This article provides nurse practitioners with important details about effective communication strategies that promote patient-centered, shared decision making during contraceptive consultations. Specific topics that should be covered during an intrauterine device (IUD) consultation are reviewed. The article outlines essential steps for safely and accurately inserting and removing an IUD. Relevant patient education for safe and effective use of the IUD is described. Simulation resources are included that can be used for practicing techniques, in addition to improving and refining IUD placement and removal skills.
Key words: simulation, intrauterine device, shared decision making, contraception consult
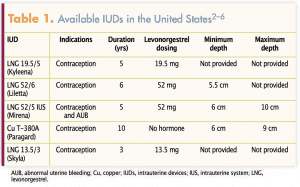
Long-acting reversible contraceptive (LARC) methods such as intrauterine devices (IUDs) are considered the most reliable reversible form of contraception. Fewer than 1 in 100 women become pregnant within 1 year of IUD placement.1 In the United States, there is one copper-containing IUD and four IUDs contain levonorgestrel (LNG): one with 13.5 mg, another with 19.5 mg, and two with 52 mg (Table 1).2–6 Regardless of parity, each IUD is considered a safe, effective contraceptive option for individuals who do not have a contraindication identified.1 The copper IUD is approved for 10 years, and the LNG IUDs are approved for 3, 5, and 6 years, respectively.2–6
Nurse practitioners (NPs) providing contraception should have knowledge of indications and contraindications, appropriate preparation and skills to perform IUD insertion and removal procedures safely and competently, an ability to recognize and address complications and side effects, and competence in providing patient education for safe and effective use. Equally important for the NP is to have the knowledge and skills to engage with the patient in a contraception consultation that promotes patient-centered, shared decision making (SDM) regarding choice of contraceptive method.
This article provides an evidence-based review of the IUDs currently available in the United States. Information about effective communication with patients using a patient-centered, shared decision approach is included. Details about specific topics that should be reviewed with patients considering an IUD are reviewed. Safe and accurate steps for inserting and removing an IUD are outlined.
Contraception consultation
A contraception consultation is conducted to assess pregnancy intentions, to understand preferences for contraceptive and noncontraceptive benefits, provide tailored information about contraceptive options, and discuss factors that may influence contraceptive decision making. IUDs offer long-acting, highly effective, reversible contraception, and their uptake among all groups of contraceptive users has increased over the last decade.7 Tiered-effectiveness contraceptive counseling, in which healthcare providers routinely discussed the most effective methods of contraception first rather than individualizing counseling to the specific patient, was once a popular method for performing a contraceptive consult.8 For many years, LARC options were rarely offered as an option during contraceptive visits. Over time, a trend developed for LARC-is-best contraceptive counseling based on concern about population-level, high unplanned pregnancy rates, favorable changes in insurance coverage, and endorsement by trusted health organizations.8 Yet, it is critical to recognize that LARC may not be the desired or right method for every woman. In fact, the high efficacy and long duration that may be seen as benefits to some may be seen by others as reproductive coercion and disempowerment over their reproductive decisions.9
SDM is considered an ideal method for preference-sensitive decisions that are highly dependent on individual values and needs.10 It is designed to assist patients in making informed decisions through a counseling process by which the NP promotes trust, establishes rapport, and facilitates effective discussion of information while providing care and concern. During SDM, the NP contributes accurate medical knowledge while the patient provides background on their values and preferences. Together, the NP and patient collaborate to achieve a goal of making a decision that is most consistent with the patient’s preferences.10 Developing a patient-centered approach to family planning, with the patient’s preferences at the forefront, has been shown to increase contraceptive continuation and satisfaction.11 Providing patient-centered contraceptive care is essential to assisting patients with making contraceptive choices that meet their individual needs.12 The NP must provide accurate and complete information on contraceptive options while engaging with women in the decision-making process, tailoring information and recommendations based on a comprehensive assessment of women’s health status and preferences, rather than simply detailing each individual method and asking women to choose from among all of them.13 Readers can access a video provided by the American College of Obstetricians and Gynecologists (ACOG) showing SDM counseling scenarios.
