no depression.10 Patients should be counseled when starting SSRIs that alleviation of symptoms can take up to 2 to 6 weeks, and medication should be taken every day.24 Side effects in the mother are not common, but can include diarrhea, headache, and nausea.22
If the patient taking an SSRI does not have remission from depression symptoms, or has minimal effect, the provider can opt to change to another medication within the SSRI drug class or can change the patient to a serotonin-norepinephrine reuptake inhibitor (SNRI). The most commonly used SNRIs include desvenlafaxine, duloxetine, and venlafaxine.10 Although there is evidence that there are no short-term adverse effects in infants, SNRIs are not recommended as first-line treatment in breastfeeding mothers.23 The RID is higher in most SNRIs than SSRIs, and it has been recommended that further studies on SNRIs in the breastfeeding mother be conducted.23
In 2019, the FDA approved brexanolone specifically for the treatment of postpartum depression. Brexanolone is a form of allopregnanolone, which acts as a positive allosteric modulator of the GABAA receptor. After delivery, the levels of endogenous allopregnanolone decline rapidly, and this is believed to contribute to postpartum depression. Brexanolone has been demonstrated to be efficacious in diverse groups of postpartum women suffering from depression.25 The medication is given intravenously over 60 hours with improvement typically occurring immediately after infusion. The patient must be monitored continuously during brexanolone administration for excessive sedation or loss of consciousness. Remission of depression after treatment has been sustained during 30 days of follow-up in clinical trials. Although brexanolone is excreted in low levels in breast milk, more studies are needed to establish safety. Cost and lack of insurance coverage limit availability with an out-of-pocket expense of approximately $34,000 for the drug. This does not include the costs incurred during infusion.26
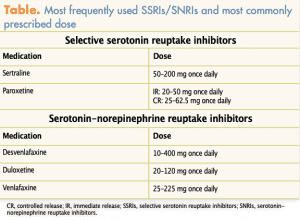
When a patient is diagnosed with postpartum depression and any pharmacologic therapy is initiated, the patient and healthcare provider should collaborate to determine if the benefits of breastfeeding outweigh the risk to the infant. Although the risk to the infant exposed to these medications is very low, this should still be discussed.10 The Table outlines the most frequently used SSRIs and SNRIs and the most commonly prescribed dose.
Prevention
Early identification and treatment of antepartum depression is recommended to reduce the incidence of postpartum depression.13 Although providers should be aware that some patients have a higher risk of developing postpartum depression, several organizations, including ACOG, American Association of Family Practice, and the Centers for Disease Control and Prevention recommend antenatal screening of all pregnant patients.13 It is also important for maternity providers to collaborate with pediatricians when there is a suspicion of postpartum depression.13
Another method for preventing postpartum depression is antenatal counseling.27 The USPSTF determined that there is a moderate benefit to counseling for pregnant women, specifically for those women who are identified as being at risk for postpartum depression.16 Researchers demonstrated in a small cohort of pregnant women that cognitive-behavioral and solution-based counseling was correlated with a lower score on the EPDS in the postpartum period.28 The use of cognitive-behavioral therapy is even more effective when the patient’s partner participates in therapy.29 These studies did not determine the timing during pregnancy or duration of counseling for best efficacy in prevention of postpartum depression, but even a brief antenatal intervention is recommended.28,29
Implications for practice
The prevalence and potentially serious adverse effects of postpartum depression warrant attention by all healthcare providers who see women during pregnancy and the first year postpartum. Repeated contact with the healthcare provider during this period of time provides for developing a trusting relationship that facilitates screening and intervention.3 Screening tools are readily available that have demonstrated reliability for use in the postpartum patient. The effectiveness of nonpharmacologic and pharmacologic treatments have been established.
All healthcare providers who see pregnant and postpartum patients should establish protocols and identify the screening tools that will be used in their clinical practice. All patients should be counseled during their pregnancy regarding signs and symptoms of postpartum depression. Patients with identified risk factors may need to be screened more frequently along with implementation of preventive interventions. Providers should also have established protocols for treatment, referrals, and follow-up so that patients receive prompt attention and have continuity of care.
Angela N. Morehead is Assistant Professor at Middle Tennessee State University in Murfreesboro. The author states that she does not have a financial interest in or other relationship with any commercial product named in this article.
References
- Sorg M, Coddington J, Ahmed A, Richards E. Improving postpartum depression screening in pediatric primary care: a quality improvement project. J Pediatr Nurs. 2019;46:83-88.
- Bina R, Harrington D. Differential predictors of postpartum depression and anxiety: the Edinburgh Postnatal Depression Scale Hebrew version two factor structure construct validity. Matern Child Health J. 2017;21(12):2237-2244.
