 By Mary Annette Hess, PhD, FNP-BC, CNS and W. Timothy Garvey, MD, FACE
By Mary Annette Hess, PhD, FNP-BC, CNS and W. Timothy Garvey, MD, FACE
Before reading the article, click here to take the pretest.
Obesity is a disease, not a condition resulting from ill-advised behavioral choices.1 After all, obesity meets the essential criteria of a disease: It has characteristic signs or symptoms; it manifests as an impairment in the normal functioning of some aspect of the body; and it results in harm or morbidity. As such, healthcare providers (HCPs) need to identify obesity in their patients, assess each patient’s risk for obesity-related complications, begin the weight-loss discussion in a thoughtful and constructive manner, and institute an individualized management plan.
Key words: obesity, risk assessment, obesity-related complications, dietary changes, weight-loss medications
Definitions and prevalence
Obesity can be defined as a body mass index (BMI) ?30 kg/m2 or it can be suggested by a waist circumference (WC) >35 inches (in women).2 But obesity is more than just a calculation or a measurement; it is a primary disease entity that can lead to cardiometabolic, biomechanical, and other complications (Figure 1).3,4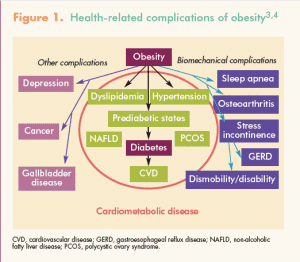
Here is a number that matters: Almost 80 million U.S. adults—almost 35% of the adult population in this country—meet criteria for obesity,5 with certain geographic areas and certain ethnic groups overrepresented. Prevalence of obesity is higher in southern states and some Midwestern states than in other parts of the country,6 as illustrated by this CDC map. Non-Hispanic blacks have the highest age-adjusted rate of obesity (47.8%), followed by Hispanics (42.5%), non-Hispanic whites (32.6%), and non-Hispanic Asians (10.8%).5 Although overall obesity prevalence is similar in women and men at any given age, women have a higher prevalence of class II obesity (BMI, 35.0-39.9) and class III obesity (BMI ?40).7
Risk assessment
Given the high prevalence of obesity, HCPs will likely encounter many patients in their practices who are candidates for weight management. In each case in which obesity is identified, the first step needed is to assess the patient’s risk for obesity-related complications. This assessment includes calculating BMI, measuring WC, and screening for the presence of cardiovascular disease (CVD) risk factors and co-morbidities.8 Compared with body weight alone, BMI is a better, albeit indirect, measure of adiposity, which is associated with a host of cardiometabolic abnormalities. WC, an indicator of abdominal adiposity, should be measured in patients with a BMI ?35 (the WC cutoff of >35 inches in women adds little predictive value in those with a BMI >35), including those who are overweight (BMI, 25-29.9). Women whose WC exceeds 35 inches are at increased risk for developing hypertension (HTN), type 2 diabetes mellitus (T2DM), and CVD.
Therefore, HCPs need to check patients’ blood pressure (BP) to assess for HTN and order a fasting blood glucose (FBG) test, and even a 2-hour oral glucose tolerance test and HbA1c in high-risk individuals, to assess for T2DM and pre-diabetes.9 The metabolic syndrome, which increases risk for T2DM, CVD, and stroke, is identified in women by the presence of at least three of these five risk factors: WC >35 inches, triglycerides ?150 mg/dL, high-density lipoprotein cholesterol (HDL-C) <50 mg/dL, BP ?130/85 mm Hg, and FBG ?100 mg/dL.10
Some obesity-associated diseases and risk factors place patients in a very-high-risk category for subsequent mortality.8 Patients with obesity and co-morbid coronary heart disease (CHD), other atherosclerotic diseases, T2DM, metabolic syndrome, pre-diabetes, or sleep apnea require aggressive modification of risk factors in addition to clinical management of the co-morbid disease. Furthermore, obesity has an aggravating effect on CVD risk factors such as cigarette smoking, HTN, high concentration of low-density lipoprotein cholesterol, low concentration of HDL-C, impaired FBG, family history of premature CHD, and age ?55 years (in women). HCPs need to identify these risk factors to determine the intensity of the clinical intervention that a patient requires.
