Faculty:
Alisa Pascale, DNP, WHNP-BC, is a women’s health nurse practitioner at the Vulvovaginal Disorders Program & Gynecology at Massachusetts General Hospital and Clinical Instructor at MGH Institute of Health Professions, both in Boston, Massachusetts.
Intended audience: This continuing education (CE) activity has been designed to meet the educational needs of nurse practitioners who provide care for women of any age.
CE approval period: Now through December 31, 2019
Estimated time to complete this activity: 1 hour CE approval hours: 1.0 contact hours, including 0.5 contact hours of pharmacology credit (NCC code 2A)
Goal statement: To understand the abnormal vaginal ecosystem in women prone to bacterial vaginosis (BV) and to use current evidence and guidelines in treating single episodes of BV and in reducing chronic/recurrent episodes of BV.
Needs assessment: This activity for Women’s Healthcare is based on a CE presentation developed by the author and presented at the NPWH annual conference held in Seattle, Washington, in October 2017. In this article, the author provides background information on BV (e.g., prevalence, risk factors, adverse sequelae, characteristics of a healthy vagina) and then focuses on the ABCDs of BV: abnormal flora, bothersome symptoms, chronicity, and the differential diagnosis.
Educational objectives: At the conclusion of this educational activity, participants should be able to:
- Describe normal vaginal flora and the alterations that result in episodic/chronic/recurrent BV.
- Discuss adverse sequelae of BV in nonpregnant and pregnant women.
- Differentiate between BV and other conditions that cause alterations of vaginal flora.
- List treatment options for episodic/chronic/recurrent BV.
Accreditation statement: This activity has been evaluated and approved by the Continuing Education Approval Program of the National Association of Nurse Practitioners in Women’s Health (NPWH), and has been approved for 1.0 contact hours, including 0.5 contact hours of pharmacology credit.
Faculty disclosures: NPWH policy requires all faculty to disclose any affiliation or relationship with a commercial interest that may cause a potential, real, or apparent conflict of interest with the content of a CE program. NPWH does not imply that the affiliation or relationship will affect the content of the CE program. Disclosure provides participants with information that may be important to their evaluation of an activity. Faculty are also asked to identify any unlabeled/unapproved uses of drugs or devices made in their presentation.
Alisa Pascale, DNP, WHNP-BC, disclosed that she served as a consultant and speaker for Symbiomix Therapeutics in 2016.
Disclosure of unlabeled use: NPWH policy requires authors to disclose to participants when they are presenting information about unlabeled use of a commercial product or device or an investigational use of a drug or device not yet approved for any use.
Disclaimer: Participating faculty members determine the editorial content of the CE activity; this content does not necessarily represent the views of NPWH. This content has undergone a blinded peer review process for validation of clinical content. Although every effort has been made to ensure that the information is accurate, clinicians are responsible for evaluating this information in relation to generally accepted standards in their own communities and integrating the information in this activity with that of established recommendations of other authorities, national guidelines, FDA-approved package inserts, and individual patient characteristics.
Successful completion of the activity: Successful completion of this activity, J-18-04, requires participants to:
- Log on to npwh.org/courses/home/details/1156 and “Sign In” at the top right-hand corner of the page if you have an NPWH account. You must be signed in to receive credit for this course. If you do not remember your username or password, please follow the “Forgot Password” link and instructions on the sign-in page. If you do not have an account, please click on “Create an Account.”*
- Read the learning objectives, disclosures, and disclaimers on the previous page.
- Study the material in the learning activity during the approval period (now through December 31, 2019).
- Complete the post-test and evaluation. You must earn a score of 70% or better on the post-test to receive CE credit.
- Print out the CE certificate if successfully completed.
*If you are an NPWH member, were once a member, or have taken CE activities with NPWH in the past, you have a username and password in our system. Please do not create a new account. Creation of multiple accounts could result in loss of CE credits as well as other NPWH services. If you do not remember your username or password, please either click on the “Forgot Username” or “Forgot Password” link or call the NPWH office at (202) 543-9693, ext. 1.
Commercial support: The content for this article was supported by an educational grant from Symbiomix Therapeutics.
Alterations in vaginal microflora cause a vaginal dysbiosis that can lead to asymptomatic or symptomatic bacterial vaginosis (BV). Because the underlying mechanism of BV is not well understood and long-term restoration of the normal vaginal flora can be challenging, BV often recurs or becomes chronic despite initially successful treatment. Healthcare providers (HCPs) caring for women should be familiar with the diagnosis and treatment of BV, as well as the management of recurrent BV. Because BV is sometimes mistaken for other vaginal conditions, HCPs should be alert for these other diagnoses, particularly in women with chronic or recurrent symptoms.