IUD preprocedure consultation
Patients who choose an IUD should have the option for same day insertion rather than needing to schedule a second appointment. Prior to insertion, a detailed discussion of benefits, risks, and potential side effects should take place. Efficacy, safety, long-term protection, and immediate return to fertility after removal are contraceptive benefits women may find beneficial. There are also noncontraceptive benefits that might be important to some women. Women with menstrual issues like dysmenorrhea or heavy menstrual bleeding may benefit from using an LNG IUD. One of the LNG IUDs is specifically indicated for treatment of heavy menstrual bleeding for women who choose to use the IUD for contraception.5 Both copper and LNG IUDs have been associated with a reduction in the risk of endometrial cancer.14 Risks associated with insertion include uterine perforation, pain, bleeding, and pelvic infection.1–6 IUD use may result in increased risk of ectopic pregnancy, abortion, or preterm delivery if IUD failure occurs; increased vaginal discharge; altered menstrual patterns; increased pelvic cramping; and hormonal IUDs can be associated with ovarian cysts and amenorrhea.1–6
A thorough evaluation of the individual’s medical history and a pelvic examination should be performed prior to insertion.1 The primary purpose of this evaluation is to identify contraindications and the need for any additional screening (Box 1).1,15 Pregnancy assessment should be performed using evidence-based criteria to be reasonably certain that the patient is not pregnant (Box 2).1 NPs should routinely follow national screening guidelines for sexually transmitted infections (STIs) and cervical cancer.1 Provided that routine screening has been performed, additional screenings are not needed at the time of IUD insertion.1 However, if high-risk patients present for an IUD, screening can be performed at the time of insertion. Insertion of the IUD should not be postponed unless purulent cervicitis, indicative of an infection, is present.1,15 It is also important to assess the patient’s desire for pain prophylaxis during the preprocedure consultation.
Box 1. Contraindications for IUD use1,15
• Allergy to component of the device
• Pregnancy or suspected pregnancy
• Known or suspected cervical, uterine, or breast cancer (LNG IUDs only for breast cancer) • Acute pelvic inflammatory disease
• Postpartum sepsis within the past 3 months
• Uterine anomaly
• Unexplained vaginal bleeding or acute cervicitis
• Untreated gonorrhea or chlamydia infection
• Infected abortion within past 3 months
• Hepatic tumors or active hepatic disease (LNG IUDs only)
• Wilson’s disease (CU IUDs only)
IUD insertion procedure
Following are detailed steps the NP should perform during IUD insertion. Pay careful attention to patient safety and comfort. Check to be sure all supplies needed are in the room, check the expiration date on the package, and confirm any allergies.
Perform a bimanual examination to confirm size and shape of the patient’s uterus, along with the position of the cervix. Gently check for cervical motion tenderness, which may indicate pelvic inflammatory disease (PID) or another pelvic abnormality.
After performing the bimanual examination, change gloves and insert a speculum into the vagina to visualize the cervix. If purulent cervicitis is present, the procedure should not be undertaken until appropriate screenings have been performed.1,15 If no signs of infection are present, proceed by cleaning the cervix with an antiseptic solution.2–6
Box 2. How to be reasonably certain a woman is not pregnant1
A practitioner can be reasonably certain a woman is not pregnant by confirming she has no signs or symptoms of a pregnancy and meets any of the following criteria:
• Is ≤ 7 days after first day of normal menstrual period
• Has not had sex since first day of last normal menstrual period
• Has been consistently and correctly using reliable contraceptive method
• Is ≤ 7 days after spontaneous miscarriage or induced abortion
• Is within 4 weeks postpartum
• Is completely or almost completely breastfeeding, amenorrheic, < 6 months postpartum
Apply sterile gloves. It is helpful to have assistance for the remainder of the procedure to facilitate maintaining sterile technique. Prior to opening the IUD package, a uterine sound should be used to confirm cervical os patency and to measure uterine depth. A tenaculum can help stabilize and straighten the cervix and uterus.2–6 If the uterus is anteverted, gentle traction should be used with the nondominant hand to pull the tenaculum downward and outward away from the patient.6 If the uterus is retroverted, however, gentle traction should be applied to the tenaculum pulling it upward and outward, or it might be helpful to apply the tenaculum to the lower portion of the cervix.6 If cervical stenosis is confirmed or patency of the cervical canal cannot be established, discontinue the procedure and consult the patient about next steps that might include performing cervical dilation manually or prescribing misoprostol to help soften the cervix and dilate the canal.1 Misoprostol should only be prescribed after a recent failed IUD insertion attempt has been confirmed.1 If patency is confirmed, uterine depth should be evaluated next and adequate depth should be confirmed based on the package insert recommendations before proceeding (Table 1).2–6
The inner IUD package containing the sterile device should be removed from the outer package. The device can be unwrapped using sterile technique. Load the IUD into the insertion tube based on package directions and maintain sterile technique.