Obesity takes a toll not just in terms of its effect on CVD risk, but also on cancer risk. The Cancer Research UK study showed that women with obesity had a 25% risk of developing a weight-related cancer—including cancer
of the bowel, gallbladder, uterus, kidney, pancreas, or esophagus, as well as post-menopausal breast cancer—in their lifetime.11 Cancer risk in these women was 40% higher than that in their slimmer counterparts.
Initiating the conversation
Either a patient or an HCP can initiate the conversation regarding the need to lose weight. The situation is generally easier to handle when a patient expresses a desire to lose weight. She has already acknowledged existence of the disease—that is, the obesity—and is seeking treatment for it on her own. However, in many cases, the HCP must broach the topic, usually after a patient has come in for a routine visit and the findings from her history, physical examination, and laboratory tests indicate that steps must be taken to lower her risk for experiencing obesity-related complications—or to treat the complications that already exist.
To avoid discomfiting a patient in this situation, a panel of nurse practitioners convened by the American Nurse Practitioner Foundation (ANPF) recommends that the HCP show her objective data reflecting her disease and her risk for future complications—with an emphasis that obesity is a health problem—and then assess her motivation and readiness for change.7 In this context, the HCP and the patient need to synchronize their expectations and goals for weight loss therapy. HCPs now have reliable tools to help patients lose 5%-10% of their body weight. This weight loss may not produce the desired cosmetic outcome but will no doubt result in clinical benefits. The emphasis is on improving the health of the patient.
To inspire a patient with obesity to want to lose weight and to commit to follow a weight-loss treatment plan, the HCP can help her identify at least one compelling reason to lose weight.7 Common patient-centered reasons include (1) decreasing the risk of having a complicated pregnancy; (2) being able to play with children or grandchildren; (3) walking without becoming short of breath; (4) preventing other chronic diseases such as T2DM; and (5) improving existing weight-related complications such as sleep apnea or T2DM. Of note, some patients may not be aware of the health risks posed by obesity and will be motivated to lose weight once educated about the risks.
Approach to management
Once a patient with obesity is motivated and ready to lose weight, the HCP needs to work with her to devise a management plan. Both of them should agree on the goals of weight-loss therapy and on the purpose of long-term therapy. A realistic initial goal for many patients is a loss of 5% of body weight in 3 months. Three major management options—lifestyle modification, pharmacotherapy, and bariatric surgery—can bring about weight loss and reduce obesity-related morbidity and mortality.1 This article focuses on the first two options.
Lifestyle modification
A comprehensive weight-loss program starts with lifestyle modification comprised of dietary changes, increased activity, and behavioral control.12
Dietary changes
With regard to energy intake, the ANPF recommends a reduction of 500-1,000 kcal/day, which can be accomplished by limiting portion size, reducing fat and sugar intake, and using commercial weight-loss meal replacements.7 The patient can follow one of many diets shown to be safe and effective; examples are a low-carbohydrate diet,13 a low-fat diet,14 a Mediterranean diet,15 a low-glycemic-load diet,16 and a portion-controlled diet.17
Practical dietary tips include avoiding skipping meals and consuming small meals and between-meal snacks every 3-4 hours. With regard to food intake, moderation is the watchword. With regard to fluid intake, however, drinking eight 8-oz glasses of water a day is crucial unless contraindicated (e.g., in patients with renal failure).
Choice of a particular diet is less important than making a commitment and adhering to the diet,18 although following a regimen tailored for a co-morbidity makes sense. Because compliance is the key to success, the diet plan should accommodate the patient’s personal and cultural preferences. Regardless of the diet chosen, patients should monitor their caloric intake via a food diary and weigh themselves at least once a week.19
Increased activity
Choice of a particular activity (e.g., walking, swimming) depends on a patient’s preference and access to, say, a pool, as well as her current weight and health status. An assessment of mobility, cardiovascular (CV) status, and perhaps pulmonary function is needed before a patient embarks on a new exercise program.20 The goal is to increase energy expenditure.20 Exercising for ?150 minutes/week can lead to modest weight loss and help prevent weight regain; doubling this amount will promote more robust weight loss.21 As with food intake, patients should record their daily physical activity.