Bacterial vaginosis (BV) is a common condition that results from a shift in the balance of a woman’s vaginal microflora. BV is manifested by a decrease in predominantly hydrogen peroxide-producing lactobacilli and an increase in anaerobic bacteria. The depletion of lactobacilli leads to a rise in vaginal pH, and enzymes produced by the anaerobes lead to some of the classic symptoms associated with BV. Before the article’s main focus on the ABCDs of BV—abnormal flora, bothersome symptoms, chronicity, and differential diagnosis—background information on BV prevalence, risk factors, and adverse sequelae is provided and the characteristics of a healthy vaginal environment are described.
Background information
Prevalence of BV varies widely from country to country, from region to region within the same country, and even within similar population groups.1 Five decades of intense research have established many risk factors for BV acquisition, but because of the condition’s complexity and the lack of a reliable animal model for studying it, its exact etiology remains elusive.2
Prevalence
A systematic review by Kenyon et al.3 suggested that BV prevalence ranged from 6% to 51% in the United States, depending on race, ethnicity, and geographic area. Based on a representative sample of U.S. women who participated in the National Health and Nutrition Examination Survey (NHANES) 2001-2004, overall BV prevalence in this country was 29% among women aged 14-49 years,4,5 making it the most common vaginal infection in this age group.6 According to this NHANES survey, non-Hispanic white women had lower rates of BV (23%) than did African American women (51%) or Mexican American women (32%).4
Risk factors
A systematic review and meta-analysis showed that BV was significantly associated with sexual contact with new and multiple male and/or female partners.7 The precise relationship between sexual activity and BV development is not known.8 General consensus among vaginitis experts is that BV can be sexually associated, but that it is not considered to be sexually transmitted at this time. Two studies showed that women with genital herpes or HIV infection had an increased risk of developing BV.9,10 Also, BV acquisition has been associated with douching.5
Adverse sequelae
Many studies have shown BV to be a risk factor for acquiring HIV infection, herpes, gonorrhea, chlamydia, and trichomoniasis.11-14 BV also may play a role in the development of pelvic inflammatory disease and cervicitis,15,16 as well as in the persistence of human papillomavirus infection17 and in the development of cervical precancerous lesions.18 In pregnant women, BV may increase the risk for miscarriage, chorioamnionitis, preterm birth, and postpartum endometritis.19-23 BV was reported to be 3 times more prevalent among infertile women than fertile women, and it doubled the risk for pregnancy loss following in vitro fertilization-embryo transfer. 24 Of note, Nasioudis et al.25 posited that most links between BV and adverse pregnancy outcomes have been derived from inadequately designed studies that did not fully evaluate other causes of pregnancy-related pathology.
Because BV is asymptomatic in many cases and because its presence increases the risk for a variety of adverse sequelae in pregnant women, healthcare providers (HCPs) may wonder about screening routinely for BV in this population. At present, the U.S. Preventive Services Task Force, the American College of Obstetricians and Gynecologists, and the CDC do not recommend routine screening for BV in asymptomatic pregnant women.26-28
This recommendation is based on evidence indicating that although treatment of BV in pregnant women can eradicate the infection, it has not been shown to decrease preterm birth rates.29 Early screening and treatment for BV may be considered in women at high risk for preterm birth, although no clear criteria/characteristics have been defined. Pregnant women with any vulvovaginal complaints or symptoms should be evaluated for BV and treated if BV is present.
Characteristics of a healthy vagina
A healthy vagina’s microbiota is characterized by a dominance of lactobacilli, which maintain the acidic vaginal pH at 4.0-4.5. The makeup of a healthy vagina in one woman may diff er from that in another. The vagina may be colonized by one or more species of lactobacilli, including Lactobacillus crispatus, L. gasseri, L. iners, and L. jensenii.30 Furthermore, the composition of the vaginal microbiota is not static; many women experience large variations within a single menstrual cycle or between successive cycles, as well as over time.25 Hydrogen peroxide and lactic acid produced by these lactobacilli and other vaginal flora enhance the antimicrobial immune response.