 Following IUD assembly, introduce the insertion tube through the speculum into the vagina and gently advance into the cervical os toward the uterus. If there is any difficulty advancing the tip of the insertion tube into the cervical os, avoid forcing that could lead to uterine perforation. Resistance may be due to instability of the cervix, internal cervical os stenosis, or the natural curvature in the cervix or uterus. If the tip of the inserter tube slides freely into the external cervical os, but then meets resistance, holding gentle, continuous pressure against the internal os, but not using force, may help the internal os relax and the insertion tube pass into the uterine cavity. If the insertion tube cannot be advanced with gentle pressure, consider using a cervical dilator to enlarge the internal os.
Following IUD assembly, introduce the insertion tube through the speculum into the vagina and gently advance into the cervical os toward the uterus. If there is any difficulty advancing the tip of the insertion tube into the cervical os, avoid forcing that could lead to uterine perforation. Resistance may be due to instability of the cervix, internal cervical os stenosis, or the natural curvature in the cervix or uterus. If the tip of the inserter tube slides freely into the external cervical os, but then meets resistance, holding gentle, continuous pressure against the internal os, but not using force, may help the internal os relax and the insertion tube pass into the uterine cavity. If the insertion tube cannot be advanced with gentle pressure, consider using a cervical dilator to enlarge the internal os.
Slowly advance the insertion tube toward the fundus. Release the IUD into the uterus per package insert directions for each type and remove the insertion tube from the uterus and vagina. Remove the tenaculum. Using long, sterile curved scissors, trim the IUD strings, leaving approximately 3 cm outside of the cervical os. Cut the strings perpendicular across.2–6 Avoid cutting the strings at an angle to prevent sharpness on the end.
Confirm the stability and comfort of the patient throughout the procedure but especially after insertion, observing for syncopal signs or symptoms. The patient should be assisted to an upright sitting position and assessed for a few minutes before standing up.
Postprocedure patient education
Allow the patient to see and touch the portion of the strings that were cut off. The patient can check their strings once a month if they desire. If the patient is not able to feel their strings, another contraceptive method should be used until returning to the clinic for the NP to evaluate the IUD location.
Inform the patient about common symptoms experienced after IUD insertion, including intermittent pelvic pain that may last a few days and intermittent vaginal bleeding.11 Advise the patient to inform you if any of the following are experienced: uterine cramping not resolved with a nonsteroidal anti-inflammatory drug, malodorous vaginal discharge, fever, if she cannot feel her strings, or if she observes anything else that is new and of concern.
Depending on the type of IUD inserted and the first day of the last menses, the patient may or may not need to use backup protection during the first month.1 However, condoms should always be encouraged with a new sex partner. If an LNG IUD is placed more than 7 days after menses onset, the patient should abstain from sex or use a backup method for 7 days.1 Backup contraception use is not necessary after placement of the copper IUD.1
Provide the patient with a reminder card that includes the IUD name and lot number, insertion date, and the date this IUD needs to be removed and replaced. A routine follow-up IUD examination is not necessary.1 Nevertheless, a follow-up IUD examination within 4 to 6 weeks post insertion or after the first menses following insertion is recommended on each IUD package insert.2–6 All women should be encouraged to return for an annual well-woman examination or sooner for follow-up if there are any concerns.16
Diagnosis and billing codes
The diagnosis used for the IUD insertion visit is “encounter for insertion of intrauterine contraception device.” The ICD-10 code is Z30.430, and the current procedural terminology (CPT) code for IUD insertion is 58300.11 There is a J code associated with IUD insertion to cover the cost of the device. ACOG provides a quick guide to LARC reimbursement that is available here.