Exercise not only facilitates weight loss but also improves CV health by reducing BP, lipid levels, and visceral fat. These reductions are linked to improved glucose tolerance and insulin sensitivity in persons without diabetes and improved glycemic control in patients with T2DM.12 Enhanced physical fitness may even lessen obesity-related mortality. Of note, patients with obesity must modify their diet and increase physical activity in order to lose weight and reduce their risk for obesity-related complications. Another note: In addition to traditional exercise, patients can aim to increase energy expenditure throughout the day by reducing sedentary behaviors. For example, car owners can park twice as far from store entrances as they used to; city dwellers can walk instead of taking a bus, subway, or taxi; and office workers can use a standing desk instead of sitting at their desk.
Behavioral therapy
As applied to weight loss, behavioral therapy entails techniques for helping patients replace habits that contribute to excess weight and poor health with those that promote weight loss and good health.12 Key components of behavioral therapy include frequent encounters with HCPs, education, stimulus control, cognitive restructuring, goal-setting, self-monitoring, and social support.20 Group weight-loss programs in community settings, commercial weight-loss programs, and programs delivered by telephone, the Internet, or text message can all be effective, depending on patient preference.12
Pharmacotherapy
If a patient has not lost about 5% of her body weight after 3 months, or if she has lost weight but regained some, most, or all of it over time, she and her HCP should consider use of weight-loss medication as an adjunct to lifestyle modification. In some patients with severe complications who require clinically meaningful weight loss quickly, lifestyle modification and pharmacotherapy can be initiated concomitantly.
Rationale
In all human beings, calorie restriction triggers various biological adaptations designed to prevent starvation.22 These adaptations may even be potent enough to reverse the initial weight-loss success achieved with lifestyle modification. In persons with obesity, additional biological adaptations function to preserve, or even increase, their highest sustained lifetime body weight. As such, more biologically-based interventions are likely to be needed to counter the compensatory adaptations that maintain a person’s highest lifetime body weight.22 Other reasons for pharmacologic intervention in facilitating weight loss include the following:
- Appetite-suppressing medication enhances a patient’s ability to adhere to a reduced-calorie diet.
- Addition of a weight-loss medication consistently achieves greater weight loss, and for a longer duration of time, than that achieved by the lifestyle intervention alone.
- Medication can help achieve the degree of weight loss needed to treat obesity-related complications.
- The American Association of Clinical Endocrinologists (AACE), the American Society of Bariatric Physicians (ASBP), the American Heart Association (AHA), the American College of Cardiology (ACC), and The Obesity Society (TOS) all recommend use of medication for patients with obesity and sufficient health risk.20,23,24
Principles for use
The FDA indication for use of weight-loss medications is a BMI ?30 or a BMI of 27-29.9 with at least one obesity-related complication. The medication should be stopped if weight loss is <5% after 12 weeks on a maximal dosage. If one agent is ineffective or poorly tolerated, a different one can be tried. All of these agents are contraindicated for use during pregnancy. Pharmacotherapy is individually tailored to each patient’s needs. More data are needed regarding the safety of combination therapy and the use of medications beyond 2 years.
