The National Association of Nurse Practitioners in Women’s Health (NPWH) supports a concerted effort to continue to improve cervical cancer screening (CCS) rates and timely, appropriate follow-up and treatment when screening results are abnormal. The goal is to reduce cervical cancer incidence, morbidity, and mortality. NPWH supports ongoing research to ensure that screening and treatment guidelines are based on the best evidence available. Furthermore, NPWH supports policies at the local, state, and federal levels that ensure access to CCS services and follow-up as needed.
Background
At one time, cervical cancer was one of the most common causes of cancer death for women in the United States. However, over a period of four decades, widespread implementation of CCS led to a significant decrease in incidence of and mortality from cervical cancer. In 1975, the cervical cancer incidence rate was 14.8 and the mortality rate was 5.5 per 100,000 women. Forty years later in 2015, the incidence rate was 6.6 and the mortality rate had dropped to 2.25 per 100,000 women.1
It is well established that high-risk types of the human papillomavirus (HPV) are the causative agents in more than 90% of cervical cancers.2,3 HPV infection, whether caused by a high-risk or a low-risk type, is usually transient, resolving on its own within 24-36 months in most women (>90%).4,5 It is persistent infection with high-risk HPV types that can lead to development of precancerous lesions/cervical intraepithelial neoplasia (CIN). Although precancerous lesions, especially those less than CIN grade 3 (CIN3), may regress spontaneously, they may also progress to invasive cervical cancer. Progression of a CIN3 lesion to cervical cancer typically takes more than 10 years.3 The relatively long time period from persistent HPV infection to the development of cervical cancer provides an opportunity to screen for both the presence of high-risk HPV and precancerous lesions.2,4,5
This understanding of the natural history of HPV infection and cervical cancer has been a driving force in the ongoing development of technological advances and evolving guidelines for CCS and follow-up for abnormal screening results. Yet, even with this progress, the National Cancer Institute estimates that 13,800 new cases of invasive cervical cancer will be diagnosed in the U.S. in 2020 and that 4,290 deaths from cervical cancer will occur.1 Most cases of cervical cancer develop in women who have not been adequately screened or who are lost to follow up when screening results were abnormal.2 It is important to note that the burden of cervical cancer incidence and mortality falls disproportionately on Black and Hispanic women despite similar screening rates as non-Hispanic White women.6 Both removing barriers to screening and ensuring access to timely, quality follow up and treatment when screening results are abnormal must be primary goals.
Populations identified as having lower rates of CCS than the general population include women with lower socioeconomic status, immigrants, women living in rural areas, American Indian/Native Alaskan women, women with physical and/or intellectual disabilities, lesbians, and transgender males.6-15 Multiple factors, in many cases co- existent, lead to these disparities. As a result, multifaceted approaches to increasing CCS rates are needed.
Barriers to CCS for these populations entail financial, logistical, linguistic, and cultural factors, as well as misperceptions or lack of knowledge about screening and cancer.8-15 In addition, individuals from some of these populations may not seek CCS because of unpleasant encounters and discrimination previously experienced in healthcare settings.11-15 These same barriers extend beyond screening to receiving appropriate follow-up and treatment for abnormal findings. Ongoing research is needed to understand these barriers and to explore effective strategies to reach women who are inadequately screened, as well as to address follow-up and treatment concerns.
Recent study data have demonstrated some promising innovations. Women who are under-screened or unscreened because of barriers such as embarrassment, discomfort, inconvenience, or lack of access may be receptive to self- sampling for high-risk HPV. 15-18 Availability of trained patient navigators to address individual barriers to follow-up and treatment for abnormal findings has also shown favorable results.19-22 Use of telecolposcopy has the capacity to provide critical timely follow-up for women who lack easy access to it because of their location and the cost and time for travel to
a distant setting. 23-25 Nurse practitioners (NPs) providing care for women are in an ideal position to implement and evaluate outcomes for these and other evidence-based strategies.