Indications for IUD removal
An IUD may be removed for various reasons, the most common of which is due to uncomfortable or undesired side effects. Irregular bleeding and abdominopelvic pain are the most common reasons women request the removal of an LNG IUD.17 Heavy menstrual bleeding is the most common reason the copper IUD is removed.18
Other indications for IUD removal include desire for pregnancy, no longer requiring contraception, partial IUD expulsion, severe anemia secondary to increased bleeding, unplanned pregnancy, and development of a malignancy.19 In the case of unplanned pregnancy, the IUD should only be removed if the strings are visible.12 It is important to evaluate the location of the pregnancy, ruling out an ectopic pregnancy and advise the patient about increased risks for a spontaneous miscarriage or for preterm delivery if the IUD remains in the uterus.1 It is controversial, but a diagnosis of PID is no longer considered an indication for IUD removal.12 With PID, the NP should treat the infection according to national treatment guidelines and reassess within 48 to 72 hours.1 If no clinical improvement is confirmed, then the NP should continue antibiotics and consider removing the IUD.1 If the patient desires to have the IUD removed, the NP should remove it after starting antibiotic treatment.1
Preprocedure consultation
A brief consultation should be performed immediately prior to removing an IUD to review the rationale and ensure the patient is aware of the risks, benefits, and potential side effects including bleeding, pelvic pain, and syncope. After reviewing potential risks and benefits, obtain the patient’s signature on the consent form.
IUD removal procedure
There are detailed steps the NP should follow in the IUD removal procedure. Pay attention to patient safety and comfort. Insert a speculum into the vagina to visualize the cervix and to confirm the presence of two IUD strings protruding out of the cervical os. Securely close a ring forceps around both IUD strings, apply gentle tension, and pull the device out of the uterus and vagina. Inspect the device to ensure it is intact and then remove the speculum from the vagina. The intact device should be shown to the patient and disposed of in a red biohazard or Sharps container. The patient should be assisted to an upright sitting position and assessed before standing up.
Postprocedure patient education
Remind the patient about the most common symptoms experienced with IUD removal, which include intermittent pelvic pain, intermittent vaginal bleeding, and fainting. Advise the patient to notify you about uterine cramping that is not resolved, abnormal vaginal discharge, or fever.
Diagnosis and billing codes
The diagnosis used for an IUD removal visit is encounter for the removal of an IUD. The ICD-10 code for this diagnosis is Z30.432, and the CPT code is IUD 58301.20 The billing code for IUD removal and replacement at the same visit is Z30.433.20
Use of simulation for practice
Simulation is a cost-effective means for practicing IUD insertion and removal procedures.
The fast-paced clinical setting is not routinely an optimal environment for the novice NP or student to obtain the necessary skills and confidence to perform IUD insertion and removal.21 Simulation provides the ability to refine skills and increase confidence in a controlled, risk-free environment. Practicing with simulation can take place in a school lab setting, at IUD workshops, or in one’s own setting with supplies that are easy to obtain (Table 2). IUD manufacturers will provide IUD training inserters, devices, and uterine models at no cost. Other supplies can be purchased from medical supply companies. The supplies needed for IUD insertion and removal simulations cost less than $20 if plastic and reusable items are purchased. Online videos demonstrating IUD insertion and removal are available online from ACOG and the IUD manufacturers (Box 3). Simulation is an excellent format for learning insertion and removal techniques as outlined by the specific IUD manufacturers. The next step to ensure safe and accurate techniques is to insert and remove several IUDs under supervision of an experienced clinician until proficiency is achieved. Supervised experiences are necessary to reduce potential for errors and to learn strategies for managing challenges that may present during the procedures.
Conclusion
Preparation prior to performing IUD consults and procedures is essential for NPs. A patient-centered, shared decision approach is an essential component of the contraceptive consult. Ensuring that safe and accurate steps are followed when performing IUD procedures is another important step in this process. Simulation provides the ability to refine skills and increase confidence in a controlled, risk-free environment prior to actually inserting and removing an IUD in the clinical setting. This article summarizes important components for NPs to consider when preparing for IUD consults and procedures.
Aimee C. Holland is Associate Professor, Brandi Shah is Assistant Professor, H. Nicole Metcalf is Adjunct Clinical Instructor, and Kathleen Pridgen is Assistant Professor at the University of Alabama at Birmingham Student Health & Wellness Center in Birmingham, Alabama. The authors have no actual or potential conflicts of interest in relation to the contents of this article.
References
- Curtis KM, Jatlaoui TC, Tepper NK, et al. U.S. Selected Practice Recommendations for Contraceptive Use, 2016. MMWR Recomm Rep. 2016;65(4):1-66.