Options
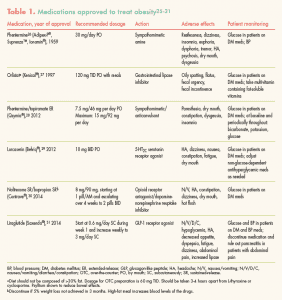
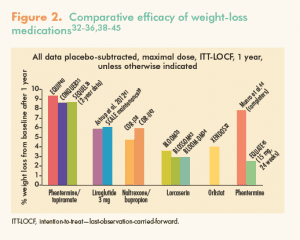
Table 1 lists FDA-approved options for treating obesity.25-31Table 2. Weight-loss medications: Clinical trial information, accessible through this link, shows the results of clinical trials demonstrating the efficacy of these agents.32-39Figure 2 illustrates the comparative efficacy of these weight-loss medications.32-36,38-45
Guidelines for practice
The spectrum of obesity treatment guidelines ranges from those that are BMI-centric, wherein treatment indication is based on BMI and the treatment goal is to lose a given amount of weight (e.g., 5%-10%), to those that are complications-centric, wherein treatment indication is based on risk for, presence of, and severity of obesity-related complications and the treatment goal is to treat or prevent the complications.46Obesity treatment guidelines from the National Heart, Lung, and Blood Institute (NHLBI), at one end of the spectrum, are based primarily on BMI and WC, although risk factors and co-morbidities are taken into account.8 The AACE, at the other end of the spectrum, recommends (1) evaluating patients with obesity for cardiometabolic and biomechanical complications; (2) selecting (a) therapeutic targets for improvements in complications, (b) treatment modality, and (c) treatment intensity; and (3) intensifying lifestyle and/or pharmacologic and/or surgical treatment modalities for greater weight loss if therapeutic targets for improvements in complications are not met.23Table 3 lists percentages of weight loss needed to achieve therapeutic benefits with regard to various obesity-related complications.4
The AHA/ACC/TOS obesity guideline, which is closer to that of the NHLBI, recommends (1) identifying patients who need to lose weight, based on BMI and WC; (2) informing patients with CVD risk factors that lifestyle changes that produce even modest sustained weight loss of 3%-5% can result in clinically meaningful health benefits, and that greater weight loss produces greater benefits; (3) devising dietary strategies for weight loss; (4) devising a comprehensive lifestyle program that helps patients adhere to a lower-calorie diet and increase physical activity through use of behavioral strategies; and (5) selecting patients for whom bariatric surgery is advised—that is, those with a BMI ?40 or a BMI ?35 with obesity-related conditions.24 The approach of the ASBP to obesity management, which is closer to that of the AACE, focuses on treating diseases related to increased body fat and its adverse metabolic and biomechanical consequences, which may improve patient health, quality of life, body weight, and body composition.20
Conclusion
Obesity is a disease that requires permanent lifestyle changes. Lifestyle modification, enabled by dietary changes, increased physical activity, and behavioral therapy, is implemented first. If a patient does not reach her goals in terms of reducing her weight and her risk for obesity-related complications, medication is added. Most medications suppress appetite, enhance a patient’s ability to follow a reduced-calorie diet, and enable significantly greater weight loss than that achieved by lifestyle changes alone. In addition, medication use can help sustain weight loss and prevent weight regain over time.
For patients with obesity and obesity-related co-morbidities, weight loss is used therapeutically to treat the obesity-related complications. The role of the HCP is to diagnose the disease of obesity, assess the patient’s risk for obesity-related complications, discuss weight-loss strategies and goals with the patient, support the patient in implementing these strategies and reaching these goals, and provide regular follow-up and encouragement over the ensuing months, years, and decades.
For readers of the online issue who wish to participate in this CE program, click here.
References
- Mechanick JI, Garber AJ, Handelsman Y, Garvey WT. American Association of Clinical Endocrinologists’ position statement on obesity and obesity medicine. Endocr Pract. 2012;18(5):642-648.
- World Health Organization. Obesity and overweight. Fact sheet No. 311. Updated January 2015. http://www.who.int/mediacentre/factsheets/fs311/en/
- Centers for Disease Control and Prevention. Overweight and Obesity. Adult Obesity Facts. Last updated September 2014. http://www.cdc.gov/obesity/data/adult.html
- Daniel S, Soleymani T, Garvey WT. A complications-based clinical staging of obesity to guide treatment modality and intensity. Curr Opin Endocrinol Diabetes Obes. 2013;20(5):377-388.
- Ogden CL, Carroll MD, Kit BK, Flegal KM. Prevalence of childhood and adult obesity in the United States, 2011-2012. JAMA. 2014;311(8):806-814.
- Centers for Disease Control and Prevention. Overweight and Obesity. Obesity Prevalence Maps. Last updated September 2014. http://www.cdc.gov/obesity/data/prevalence-maps.html
- American Nurse Practitioner Foundation. Nurse Practitioners and the Prevention and Treatment of Adult Obesity. 2013. http://anp-foundation.org/wp-content/uploads/2013/07/ObesityWhitePaperv11.pdf
- NHLBI Obesity Education Initiative. The Practical Guide: Identification, Evaluation, and Treatment of Overweight and Obesity in Adults. October 2000. www.nhlbi.nih.gov/guidelines/obesity/prctgd_c.pdf
- American Diabetes Association. Standards of Medical Care in Diabetes—2012. Diabetes Care. 2012;35:S11-S63.