Recommendations regarding which test should be used for primary screening (eg, Pap test alone, HPV test alone, co-testing), which sampling methods are optimal, and how services are best delivered will likely continue to evolve as technology advances and more data become available. What is certain is that CCS performed on a regular basis, with access to follow-up and early treatment, reduces cervical cancer morbidity and mortality.
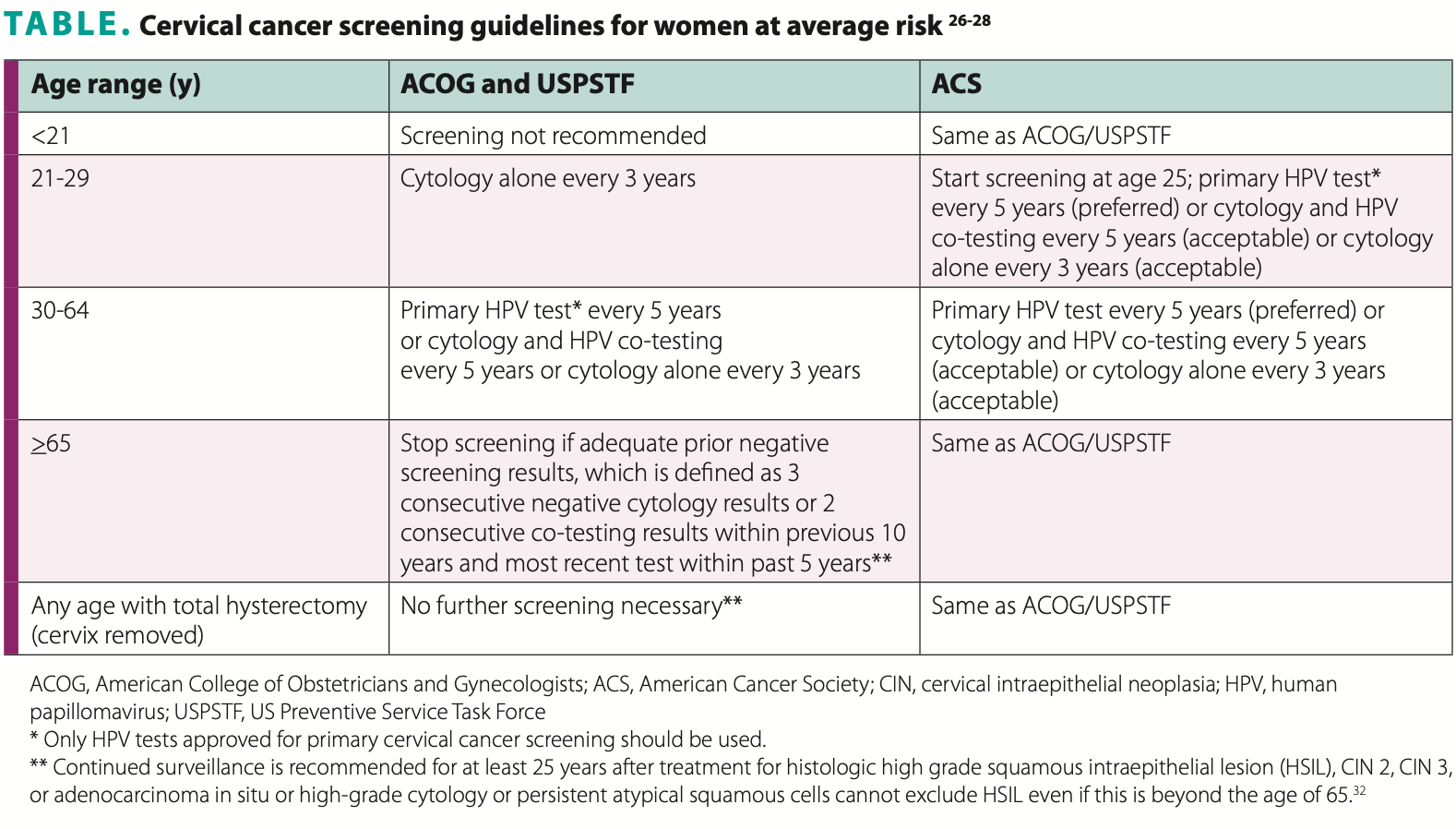
CCS guidelines by age group are listed in Table.26-28 Of note, these guidelines are intended for women at average risk for developing cervical cancer. Women with certain additional risk factors may require a different screening schedule than that recommended for the general population. Women at higher than average risk include those infected with HIV or who are otherwise immunocompromised, those who were exposed to diethylstilbestrol in utero, and those previously treated for CIN grade 2 or higher. The same CCS recommendations apply to any individual with a cervix, regardless of gender identity.26-28
Implications for women’s healthcare and NP practice
Identifying populations within one’s own community that are facing barriers to cervical cancer screening and follow-up is essential as a first step. Use of a variety of evidence- based strategies can reduce barriers and facilitate preventive healthcare for these populations. NPs can participate in community-based approaches to reach vulnerable populations with culturally appropriate education focused on addressing misperceptions and lack of knowledge about screening and cervical cancer.