- Cooper Surgical. Paragard intrauterine copper contraceptive: Prescribing information. 2020. https://14wub23xi2gmhufxjmvfmt1d-wpengine.netdna-ssl.com/wp-content/uploads/2018/10/PARAGARD-PI.pdf.
- Bayer Healthcare Pharmaceuticals. Skyla (levonorgestrel-releasing intrauterine system): Prescribing information. 2018. https://labeling.bayerhealthcare.com/html/products/pi/Skyla_PI.pdf.
- Bayer Healthcare Pharmaceuticals. Kyleena (levonorgestrel-releasing intrauterine system): Prescribing information. 2018. https://www.kyleena-us.com/pi/.
- Bayer Healthcare Pharmaceuticals. Mirena (levonorgestrel-releasing intrauterine system): Prescribing information. 2017. https://labeling.bayerhealthcare.com/html/products/pi/Mirena_PI.pdf.
- Allergan. Liletta (levonorgestrel-releasing intrauterine system): Prescribing information. 2019. https://media.allergan.com/actavis/actavis/media/allergan-pdf-documents/product-prescribing/liletta_shi_pi.pdf.
- Kavanaugh ML, Jerman J. Contraceptive method use in the United States: trends and characteristics between 2008, 2012 and 2014. Contraception. 2018;97(1):14-21.
- Brandi K, Fuentes L. The history of tiered-effectiveness contraceptive counseling and the importance of patient-centered family planning care. Am J Obstet Gynecol. 2020;222(4S):S873-S877.
- Gomez AM, Fuentes L, Allina A. Women or LARC first? Reproductive autonomy and the promotion of long-acting reversible contraceptive methods. Perspect Sex Reprod Health. 2014;46(3):171-175.
- Elwyn G, Dehlendorf C, Epstein RM, et al. Shared decision making and motivational interviewing: achieving patient-centered care across the spectrum of health care problems [erratum Ann Fam Med. 2014;12(4):301]. Ann Fam Med. 2014;12(3):270-275.
- Schivone GB, Glish LL. Contraceptive counseling for continuation and satisfaction. Curr Opin Obstet Gynecol. 2017;29(6):443-448.
- Antell K, Deshmukh P, Brown EJ. Contraception update: intrauterine devices. FP Essent. 2017;462:20-24.
- American College of Obstetrician and Gynecologists. ACOG committee opinion no. 755: Well-woman visit. Obstet Gynecol. 2018;132(4):e181-e186.
- Adeyemi-Fowode OA, Bercaw-Pratt JL. Intrauterine devices: effective contraception with noncontraceptive benefits for adolescents. J Pediatr Adolesc Gynecol. 2019;32(5S):S2-S6.
- Curtis KM, Tepper NK, Jatlaoui TC, et al. U.S. Medical Eligibility Criteria for contraceptive use, 2016. MMWR Recomm Rep. 2016;65(3):1-103.
- Dehlendorf C. Contraception: counseling and selection. UpToDate. February 28, 2020. https://www.uptodate.com/contents/contraceptive-counseling-and-selection-for-women.
- Bahamondes L, Brache V, Meirik O, et al; WHO Study Group on Contraceptive Implants for Women. A 3-year multicentre randomized controlled trial of etonogestrel- and levonorgestrel-releasing contraceptive implants, with non-randomized matched copper-intrauterine device controls. Hum Reprod. 2015;30(11):2527-2538.
- Bateson D, Harvey C, Trinh L, et al. User characteristics, experiences and continuation rates of copper intrauterine device use in a cohort of Australian women. Aust N Z J Obstet Gynaecol. 2016;56(6):655-661.
- Cario HA, Secor RM. Advanced Health Assessment of Women: Clinical Skills and Procedures. 4th ed. New York, NY: Springer Publishing Company; 2018.
- American College of Obstetricians and Gynecologists. 2020. Long-Acting Reversible Contraception (LARC) Quick Coding Guide. https://www.acog.org/education-and-events/publications/larc-quick-coding-guide/basic-iud.
- Nakajima AK, Posner GD. Human Simulation for Women’s Health. New York, NY: Springer; 2012.
Web resources
acog.org/programs/long-acting-reversible-contraception-larc/video-series/insertion/paragard
acog.org/programs/long-acting-reversible-contraception-larc/video-series/insertion/kyleena
lilettahcp.com/iud-product-features#insertion-video
acog.org/programs/long-acting-reversible-contraception-larc/video-series/removal