Some healthy women have low numbers of vaginal lactobacilli and high numbers of other lactic acid-producing bacteria and/or variable concentrations of anaerobic bacteria that have been associated with BV.30 The percentage of apparently healthy asymptomatic women with a vaginal microbiota not dominated by lactobacilli is higher among women with African and/or Hispanic heritage, who also have a higher vaginal pH than do white or Asian women.30
Abnormal vaginal flora
Although Gardnerella vaginalis is the best known pathogen linked to BV, the condition is associated with at least a dozen other species (spp) as well, including Atopobium vaginae, Ureaplasma urealyticum, Mycoplasma spp, and others.31 Absence of localized inflammation associated with infection by any of these bacteria is the basis for the term vaginosis rather than vaginitis.
Vaginal biofi lms are well-described microbial communities embedded in a self-produced extracellular matrix to which other species also can adhere.32,33 In one study, G. vaginalis comprised 90% of bacteria in the biofilm and A. vaginae accounted for most of the remainder.34 This biofilm makes abnormal flora more resistant both to the vagina’s own natural defenses and to antibiotics, likely accounting for the frequency with which women have persistent and recurrent infections.35
Bothersome symptoms
Many women with BV are asymptomatic8 and do not learn of this diagnosis until they undergo a routine gynecologic examination by an HCP who notes the typical signs. But many women with BV do have signs and symptoms, typically a thin white or gray vaginal discharge; itching or burning in the vagina; a strong fish-like odor, especially after sex; burning when urinating; and/or itching around the outside of the vagina.8 Many women associate BV onset with recent sexual activity,36 which can cause embarrassment and self-consciousness and prompt some to change or limit their sexual relationships or activities. Self-help remedies such as douching may only exacerbate the problem.37
Making the diagnosis
The telephone is not an effective tool for diagnosis of BV. Because patient self-diagnosis and telephone triage diagnosis are notoriously inaccurate,38 women experiencing any of the aforementioned vaginal signs/symptoms, particularly recurrent symptoms, should see their HCP for an evaluation and clinical diagnosis.
Physical examination
On physical exam of women who may have BV, the external genitalia usually appear normal, although a thin milky discharge may be present at the introitus. The characteristic vaginal discharge is thin, homogeneous, and white, gray, or even yellow. A fishy/amine-positive odor may be perceptible. In many women with BV, physical exam findings can appear normal.
Office-based testing
In the clinical setting, BV diagnosis is made based on Amsel criteria39:
- Vaginal pH >4.5;
- Homogeneous white, gray, or even yellow (milky) discharge;
- Release of an amine (fi shy) odor after addition of 10% potassium hydroxide (KOH) solution to the vaginal fluid; and
- Presence of clue cells on saline wet prep microscopy.
At least three of these four criteria must be met to make the diagnosis. Vaginal pH paper, saline, KOH, slides, and a microscope are all that are needed to make a quick office diagnosis of BV. This testing is inexpensive and can often yield a diagnosis at the time of the visit, facilitating treatment.
Other laboratory tests
Several commercial products such as the BD Affirm™ VPIII Microbial Identification System and the OSOM® BVBlue® point-of-care testing can identify the microbes present in a patient’s vaginal fluid. Use of such a product adds cost to the visit and can delay diagnosis. Of note, BV diagnosis should not be made solely on the basis of a positive G. vaginalis culture because this bacterium is present in ~50%- 80% of healthy, asymptomatic women.40,41 Positive test results should be interpreted in the context of the entire clinical picture to make the diagnosis.
Treating symptoms and infection
According to the CDC, the benefits of BV therapy in nonpregnant women are symptom relief and infection cure.42 Another potential benefit is a reduction of the risk of acquiring a sexually transmitted infection.
Multiple-dose regimens
For a single episode of BV, the CDC recommends these regimens42:
- metronidazole 500 mg orally twice daily for 7 days OR
- metronidazole gel 0.75%, 1 full applicator (5 g) intravaginally, once daily for 5 days OR
- clindamycin cream 2%, 1 full applicator (5 g) intravaginally at bedtime for 7 days.
Alternative regimens include the following42:
- tinidazole 2 g orally once daily for 2 days OR
- tinidazole 1 g orally once daily for 5 days OR
- clindamycin 300 mg orally twice daily for 7 days OR
- clindamycin ovules 100 mg intravaginally once at bedtime for 3 days.
With regard to treatment with nitroimidazoles such as metronidazole and tinidazole, users should abstain from alcohol use for 24 hours after completion of the antibiotic regimen to avoid the chance of a disulfi ram-like reaction. Clindamycin cream and ovules are oil based and might weaken latex condoms and diaphragms for 3-5 days after the regimen is completed.