- ACOG Committee Opinion no. 757. Screening for perinatal depression. Obstet Gynecol. 2018;132(5):e208-e212.
- Prevatt BS, Desmarais SL, Janssen PA. Lifetime substance use as a predictor of postpartum mental health. Arch Womens Ment Health. 2017;20(1):189-199.
- Bauman BL, Ko JY, Cox S, et al. Vital signs: postpartum depressive symptoms and provider discussions about perinatal depression – United States, 2018. MMWR Morb Mortal Wkly Rep. 2020;69(19):575-581.
- Kallem S, Matone M, Boyd RC, Guevara JP. Mothers’ mental health care use after screening for postpartum depression at well-child visits. Acad Pediatr. 2019;19(6):652-658.
- Kendig S, Keats JP, Hoffman MC, et al. Consensus bundle on maternal mental health: perinatal depression and anxiety. Obstet Gynecol. 2017;129(3):422-430.
- van der Zee-van den Berg AI, Boere-Boonekamp MM, IJzerman MJ, et al. Screening for postpartum depression in well-baby care settings: a systematic review. Matern Child Health J. 2017;21(1):9-20.
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, 5th ed. Arlington, VA: American Psychiatric Association; 2013.
- Viguera A. Postpartum unipolar major depression: epidemiology, clinical features, assessment, and diagnosis. UptoDate. November 20, 2018.
- Siu AL; US Preventive Services Task Force; Bibbins-Domingo K, Grossman DC, Baumann LC, et al. Screening for depression in adults: US Preventive Services Task Force recommendation statement. JAMA. 2016;315(4):380-387.
- ACOG Committee Opinion no. 736. Optimizing postpartum care. Obstet Gynecol. 2018;131(5):e140-e150.
- Rafferty J, Mattson G, Earls MF, Yogman MW; Committee on Psychosocial Aspects on Child and Family Health. Incorporating recognition and management of perinatal depression into pediatric practice. Pediatrics. 2019;143(1):1-29.
- Moraes GP, Lorenzo L, Pontes GA, et al. Screening and diagnosing postpartum depression: when and how? Trends Psychiatry Psychother. 2017;39(1):54-61.
- Lee E-H, Lee S-J, Hwang S-T, et al. Reliability and validity of the Beck Depression Inventory-II among Korean adolescents. Psychiatry Invest. 2017;14(1):3-36.
- US Preventive Services Task Force; Curry SJ, Krist AH, Owens DK, et al. Interventions to prevent perinatal depression: US Preventive Services Task Force recommendation statement. JAMA. 2019;321(6):580-587.
- Guille C, Newman R, Fryml LD, et al. Management of postpartum depression. J Midwifery Womens Health. 2013;58(6):643-653.
- Nakamura A, van der Waerden J, Melchior M, et al. Physical activity during pregnancy and postpartum depression: systematic review and meta-analysis. J Affect Disord. 2019;246:29-41.
- Pritchett R, Jolly K, Daley AJ, et al. Women’s experiences of exercise as a treatment for their postnatal depression: A nested qualitative study. J Health Psychology. 2020;25(5):684-691.
- American College of Obstetricians and Gynecologists. Patient resources. FAQs. Exercise during pregnancy. 2019. https://www.acog.org/patient-resources/faqs/pregnancy/exercise-during-pregnancy.
- Craig MC. Postnatal depression: drug treatments. BMJ Clin Evid. 2016;2016:1407.
- Kovich H, DeJong A. Common questions about the pharmacologic management of depression in adults. Am Fam Physician. 2015;92(2):94-100.
- Scrandis DA. Antidepressant use in breastfeeding women. Nurse Pract. 2018;43(10):18-20.
- Gorun A. Choosing and discussing SSRIs for depression in pregnancy: a basic guide for residents. Am J Psychiatry Residents J. 2018;13(6):3-5.
- English C, Goodwin H, Dickinson JA, Rey JA. Brexanolone: a novel treatment option for postpartum depression. PT. 2019;44(12):732-736.
- Fantasia HC. Brexanolone is the first drug specifically for postpartum depression. Nurs Womens Health. 2019;23(5):450-454.
- Langan R, Goodbred AJ. Identification and management of peripartum depression. Am Fam Physician. 2016;93(10):852‐858.
- Ramezani S, Khosravi A, Motaghi Z, et al. The effect of cognitive-behavioral and solution-focused counselling on prevention of postpartum depression in nulliparous pregnant women. J Reprod Infant Psychol. 2017;35(2):172-182.
- Ngai F-W, Wong Pw-C, Chung K-F, et al. Effect of couple-based cognitive behavioural intervention on prevention of postnatal depression: multisite randomised controlled trial. BJOG. 2020;127(4):500‐507.