- Grundy SM, Brewer HB Jr, Cleeman JI, et al. Definition of metabolic syndrome: Report of the National Heart, Lung, and Blood Institute/American Heart Association conference on scientific issues related to definition. Circulation. 2004;109(3):433-438.
- MedlinePlus. Obesity Raises Women’s Cancer Risk by 40 Percent, Study Finds. March 17, 2015. http://www.nlm.nih.gov/medlineplus/news/fullstory_151517.html
- Wadden TA, Webb VL, Moran CH, Bailer BA. Lifestyle modification for obesity: new developments in diet, physical activity, and behavior therapy. Circulation. 2012;125(9):1157-1170.
- Atkins RC. Dr. Atkins’ New Diet Revolution. New York, NY: Avon; 2002.
- Krauss RM, Eckel RH, Howard B, et al. AHA Dietary Guidelines: revision 2000: A statement for healthcare professionals from the Nutrition Committee of the American Heart Association. Circulation. 2000;102(18):2284-2299.
- Shai I, Schwarzfuchs D, Henkin Y, et al; Dietary Intervention Randomized Controlled Trial (DIRECT) Group. Weight loss with a low-carbohydrate, Mediterranean, or low-fat diet. N Engl J Med. 2008;359(3):229-241.
- Makris AP, Foster GD. Dietary approaches to the treatment of obesity. Psychiatr Clin North Am. 2005;28(1):117-139, viii-ix.
- Tsai AG, Wadden TA. The evolution of very-low-calorie diets: an update and meta-analysis. Obesity (Silver Spring). 2006;14(8):1283-1293.
- Dansinger ML, Gleason JA, Griffith JL, et al. Comparison of the Atkins, Ornish, Weight Watchers, and Zone diets for weight loss and heart disease risk reduction: a randomized trial. JAMA. 2005;293(1):43-53.
- The National Weight Control Registry. http://www.nwcr.ws/
- Seger JC, Horn DB, Westman EC, et al. American Society of Bariatric Physicians. Obesity Algorithm: Adult Adiposity Evaluation and Treatment. 2013. www.obesityalgorithm.org
- Strasser B. Physical activity in obesity and metabolic syndrome. Ann N Y Acad Sci. 2013;1281:141-159.
- Ochner CN, Tsai AG, Kushner RF, Wadden TA. Treating obesity seriously: when recommendations for lifestyle change confront biological adaptations. Lancet Diabetes Endocrinol. 2015;3(4):232-234.
- Garber AJ, Abrahamson MJ, Barzilay JI, et al; American Association of Clinical Endocrinologists. AACE comprehensive diabetes management algorithm 2013. Endocr Pract. 2013;19(2):327-336.
- Jensen MD, Ryan DH, Apovian CM, et al. American College of Cardiology/American Heart Association Task Force on Practice Guidelines; Obesity Society. 2013 AHA/ACC/TOS guideline for the management of overweight and obesity in adults: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines and The Obesity Society. J Am Coll Cardiol. 2014;63(25 pt B):2985-3023.
- U.S. Food and Drug Administration. Drugs @FDA. http://www.accessdata.fda.gov/Scripts/cder/DrugsatFDA/
- Phentermine prescribing information. Drugs.com. http://www.drugs.com/pro/phentermine.html
- Xenical prescribing information. Genentech USA, Inc. 2013. http://www.gene.com/download/pdf/xenical_prescribing.pdf
- Qsymia prescribing information. Vivus, Inc. 2012-2014. https://qsymia.com/hcp/include/media/pdf/prescribing-information.pdf
- Belviq prescribing information. Arena Pharmaceuticals. August 2012. https://www.belviqhcp.com/media/1001/belviq_prescribing_information.pdf
- Contrave prescribing information. Takeda Pharmaceuticals America, Inc. 2014. http://general.takedapharm.com/content/file.aspx?filetypecode=CONTRAVEPI&CountryCode=US&LanguageCode=EN&cacheRandomizer=ef10a935-77f9-4f19-8bfd-5eefd179c8d1
- Saxenda prescribing information. Novo Nordisk. December 2014. http://www.novo-pi.com/saxenda.pdf
- Torgerson JS, Hauptman J, Boldrin MN, Sjöström L. Xenical in the prevention of diabetes in obese subjects (XENDOS) study: a randomized study of orlistat as an adjunct to lifestyle changes for the prevention of type 2 diabetes in obese patients. Diabetes Care. 2004;27(1):155-161.