Single-dose regimens
Because a medication regimen requiring fewer doses may improve adherence, four different single-dose regimens are available. Single-dose intravaginal regimens include clindamycin 2% cream (Clindesse®) and metronidazole gel 1.3% (Nuvessa®); these intravaginal products are not recommended for pregnant women and are less effective than multiple-dose regimens. 43,44 New in 2018 is secnidazole 2g (Solosec™), a novel, single-dose oral product. Secnidazole is formulated as a packet of granules that are sprinkled onto applesauce, yogurt, or pudding and then consumed. 45,46 Unlike metronidazole, this product has no warning to avoid alcohol consumption. An FDA pregnancy category has not yet been assigned for secnidazole. Although single-dose treatments are well liked by patients, they tend to increase out-of-pocket cost.
Chronic/recurrent BV
Chronic or recurrent BV is defined as three or more episodes per year.47 The recurrence rate may be as high as 80% in some populations.48 As discussed previously, the underlying mechanism of the shift to abnormal flora in BV is not well understood. In addition, to date, no treatments target the biofilm, leading to challenges in resolving chronic cases. Women express considerable frustration, embarrassment, and distress with recurrences and the need for repeated or ongoing treatment.37 Until novel treatment options come along, treatment regimens for recurrent BV aim to first treat the current infection and then suppress recurrence(s). HCPs should treat the current infection as per CDC guidelines—that is, with one of the recommended or alternative regimens.
Some experts suggest extending the initial course of oral metronidazole from 7 to 10 days in patients with recurrent BV.49 The addition of boric acid (compounded 600 mg vaginally x21 days) to the initial course of oral nitroimidazole may improve results.49,50 Another option is high-dose metronidazole (compounded 750 mg vaginally x 7 days), which has a higher cure rate than does the 500-mg dose.51 Once the current infection is treated, a regimen of metronidazole vaginal gel 2%, 1 applicator intravaginally twice weekly for 4-6 months, has shown efficacy in suppressing or preventing recurrences.31 All of the regimens discussed in this paragraph are prescribed off label.
Because BV is a sexually associated infection, condom use may prevent BV recurrences and should be suggested to patients with recurrent BV. Probiotics are popular with patients; two particular lactobacilli strains—L. rhamnosus and L. reuteri—taken orally for 30 days, may help reduce BV recurrences.52 However, strong evidence of lactobacilli benefit for the treatment or prevention of recurrent BV is lacking. Novel agents that disrupt the vaginal biofilm, including antiseptics, probiotics/prebiotics, plant-derived compounds, natural antimicrobials, acidifying/buffering agents, and DNases, are being investigated as treatments for recurrent BV.53 Marrazzoetal.54 have described a novel boric acid-based vaginal anti-infective with enhanced anti-biofilm activity (TOL-463) that may show promise for treating recurrent BV in the future.
Differential diagnosis
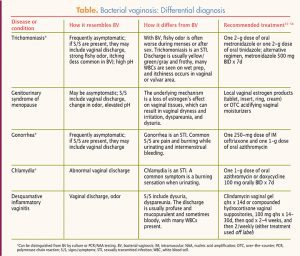
Bacterial vaginosis is distinctive in terms of the characteristics of the vaginal discharge, the fi shy odor, the elevated vaginal pH, and the presence of clue cells on wet prep. Nevertheless, other conditions and diseases bear similarities to BV and may need to be ruled out. For example, the genitourinary syndrome of menopause may present with an elevated vaginal pH and a shift in vaginal flora. Correction of the underlying low estrogen state with local estrogen or acidic vaginal moisturizers may sometimes correct the vaginal dysbiosis without a need for antibiotics. Coexistence of candidiasis along with BV, or as a result of antibiotic treatment for BV, should be considered. Desquamative inflammatory vaginitis is a less well known condition that can be confused with BV because it causes pH elevation, vaginal discharge, and loss of lactobacilli. Disease entities and conditions that may need to be considered because of their overlapping symptomatology or their coexistence with BV are listed in the Table, along with recommended treatments.55-58
Conclusion
Many woman are unfamiliar with the condition of BV, which is far more common than women or their HCPs may realize. BV involves a disruption in healthy vaginal microflora and is not always symptomatic, but it can have major adverse sequelae. In addition, recurrence rates are high and are quite bothersome for many women. Standard treatment includes metronidazole or clindamycin, although new treatments are emerging. More research is needed for both better understanding of BV pathogenesis and new and novel treatment options.
References appear in the downloadable PDF above and below.