Women may be uncertain about the appropriate screening frequency and type of test for their age group as recommendations change and vary. Women who have received HPV vaccination may have the misconception that they have ensured themselves lifelong immunity to HPV infection and that they can forgo regular CCS. It is crucial for NPs to provide women with evidence-based information in a way that is meaningful to them. This facilitates client-centered care and shared decision making in promoting health and preventing disease.
For women aged 65 years or older, careful review of their health histories is needed to confirm that they meet certain criteria before discontinuing CCS. A large study showed
that most cervical cancers in women aged 65 or older occurred among those who had not met criteria for stopping screening.29 An analysis of 2013 and 2015 National Health Interview Surveys data indicated that 12.1% of women aged 41-45 years and 18.4% of women aged 61-65 years had never had CCS or that their most recent screening was greater than 5 years ago.30 NPs should not make assumptions that a woman has undergone recommended screenings prior to age 65.
Effective reminder and follow-up systems are crucial. NPs who provide women’s healthcare must be innovative in designing and implementing reminder systems that reach all patients and engage them to return for both routine screening and any additional follow-up needed. NPs who provide women’s healthcare are also in an opportune position to participate in surveillance to track outcomes of screening and follow-up strategies. The data obtained can drive informed decision making about what works to improve CCS and to improve service delivery.31
Improving CCS rates and HPV vaccination rates go hand in hand to reduce the incidence of cervical cancer. NPs must also take steps in their clinical practice to identify patients who need HPV vaccination, strongly recommend this vaccination, provide the vaccination on the same day that the need is identified, and use reminder systems to ensure patients return to the office to complete the vaccination series. Useful resources are available in Box.
BOX: Useful Resources |
|
2019 ASCCP Risk Based Management Consensus Guidelines |
|
CDC National Breast and Cervical Cancer Early Detection Program (NBCCEDP) https://www.cdc.gov/cancer/nbccedp/index.htm |
|
CDC Increasing Population-based Breast and Cervical Cancer Screenings: An Action Guide to Facilitate Evidence-based Strategies. Atlanta, GA: CDC, U.S. Department of Health and Human Services; 2014. https:/stacks.cdc.gov/view/cdc/40706 |
|
NPWH Well-Woman Visit App |
|
National HPV Vaccination Roundtable |
NPs who provide healthcare for women aged 21 years or older should:
- Identify those populations in the community they serve who are at risk for not receiving regular CCS and follow-up.
- Advocate for culturally appropriate outreach to populations in the community they serve who are at risk for not receiving regular CCS and follow-up.
- Create healthcare environments that are welcoming and nonjudgmental and that promote a comfortable, affirming CCS experience.
- Follow current CCS guidelines.
- Educate patients about current CCS guidelines
- Utilize effective reminder and follow-up systems.
- Establish resources for referral and treatment.
- Confirm the status of every patient aged 65 years or older to determine whether the criteria are met to discontinue CCS.