- Smith SR, Weissman NJ, Anderson CM, et al; Behavioral Modification and Lorcaserin for Overweight and Obesity Management (BLOOM) Study Group. Multicenter, placebo-controlled trial of lorcaserin for weight management. N Engl J Med. 2010;363(3):245-256.
- O’Neil PM, Smith SR, Weissman NJ, et al. Randomized placebo-controlled clinical trial of lorcaserin for weight loss in type 2 diabetes mellitus: the BLOOM-DM study. Obesity (Silver Spring). 2012;20(7):1426-1436.
- Gadde KM, Allison DB, Ryan DH, et al. Effects of low-dose, controlled-release, phentermine plus topiramate combination on weight and associated comorbidities in overweight and obese adults (CONQUER): a randomised, placebo-controlled, phase 3 trial. Lancet. 2011;377(9774):1341-1352.
- Garvey WT, Ryan DH, Look M, et al. Two-year sustained weight loss and metabolic benefits with controlled-release phentermine/topiramate in obese and overweight adults (SEQUEL): a randomized, placebo-controlled, phase 3 extension study. Am J Clin Nutr. 2012;95(2):297-308.
- Garvey WT, Ryan DH, Henry R, et al. Prevention of type 2 diabetes in subjects with prediabetes and metabolic syndrome treated with phentermine and topiramate extended release. Diabetes Care. 2014;37(4):912-921.
- Greenway FL, Fujioka K, Plodkowski RA, et al; COR-I Study Group. Effect of naltrexone plus bupropion on weight loss in overweight and obese adults (COR-I): a multicentre, randomised, double-blind, placebo-controlled, phase 3 trial. Lancet. 2010;376(9741):595-605.
- Wadden TA, Hollander P, Klein S, et al; NN8022-1923 Investigators. Weight maintenance and additional weight loss with liraglutide after low-calorie-diet-induced weight loss: the SCALE Maintenance randomized study. Int J Obes (Lond). 2013;37(11):1443-1451.
- Allison DB, Gadde KM, Garvey WT, et al. Controlled-release phentermine/topiramate in severely obese adults: a randomized controlled trial (EQUIP). Obesity (Silver Spring). See comment in PubMed Commons below2012;20(2):330-342.
- Astrup A, Carraro R, Finer N, et al; NN8022-1807 Investigators. Safety, tolerability and sustained weight loss over 2 years with the once-daily human GLP-1 analog, liraglutide. Int J Obes (Lond). 2012;36(6):843-854.
- Apovian CM, Aronne L, Rubino D, et al; COR-II Study Group. A randomized, phase 3 trial of naltrexone SR/bupropion SR on weight and obesity-related risk factors (COR-II). Obesity (Silver Spring). 2013;21(5):935-943.
- Fidler MC, Sanchez M, Raether B, et al; BLOSSOM Clinical Trial Group. A one-year randomized trial of lorcaserin for weight loss in obese and overweight adults: the BLOSSOM trial. J Clin Endocrinol Metab. 2011;96(10):3067-3077.
- Munro JF, MacCuish AC, Wilson EM, Duncan LJ. Comparison of continuous and intermittent anorectic therapy in obesity. Br Med J. 1968;1(5588):352-354.
- Ryan DH, Peterson C, Troupin B, et al. Weight loss at 6 months with V1-0521 (PEN/TPM combination) treatment. Presented at: 69th Annual Scientific Sessions of the American Diabetes Association; June 5, 2009.
- Garvey WT. New tools for weight-loss therapy enable a more robust medical model for obesity treatment: rationale for a complications-centric approach. Endocrine Pract. 2013;19(5):864-874.