- Advocate for accessible and affordable CCS services.
- Participate in surveillance programs to track outcomes of CCS and follow-up strategies.
- Recommend and provide HPV vaccination when indicated.
NPWH will provide leadership and resources to ensure that:
- Continuing education programs are available for NPs to learn about evidence-based strategies to improve CCS rates as well as timely follow-up and treatment.
- NPs have resources to develop and/or implement community-based approaches to reach vulnerable populations for cervical cancer awareness, screening, and follow-up.
- Timely updates are provided on CCS guidelines.
- NPs have an opportunity to participate in surveillance programs to obtain data on what works to improve CCS rates.
- Research moves forward in all aspects of cervical cancer prevention, screening, and treatment to improve healthcare outcomes.
- Policies support equitable access to CCS, appropriate follow-up, and treatment when needed
References
1. National Cancer Institute Surveillance, Epidemiology, and End Results (SEER) Program. Cancer Stat Facts: Cervical Cancer. 2018. seer.cancer.gov/statfacts/html/cervix.html
- Melinkow J, Henderson JT, Burda BU, et al. Screening for Cervical Cancer with High-Risk Human Papillomavirus Testing: A Systematic Evidence Review for the U.S. Preventive Services Task Force: Evidence Synthesis No. 158. Rockville, MD: Agency for Healthcare Research and Quality. 2018. Publication No. 15-05224 -EF-1.
- Moscicki AB, Schiffman M, Burchell A, et al. Updating the natural history of human papillomavirus and anogenital cancers. Vaccine. 2012;30(suppl 5):F24-F33.
- Insinga RP, Dasbach EJ, Elbasha EH, et al. Incidence and duration of cervical human papillomavirus 6, 11, 16, and 18 infections in young women: an evaluation from multiple analytic perspectives. Cancer Epidemiol Biomarkers Prev. 2007;16(4):709-715.
- Plummer M, Schiffman M, Castle PE, et al. A 2-year prospective study of human papillomavirus persistence among women with a cytological diagnosis of atypical squamous cells of undetermined significance or low-grade squamous intraepithelial lesion. J Infect Dis. 2007;195(11):1582-1589.
- U.S. Department of Health and Human Services. Office of Minority Health. Minority population profiles. October 2, 2018. https://minorityhealth.hhs.gov/omh/browse.aspx?lvl=2&lvlID=26
- White A, Thompson TD, White MC, et al. Cancer Screening Test Use — United States, 2015. Morb Mortal Wkly Rep. 2017;66(8):201-206.
- Akinlotan M, Bolin JN, Helduser J, et al. Cervical cancer screening barriers and risk factor knowledge among uninsured women. J Community Health. 2017;42(4):770-778.
- Adunlin G, Cyrus JW, Asare M, Sabik L. Barriers and facilitators to breast and cervical cancer screening among immigrants in the United States. Journal of Immigrant and Minority Health. 2019;21:606- 658.
- Schoenberg NE, Studts CR, Hatcher-Keller J, et al. Patterns and determinants of breast and cervical cancer non-screening among Appalachian women. Women Health. 2013;53(6):552-571.
- Andresen EM, Peterson-Besse JJ, Krahn GL, et al. Pap, mammography, and clinical breast examination screening among women with disabilities: a systematic review. Women’s Health Issues. 2013;23(4):e205-e214.
- Parish SL, Swaine JG, Son E, Luken K. Determinants of cervical cancer screening among women with intellectual disabilities: evidence from medical records. Public Health Rep. 2013;128(6):519-526.
- Tracy JK, Schluterman NH, Greenberg DR. Understanding cervical cancer screening among lesbians: a national survey. BMC Public Health. 2013;13(442):1-9.
- Peitzmeier SM, Khullar S, Reisner SL, Potter J. Pap test use is lower among female-to-male patients than non-transgender women. Am J Prev Med. 2014;47(6):808-812.
- Reiter PL, McRee AL. Cervical cancer screening (Pap testing) behaviours and acceptability of human papillomavirus self-testing among lesbian and bisexual women aged 21–26 years in the USA. J Fam Plann Reprod Health Care. 2015;41:259–264
- Crosby RA, Hagensee ME, Vanderpool R, et al. Community-based screening for cervical cancer: a feasibility study of rural Appalachian women. Sex Transm Dis. 2015;42(11):607-611.
- Penaranda E, Molokwu J, Hernandez I, et al. Attitudes toward self-sampling for cervical cancer screening among primary care attendees living on the US-Mexico border. South Med J. 2014;107(7);426-432.
- Winer RL, Gonzales AA, Noonan CJ, et al. Assessing acceptability of self-sampling kits, prevalence, and risk factors for human papillomavirus infection in American Indian women. J Community Health. 2016;41(5):1049-1061.
- Freund KM, Battaglia TA, Calhoun E, et al. Impact of patient navigation on timely cancer care: the Patient Navigation Research Program. J Natl Cancer Inst. 2014;106(6):1-9.
- Luckett F, Pena N, Vitonis A, et al. Effect of patient navigator program on no-show rates at an academic referral colposcopy clinic. J Womens Health (Larchmt). 2015;24(7):608-615.
- Paskett ED, Dudley D, Young GS, et al. Impact of patient navigation interventions on timely diagnostic follow up for abnormal cervical cancer screening. J Womens Health (Larchmt). 2016;25(1):15-21.
- Simon MA, Tom LS, Nonzee NJ, et al. Evaluating a bilingual patient navigation program for uninsured women with abnormal screening tests for breast and cervical cancer: implications for future navigator research. Am J Public Health. 2015;105(5):e87-e94.
- Hitt WC, Low GM, Lynch CE, et al. Application of a telecolposcopy program in rural settings. Telemed J E Health. 2016;22(10):816-820.
- Hitt WC, Low G, Bird TM, Ott R. Telemedical cervical cancer screening to bridge Medicaid service gap for rural women. Telemed J E Health. 2013;19(5):403-408.
- Lee S, Hitt WC. Clinical applications of telemedicine in gynecology and women’s health. Obstet Gynecol Clin N Am. 2020;47(2):259–270.
26. American College of Obstetricians and Gynecologists. Updated cervical cancer screening guidelines practice advisory. April 2021. https://www.acog.org/clinical/clinical-guidance/practice-advisory/ articles/2021/04/updated-cervical-cancer-screening-guidelines.
27. U.S. Preventive Services Task Force. Screening for cervical cancer: U.S. Preventive Services Task Force recommendation statement. JAMA. 2018;320(7):674-686.
28. Fontham ETH, Wolf AMD, Church TR. Cervical cancer screening for individuals at average risk: 2020 guideline update from the American Cancer Society. CA Cancer J Clin. 2020;0:1-26.
29. Dinkelspiel H, Fetterman B, Poitras N, et al. Screening history preceding a diagnosis of cervical cancer in women age 65 and older. Gynecol Oncol. 2012;126(2):203-206.
30. White MC, Shoemaker ML, Benard VB. Cervical cancer screening and incidence by age: unmet needs near and after the stopping age for screening. Am J Prev Med. 2017;53(3):392-395.
31. CDC. Increasing Population-based Breast and Cervical Cancer Screenings: An Action Guide to Facilitate Evidence-based Strategies. Atlanta, GA: CDC, U.S. Department of Health and Human Services; 2014. cdc.gov/cancer/nbccedp/pdf/breastcanceractionguide.pdf
32. Perkins RB, Guido RS, Castle PE, et al. 2019 ASCCP Risk-based management consensus guidelines for abnormal cervical cancer screening tests and cancer precursors. Low Genit Tract Dis. 2020;24:102-131.
Approved by the NPWH Board of Directors, March 2018.
Updated and reaffirmed by the Board of Directors, November 2020.
Table revised to reflect ACOG April 2021 Updated Cervical Cancer Screening Guidelines Practice Advisory, May 2021.

